
��14�֣���������ͭ���輰��Ͻ�������������������Ź㷺��Ӧ�á���ش������й����⡣
��1��Ŀǰ��ұ�������ȴ�99��9999�����������й��ڴ����������������
������ĸ����
| A��Ӳ�ȱȸ�С���۵�ȸָ� | B�����������ᷴӦ |
| C���벻��ֳɷ���ͬ | D�������Ũ�����жۻ� |
 6Cu+SO2�����÷�Ӧ��������������������
6Cu+SO2�����÷�Ӧ�������������������� 
��14�֣�ÿ��2�֣�(1)BC
(2)10Al+3V2O5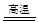 10V+5Al2O3
10V+5Al2O3
(3)��2Cu+O2+H2O+CO2=Cu2(OH)2CO3
�� Cu2O��Cu2S
��2H2SO4+2Cu+O2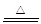 2CuSO4+2H2O
2CuSO4+2H2O
��ȡ��ͬ��������ͭ���������٣��������ж�����
(4)2C+SiO2 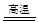 Si+2CO
Si+2CO
���������������1��A�������ȺϽ���۵�ߣ�Ӳ��С����ȷ��B���������ᷴӦ������C������ֵijɷ��г������⣬�����зǽ���̼���ɷֲ�ͬ������D���������Ũ�����з����ۻ�����ȷ��E���ߴ�����Ϊ����̼����ʹ�ڳ�ʪ������Ҳ�����γ�ԭ��ض����⣬��ȷ����ѡBC��
��2��V2O5��Al��Ӧұ��������ѧ����ʽ��10Al+3V2O5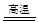 10V+5Al2O3
10V+5Al2O3
��3����ͭ������е�������ˮ��������̼��Ӧ���ɼ�ʽ̼��ͭ����ѧ����ʽΪ2Cu+O2+H2O+CO2=Cu2(OH)2CO3
��������ԭ��Ӧ����������ָԪ�ػ��ϼ۽��ͣ��õ��ӵ����ʣ�2Cu2O+Cu2S 6Cu+SO2���У�Ԫ�ػ��ϼ۽��͵���Cu��������������Cu2O��Cu2S��
6Cu+SO2���У�Ԫ�ػ��ϼ۽��͵���Cu��������������Cu2O��Cu2S��
��ͭ��������ϡ�����ڼ��������·�Ӧ��������ͭ��ˮ����ѧ����ʽΪ2H2SO4+2Cu+O2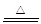 2CuSO4+2H2O���˷���ֱ����Ũ������ͭ��Ӧ���������Ե��ŵ㣺��ȡ��ͬ��������ͭ���������٣��������ж��������Ⱦ������
2CuSO4+2H2O���˷���ֱ����Ũ������ͭ��Ӧ���������Ե��ŵ㣺��ȡ��ͬ��������ͭ���������٣��������ж��������Ⱦ������
��4��ʯӢɰ����Ҫ�ɷ��Ƕ������裬��̼�ڸ��������·�Ӧ���ɹ赥�ʺ�һ����̼����ѧ����ʽ:
2C+SiO2 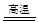 Si+2CO
Si+2CO
���㣺������������ͭ�����ʼ��Ʊ����ֹ���Ʊ�����ѧ����ʽ����д


 �����߿����ϵ�д�
�����߿����ϵ�д� �㾦�½̲�ȫ�ܽ��ϵ�д�
�㾦�½̲�ȫ�ܽ��ϵ�д� Сѧ�̲���ȫ���ϵ�д�
Сѧ�̲���ȫ���ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(15��)�ռ�(NaOH)�ʹ���(Na2CO3)�����ᡢ��������Ტ��Ϊ��ҵ�ϵġ������������Ҫ��ش��������⣺
��1����������й�NaOH��Һ�μӷ�Ӧ�����ӷ���ʽ
����NaOH��Һ�м�����Ƭ��_______________________��
����NaOH��Һ�еμ������Ȼ�����Һ___________________��
�۶���������NaOH��Һ����������ԭ��Ӧ��___________________��
�����ȵ�NaOH��Һϴ��մ�����ʵ��Թܣ�___________________��
