
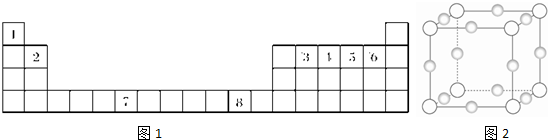
�±�����Ԫ�����ڱ���ǰ������ȥ�������Ϸ��Ŀհ��������϶��ɣ��������ߴ�Ϊ��A����A������Ӵ���������Ӧ����ѧ�����ش��������⣺
(1)eԪ��ԭ�ӽṹʾ��ͼΪ________��e��g�����γ�1��1�ͻ�����û��������ʽΪ________��������________������(����ӡ��������ۡ�)��
(2)h�ĵ�������gԪ�ص�����������Ӧ��ˮ�������Һ��Ӧ����д���÷�Ӧ�����ӷ���ʽ________��
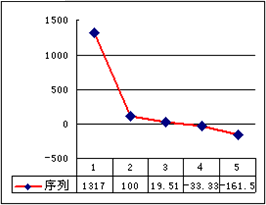
(3)��ͼ�DZ�ʾ��������8��Ԫ�ص��ʵ��۵�(��)����ͼ����֪���Ρ�1������Ar�����������Ρ�8������________(�ѧʽ)���ж�������________��
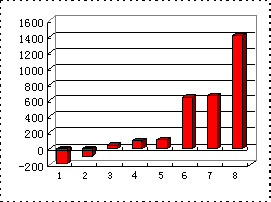
(4)b��c��d��e��f����Ӧ������⻯��ķе�(��)ֱ������ͼ(��ͼ)�����С�5�����⻯���������________����֪cԪ������⻯��8 g�ڿ�������ȫȼ������Һ̬ˮʱ���ų�445.15 kJ�������÷�Ӧ���Ȼ�ѧ����ʽΪ��________��
 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| �� | |||||||||||||||||
| �� | �� | �� | �� | �� | |||||||||||||
| �� | �� | ||||||||||||||||
| �� | �� |

| M | ||
4
|
| M | ||
4
|
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 �±�ΪԪ�����ڱ���һ���֣������Ԫ�آ�-���ڱ��е�λ�ã��û�ѧ����ش��������⣺
�±�ΪԪ�����ڱ���һ���֣������Ԫ�آ�-���ڱ��е�λ�ã��û�ѧ����ش��������⣺| �� ���� |
IA | 0 | 1 | �� | ��A | ��A | ��A | ��A | ��A | ��A | 2 | �� | �� | �� | 3 | �� | �� | �� | �� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com