
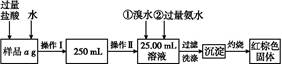
 2Fe3++2Br-£Ø2·Ö£©
2Fe3++2Br-£Ø2·Ö£©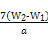 ”Į100%£Ø1·Ö£©
”Į100%£Ø1·Ö£©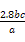 ”Į100%£Ø1·Ö£©
”Į100%£Ø1·Ö£©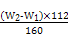 ”Į
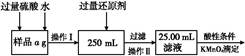
”Į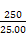 g=7£ØW2-W1£© g,ĘäÖŹĮæ·ÖŹżĪŖ
g=7£ØW2-W1£© g,ĘäÖŹĮæ·ÖŹżĪŖ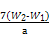 ”Į100%”£
”Į100%”£ ”«Mn
”«Mn ,ŌņŌѳʷ֊n£ØFe£©=n£ØFe2+£©=5”Įbc”Į10-3”Į
,ŌņŌѳʷ֊n£ØFe£©=n£ØFe2+£©=5”Įbc”Į10-3”Į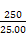 mol="0.05bc" mol,Ōņm£ØFe£©="0.05bc”Į56" g="2.8bc" g,ĘäÖŹĮæ·ÖŹżĪŖ
mol="0.05bc" mol,Ōņm£ØFe£©="0.05bc”Į56" g="2.8bc" g,ĘäÖŹĮæ·ÖŹżĪŖ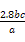 ”Į100%”£
”Į100%”£

 æŖŠÄĶÜדŌŖ²āŹŌ¾ķĻµĮŠ“š°ø
æŖŠÄĶÜדŌŖ²āŹŌ¾ķĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
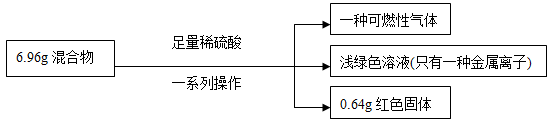
| A£®Éś³ÉĘųĢåÖŹĮæĪŖ0.2g |
| B£®Ō»ģŗĻĪļÖŠĢśµÄÖŹĮæ·ÖŹżĪŽ·Ø¼ĘĖć |
| C£®ŹµŃéÖŠ·“Ó¦µÄH2SO4ÖŹĮæĪŖ10.78g |
| D£®Ō»ģŗĻĪļÖŠĶŌŖĖŲµÄÖŹĮæ²»Ņ»¶ØĪŖ0.64g |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
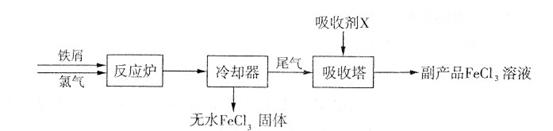
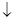 +A”£ŌņAµÄ»ÆѧŹ½ĪŖ ”£
+A”£ŌņAµÄ»ÆѧŹ½ĪŖ ”£²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®Ķعż»ÆŗĻ·“Ó¦æÉÖʵĆFeCl2ŗĶFe(OH)3 |
| B£®ĢśŹĒ½Ļ»īĘĆµÄ½šŹō£¬²»ÄÜÓĆĢśÖŹČŻĘ÷“¢“ę”¢ŌĖŹäÅØĻõĖį”¢ÅØĮņĖį |
| C£®²»ŠāøÖŹĒĢśµÄŗĻ½š£¬Ö»ŗ¬½šŹōŌŖĖŲ |
| D£®Fe3O4Ė׳ĘĢśŗģ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
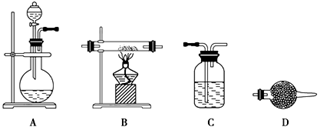
| A£®Ļ”ĮņĖį | B£®°±Ė® | C£®ĒāŃõ»ÆÄĘČÜŅŗ | D£®øßĆĢĖį¼ŲČÜŅŗ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗŹµŃéĢā
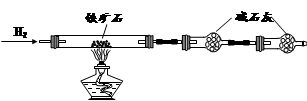

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā

²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com