
| 8.36(t2-t1) |
| c1 |
| 8.36(t2-t1) |
| c1 |
| Q |
| n |
| c1V |
| 1000 |
| c1V |
| 1000 |
| 0.00836(t2-t1)��VkJ�� | ||
|
| 8.36(t2-t1) |
| c1 |
| 8.36(t2-t1) |
| c1 |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(2)�к��Ȳⶨ��ʵ���У��õ��IJ����������ձ����¶ȼơ�_______��_______��
(3)��������ѧ��ѧ�г��ò���������ɵ�ʵ��װ��ͼ(������Ҫ�������м���Һ������)��

��ش��������⣺
�����������ﰱ����װ����_____________(����ĸ)��
�ڼ��������ռ��������������ռ�һ�����������װ����____________ (����ĸ)��
����ʵ�����Ʊ�������ʵ���У����Գ�ȥ�������Ȼ�������������װ����___________(����ĸ)��
����������ϩ����ˮ��Ӧ�ƶ��������ʵ��װ����____________(����ĸ)��
����Cװ���У������ձ��ڵ�����������Һ����β��������������ƿ��������____________��
(4)Ϊ�Ƚ�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч����ijͬѧ�������ͼ��ʾ��ʵ�顣
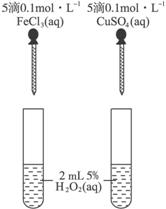
�ٿ�ͨ���۲�__________________________________________�����Եĵó����ۣ�
����Aͬѧ�����CuSO4��ΪCuCl2��Ϊ��������������_____________________________
_____________________________________________________________________��
��������Aͬѧ�ĸĽ�����������Ϊ��������θĽ���
____________________________________________________________________
___________________________________________________________________
____________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ��ɶ����и߶�3�¼�⻯ѧ�Ծ� ���ͣ������
��21�֣���1����50mL 0.55mol/L NaOH��Һ��50mL 0.25mol/L H2SO4��Һ�����к��Ȳⶨ��ʵ�飬�����Һ�ڷ�Ӧǰ����¶ȱ仯Ϊt1�桫t2�棨t2>t1��,��Ϻ���Һ�ı�����Ϊc = 4.18J����g���棩,��Һ���ܶȶ�����Ϊ1g/mL���к��ȡ�H=__________(�����ʽ�����û���)������H2SO4��Һ������ͬ���ʵ���Ũ�ȵ�CH3COOH��Һ����õġ�H______��(��ƫ����ƫС������ͬ����������ϡ���ỻ��Ũ����������ʵ�飬��õġ�H_______���ƫ����ƫС������ͬ������
��2��ij���ܻ�ѧ����N2H2�ڣ���Ԫ�ص��ӻ�����Ϊ____������ʽΪ____��һ���������ЦҼ� �� ���� �� ����
��3�������ܱ������н��п��淴Ӧ�� CO(g)��NO2(g) CO2(g)��NO(g)��(����Ӧ����)���ﵽƽ���ֻ�ı�����һ����������ƽ���Ӱ���ǣ�
CO2(g)��NO(g)��(����Ӧ����)���ﵽƽ���ֻ�ı�����һ����������ƽ���Ӱ���ǣ�
�����������������ƽ�������ƶ�(�������������)����Ӧ��������ɫ��������(��������dz�������䡱)
������������䣺��ͨ��CO2���壬ƽ���� ���ƶ�����Ӧ��������ɫ��������ͨ��N2���壬ƽ���� ���ƶ�����Ӧ��������ɫ�������ۼ��������ƽ���������ƶ���
(4)�±��Ǽ��ֳ���ȼ��(1 mol)��ȫȼ��ʱ�ų���������
| ���� | ̿��(C) | һ����̼(CO) | ����(H2) | ����(CH4) | �Ҵ�(C2H5OH) |
| ״̬ | ���� | ���� | ���� | ���� | Һ�� |
| ����(kJ) | 392.8 | 282.6 | 285.8 | 890.3 | 1 367 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013��ɶ����и߶�3�¼�⻯ѧ�Ծ� ���ͣ������
��21�֣���1����50mL 0.55mol/L NaOH��Һ��50mL 0.25mol/L H2SO4��Һ�����к��Ȳⶨ��ʵ�飬�����Һ�ڷ�Ӧǰ����¶ȱ仯Ϊt1�桫t2�� ��t2>t1��,��Ϻ���Һ�ı�����Ϊc = 4.18J����g���棩,��Һ���ܶȶ�����Ϊ1g/mL���к��ȡ�H=__________(�����ʽ�����û���)������H2SO4��Һ������ͬ���ʵ���Ũ�ȵ�CH3COOH��Һ����õġ�H______��(��ƫ����ƫС������ͬ����������ϡ���ỻ��Ũ����������ʵ�飬��õġ�H_______���ƫ����ƫС������ͬ������
��2��ij���ܻ�ѧ����N2H2�ڣ���Ԫ�ص��ӻ�����Ϊ____������ʽΪ____��һ���������ЦҼ� �� ���� �� ����
��3�������ܱ������н��п��淴Ӧ�� CO(g)��NO2(g) CO2(g)��NO(g)��(����Ӧ����)���ﵽƽ���ֻ�ı�����һ����������ƽ���Ӱ���ǣ�
CO2(g)��NO(g)��(����Ӧ����)���ﵽƽ���ֻ�ı�����һ����������ƽ���Ӱ���ǣ�

