
ij�¶�ʱ�����ܱ������У�X��Y��Z��������Ũ�ȵı仯��ͼ����ʾ���������������䣬���¶ȷֱ�ΪT1��T2ʱ��Y�����������ʱ���ϵ��ͼ����ʾ�������н�����ȷ����
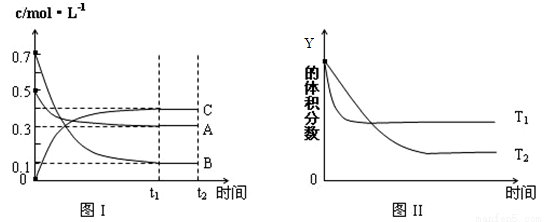
A���÷�Ӧ���Ȼ�ѧ����ʽΪ��X(g)+3Y(g) 2Z(g������H<0
2Z(g������H<0
B���������������䣬�����¶ȣ������淴Ӧ���ʾ�����X��ת��������
C���ﵽƽ����������������䣬��С�����ƽ�����淴Ӧ�����ƶ�
D���ﵽƽ����������������䣬ͨ��ϡ�����壬ƽ��������Ӧ�����ƶ�
 ̽���빮�̺��Ͽ�ѧ����������ϵ�д�
̽���빮�̺��Ͽ�ѧ����������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2016�������и�����ѧ�ڵ������¿����ۻ�ѧ�Ծ��������棩 ���ͣ������
[��ѧ��ѡ�� ���ʽṹ������]
�ڶ�������̼���������ǹ����������ʵ�������ҪԪ�أ�������������Ҳ������Ҫ��Ӧ�á�
��1���ڶ������У���һ�����ܴ���B��N֮���Ԫ����_______�֡�
��2��ijԪ��λ�ڵ������ڢ��壬���̬ԭ�ӵ�δ�ɶԵ��������̬̼ԭ�ӵ�δ�ɶԵ�������ͬ�������̬ԭ�ӵļ۲�����Ų�ʽΪ____________��
��3��NH3���ӵ�VSEPRģ��Ϊ_________��SO3���ӵĿռ乹��Ϊ_________��
��4����ϩͪ��CH2=C=O����һ����Ҫ���л��м��壬����CH3COOH�ں�����C2H5O��3P=O�����¼�����H2O�õ�����ϩͪ������̼ԭ���ӻ����������_________��CH3COOH�ķе��HCOOCH3�ĸߣ�����Ҫԭ����_______________________________��
��5 CH2=CHCN���Ʊ����ڵ�ԭ�ϣ�������ЦҼ��ͦм��ĸ���֮��Ϊ��_________������������ȣ���
��6��[LiC60]PF6��NaCl����������Ƶľ����ṹ��[LiC60]+��ʾ1��Li+λ��1��C60�ڲ�����[LiC60]PF6�����а����Ļ�ѧ����__________������ĸ����
A ������ B ���Ӽ� C ���Լ� D �Ǽ��Լ� E ���»���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ���㽭ʡ��һ�����в��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
�������滯ѧ�Լ��ķ�����ȷ����
A����ʢҺ����Լ�ƿ�м�ˮ���γɡ�ˮ�⡱���Լ�����ӷ�
B�������й�Ƭ���廯����������ɫ�Լ�ƿ��
C���ռ���Һ���ڴ�ĥ�ڲ��������Լ�ƿ��
D�����Ƶ���ˮ��������ɫ���ƿ�У�������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�����ʡ�߶������У��ģ���ѧ�Ծ��������棩 ���ͣ�ѡ����
���з�Ӧ�����ӷ���ʽ����ȷ����
A���������ᷴӦ��2Fe + 6H+ = 2Fe3+ + H2��
B������������Һ�����ᷴӦ��OH- + H+ = H2O
C��п������ͭ��Һ��Ӧ��Zn + Cu2+ = Zn2+ + Cu
D���Ȼ�����Һ��ϡ���ᷴӦ��Ba2+ + SO42- = BaSO4��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�����ʡ��һ�ϰ��ڲ��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
ij��Ӧ������ʽ��ʾ�� xR2+ + yH+ + O2 == mR3+ + nH2O �� ��m��ֵΪ
A��2x B��4 C��y/2 D��7
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�꺣��ʡ��һ��12�¶ο���ѧ�Ծ��������棩 ���ͣ������
�������ʣ���H2O ��NaCl ��H2SO4 ��Ca(OH)2 ��Fe2O3 ��CuSO4��5H2O ��HNO3 ��AgNO3 ��NaHCO3 �����������������________�����ڼ����________�����������________�������ε���______
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ��ӱ�ʡ�߶���12���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
����������ȷ����( )
A�����Ӿ����У�ֻ�������Ӽ��������ܴ���������ѧ��
B������Ԫ��(O��S��Se��Te)���⻯��ķе���������
C��NaHSO4��Na2O2�����е��������Ӹ����Ⱦ�Ϊ1��1
D�������Ӳ�ȣ����ʯ>̼����>ʯӢ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ��ӱ�ʡ��һ�ϵ������¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
���з�Ӧ�����ӷ���ʽ��д��ȷ����
A���Ȼ�����Һ�м�������İ�ˮ��Al3����4NH3��H2O = AlO��4NH��2H2O
B��Ũ�ռ���Һ�м�����Ƭ��Al��2OH? = AlO��H2��
C����ŨNaOH��Һ�ܽ�Al2O3��2OH?��Al2O3 = 2AlO��H2O
D����NaAlO2��Һ��ͨ�������CO2��2 AlO+CO2+3H2O = 2Al(OH)3��+CO32?
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ���㽭ʡ�߶������в��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
����ʵ���ܴﵽԤ��Ŀ�ĵ��ǣ� ��
�����Ҵ���Ũ�����ȥ���������е������������NaOH��Һ��ȥ���е��������ӣ����ñ���NaHCO3��Һ��ȥCO2�е�����SO2�����ü��ȵķ�����ȡNH4Cl�����л��е������⣻���þƾ���ȡ��ˮ�еĵ⡣
A���٢� B���٢ڢܢ� C���ڢ� D���ڢۢ�
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com