��2��������̼���ƺ�̼�����Ƶ����ʱȽϣ��á�������������������գ�
�����ȶ��ԣ�Na2CO3_______NaHCO3��
����ϡ���ᷴӦ������Na2CO3_______NaHCO3��
�����ʵ�����ͬʱ��������������ʵ�����Na2CO3_______NaHCO3��
��3����10 mL 1 mol/L�Ĵ�����Һ�У����Ͻ��貢��μ���10 mL 1.5 mol/L���ᣬ��ȫ��Ӧ���ڱ�״�������ɶ�����̼�����Ϊ____________L��
��4��̼�����ڹ�ҵ�Ͼ��й㷺�����ã�̼���ƿ����������й�ҵ������Ҫԭ�ϵ���_____(����)
a������ b��ˮ�� c����ֽ d����ˮ��ȡþ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ijʵ��С���ù�ҵ�������壨��Ҫ�ɷ�ΪCu2S��Fe2O3���Ʊ��й����ʣ�������������ͼ��ʾ����ش�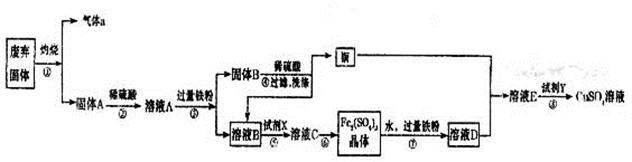
��1������a�Ļ�ѧʽΪ ��
��2����ҺB���������ữ���ټ�������������X�õ���ҺC��д���÷�Ӧ�����ӷ���ʽ ��
��3���Ʊ�����ͭ��Һ�������£�O2��ͭ�ۺ�ϡ����������һ�𣬼�������Ӧ����������ҺD���漴��������ͭ�����������Ϸ���FeSO4��ͭ������������á�
A.��һ����Ӧ�����ӷ���ʽΪ��4Fe2����O2��4H��=4Fe3����2H2O����ڶ�����Ӧ�Ĺ��ӷ���ʽΪ ��
B.�߲����У�����Fe2(SO4)3��ҺʱӦע�� ��
��4���������Ŀ���ǵõ��ϴ�������ͭ��Һ�����������Լ�Y����pH����Ԫ��ȫ������������Ũ��С��10��5mol/L����Ȼ���ٹ��ˣ�Ũ�����ᾧ�ȣ���pH���ٵ���Ϊ_____��
��֪��Ksp[Cu(OH)2]��1��10��22��Ksp[Fe(OH)2] ��1��10��16��Ksp[Fe(OH)3] ��1��10��38
��5����ѧ�ҷ���������Cu2O��̫���������¿��Դ��ֽ�ˮ��
A.һ���¶��£���2L�ܱ������м�������Cu2O��ͨ��2molˮ�������������·�Ӧ��
2H2O(g)��2H2(g)��O2(g) ��H����484kJ/mol
20minĩ���n(O2)��0.16mol�������ʱ��ķ�Ӧ���ʦ�(H2)��_________�����¶��£��˷�Ӧ��ƽ�ⳣ������ʽK��___________________��
B.��֪��2Cu2O(s)��O2(g)��4CuO(s) ��H����292kJ/mol
2C(s)��O2(g)��2CO(g) ��H����221kJ/mol
��д��̿�ۻ�ԭCuO(s)�Ʊ�Cu2O(s)���Ȼ�ѧ����ʽ_________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
��1���֣�
һλͬѧ�ڸ�ϰʱ��������һ��ϰ�⣺ij��ɫ��Һ�п��ܺ��С�H+��OH-��Na+��NO3-�����������ۺ�ֻ����H2���ʸ���ɫ��Һ���ܴ��������ļ������ӡ�
��1���������۲���H2��˵��������______��������ԡ���ԭ�ԡ�����
��2����ͬѧ��������H+�������ڣ���NO3-�Ͳ��ܴ������ڡ�
���ʵ��֤ʵ���£�
| װ �� | �� �� |
 | ��. ʵ���ʼ��δ���������� ��. ��һ������������ݣ�Һ���Ϸ���dz��ɫ ��. �Թܱ��ȣ���Һ���� |
| ʵ �� | �� �� | �� �� |
| ʵ��1 | ��ʪ��KI��������ֽ���ڿ����� | δ���� |
| ʵ��2 | ��ʪ��KI��������ֽ����dz��ɫ���� | ��ֽ���� |
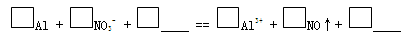
| װ �� | �� �� |