�����������������ƽ���� �� �ƶ�(�������������)����Ӧ��������ɫ��������(��������dz�������䡱)
������������䣺��ͨ��CO2���壬ƽ���� ���ƶ�����Ӧ��������ɫ�� �� ����ͨ��N2���壬ƽ���� ���ƶ�����Ӧ��������ɫ�� �� ���ۼ��������ƽ���������ƶ���
(4)�±��Ǽ��ֳ���ȼ��(1 mol)��ȫȼ��ʱ�ų���������
|
���� |
̿��(C) |
һ����̼(CO) |
����(H2) |
����(CH4) |
�Ҵ�(C2H5OH) |
|
״̬ |
���� |
���� |
���� |
���� |
Һ�� |
|
����(kJ) |
392.8 |
282.6 |
285.8 |
890.3 |
1 367 |
�ٴ������Ƕȷ�����Ŀǰ���ʺϼ�ͥʹ�õ���������ȼ����________��
��д���ܵ�ú���е�һ����̼ȼ���ȵ��Ȼ�ѧ����ʽ_______________________
�۳��ȼ��1 mol���и���ȼ�ϣ��ŷų�������̼����������________��
�ܿ���ȼ�ϴ������ޣ�������ȼ�չ����л������Ⱦ��������Դ�������ķ�չս�ԣ��ҹ��������õ���ɫ��Դ��________�ȡ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�����ʡ�߶���ѧ����ĩ���Ի�ѧ�Ծ� ���ͣ�ʵ����
��13�֣�
��1��������ͭ������ᾧˮ�����IJⶨʵ���У�������������Ҫ���� �Ρ�
��2���к��Ȳⶨ��ʵ���У��õ��IJ����������ձ����¶ȼơ� �� ��
��3����������ѧ��ѧ�г��ò���������ɵ�ʵ��װ��ͼ(������Ҫ�������м���Һ������)��

��ش��������⣺
�����������ﰱ����װ����_______________(����ĸ)��
�ڼ��������ռ��������������ռ�һ�����������װ����_______________(����ĸ)��
����ʵ�����Ʊ�������ʵ���У����Գ�ȥ�������Ȼ�������������װ����__________ (����ĸ)��
����������ϩ����ˮ��Ӧ�ƶ��������ʵ��װ����__________(����ĸ)��
����Cװ���У������ձ��ڵ�����������Һ����β��������������ƿ��������____��
��4��Ϊ�Ƚ�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч����ijͬѧ�������ͼ��ʾ��ʵ�顣

�ٿ�ͨ���۲� �����Եĵó����ۣ�
����Aͬѧ�����CuSO4��ΪCuCl2��Ϊ�������������� ��
��������Aͬѧ�ĸĽ�����������Ϊ��������θĽ��� ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com