 | ��. ʵ���ʼ��δ���������� ��. ��һ������������ݣ��д̼�����ζ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
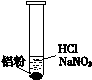
��ˮAlCl3���������������л��ϳɵĴ����ȣ���ҵ����������(��Ҫ�ɷ���Al2O3��Fe2O3,������������SiO2 )��ʯ�ͽ�(��Ҫ�ɷ���C)Ϊԭ���Ʊ���ˮAlCl3��2Al2O3��6Cl2===4AlCl3��3O2
�ش��������⣺
(1)Cl���ĵ���ʽΪ ________��Al��Ԫ�����ڱ����е�λ����________��
(2)�����м���ʯ�ͽ�����Ŀ����_____________________________________��
(3)���뽹̿��Ļ�ѧ��Ӧ�ɱ�ʾΪAl2O3��C��Cl2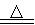 AlCl3��X�������ʵ��ȷ������X�ijɷ֣�
AlCl3��X�������ʵ��ȷ������X�ijɷ֣�
____________________________________________________________��
(4)���ᴿAlCl3�ֲ�Ʒʱ��������������ۣ���ʹ�۵�ϵ͵�FeCl3ת��Ϊ�۵�ϸߵ�FeCl2���Ӷ�������AlCl3�л��������Ȼ���÷�Ӧ�Ļ�ѧ����ʽΪ _____________________________________________________________��
(5)Ϊ�ⶨ�Ƶõ���ˮAlCl3��Ʒ(��FeCl3����)�Ĵ��ȣ���ȡ16.25 g��ˮAlCl3��Ʒ�����ڹ�����NaOH��Һ�����˳�����������ᆳϴ�ӡ����ա���ȴ�����أ�������������Ϊ0.32 g��
�ٹ����漰�����ӷ���ʽ__________________________��___________��
��AlCl3��Ʒ�Ĵ���Ϊ__________________________________��
(6)��������Ϊԭ�Ͽ���ͨ������;���ᴿ������
��д����Һ�������ʵĻ�ѧʽ______________________________��
��д����Һ���м��������ˮ�õ�Al(OH)3�����ӷ���ʽ__________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
��ҵ�����÷���м�����������������������ȣ�������ʽ������[Fe��OH��SO4]�Ĺ����������£�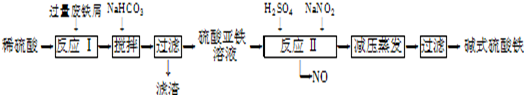
��֪������������������������ʽ����ʱ��Һ��pH���±���
| ������ | Fe��OH��3 | Fe��OH��2 | Al��OH��3 |
| ��ʼ���� | 2.3 | 7.5 | 3.4 |
| ��ȫ���� | 3.2 | 9.7 | 4.4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
�����仯�����ڹ��õķ�չ��������Ҫ���á�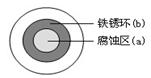
��1����֪��4Fe(s)��3O2(g)=2Fe2O3(s) ��H����1641.0kJ��mol-1 C(ʯī)��1/2O2(g)=CO(g) ��H����110.5 kJ��mol-1��Fe2O3(s)��3C(ʯī)=2Fe(s)��3CO(g)�ġ�H�� kJ��mol-1��
��2�����ڳ�ʪ�Ŀ����������绯ѧ��ʴ��ijͬѧ��NaCl��Һ����һ�����������������ϣ�һ��ʱ�����Һ�θ��ǵ�Բ��������(a)�ѱ���ʴ���䰵����Һ�������γ���ɫ���(b)����ͼ��ʾ��Һ�α�Ե�� �����������������������缫��ӦʽΪ ��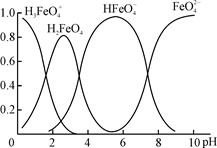
��3�����ѺϽ���һ�ֳ��õIJ���ֲ��ϣ�ijͬѧ��̽���úϽ������ʱ��������TiO2+��Fe3+��Һ�м�����м����Һ����ɫ���ù����з����ķ�Ӧ�У�
��2TiO2��(��ɫ)��Fe��4H��=2Ti3��(��ɫ)��Fe2����2H2O
��Ti3��(��ɫ)��Fe3����H2O=TiO2��(��ɫ)��Fe2����2H��
�� ��
��4���ٸ������(K2FeO4)��һ��������ˮ��������FeO��ˮ��Һ�еĴ�����̬����ͼ��ʾ���������ʾ��������̬�ķ����ֲ���
����˵������ȷ���� ��������ĸ��
A��������Һ�������α仯����Ԫ�ض���4�ִ�����̬
B����pH��10��������Һ�м�������pH��2��HFeO�ķֲ�����������
C����pH��6��������Һ�м�KOH��Һ��������Ӧ�����ӷ���ʽΪ��
HFeO��OH��=FeO��H2O
��K2FeO4����ˮ��ų�һ����ɫ��ζ���壬��ɱ������������ˮ�е��������ʵ�ԭ���������ӷ���ʽ��ʾΪ ��
��5����һ������Fe��FeO��Fe3O4�Ļ�����м���100 mL 1 mol��L-1�����ᣬǡ��ʹ�������ȫ�ܽ⣬�ų�224 mL����״�������壬����KSCN��Һ���Ժ�ɫ������������CO�ڸ����»�ԭ��ͬ�����Ĵ˻����ɵ��� g��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij�������д�����CuS����������������������ʡ�ʵ�������Ըÿ���Ϊԭ���Ʊ�CuCl2��2H2O���壬�������£�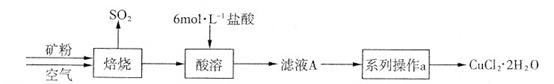
��1����ʵ�����У�����37��(�ܶ�Ϊ1��19 g��mL-1)����������500 mL 6 mol��L-1�����ᣬ��Ҫ����������Ͳ���ձ����������⣬���� �� ��
��2��������ʵ���������ϵ�в���a��������ʵ������У�����Ҫ���� (�����и��������)��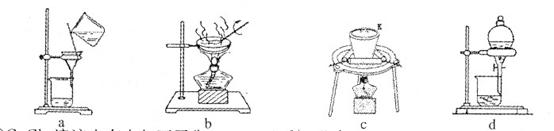
��CuCl2��Һ�д�������ƽ�⣺Cu(H2O)42+(��ɫ)+4Cl-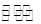 CuCl42-(��ɫ)+4H2O��
CuCl42-(��ɫ)+4H2O��
����ʵ��֤����ҺA(��ɫ)�д�������ƽ�⣬����ҺA�⣬�����Լ��У�����Ҫ���� (�����и��������)��
a��FeCl3���� b��CuCl2���� c������ˮ
��3��ij��ѧС������ʵ�������о�CuS���յķ�Ӧ���̣��������ϵ�֪�ڿ��������±���CuSʱ�����������仯����SO2������������ͼ��ʾ��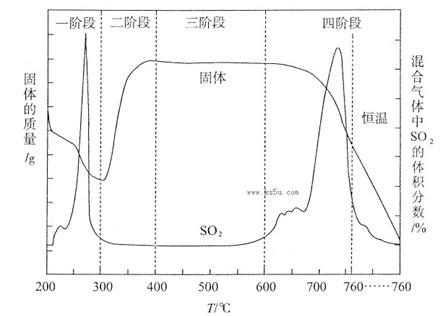
��CuS�����ڱ��չ����У���Cu2S��CuO��CuSO4��CuSO4��CuO���ɣ�ת��˳��Ϊ��
�ڢٲ�ת����Ҫ��200��300oC��Χ�ڽ��У��ò�ת���Ļ�ѧ����ʽΪ ��
��300��400oC��Χ�ڣ����������������ӵ�ԭ���� ����ͼ��ʾ�����У�CuSO4�������ȶ����ڵĽ��� (�����и��������)��
a��һ�� b������ c������ d���Ľ�
�۸û�ѧС���������װ��ģ��CuS�����������б��յ��ĽεĹ��̣�����֤��������ΪSO2��O2�Ļ���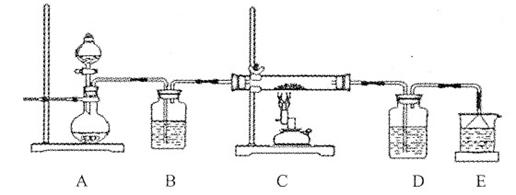
a��װ����װ��ɺ�Ӧ�������е�һ������� ��
b����Dװ���в�����ɫ����ʱ������˵�����Ľ���������ΪSO2��O2�Ļ�������Ϊװ��D��ԭ��ʢ�е���ҺΪ ��Һ��
c����ԭCuS����������Ϊl0��0 g����ʵ������У������¶���760oC���ҳ������ȣ���������ַ�Ӧ��ʯӢ�����������ù��������Ϊ8��0 g����ԭ������CuS����������Ϊ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com