
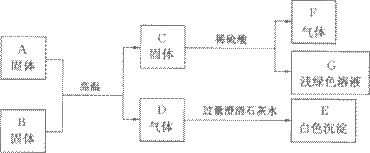
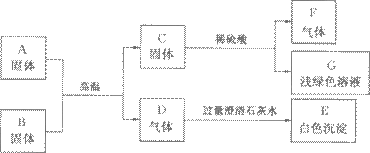
AŹĒij¹¤³§·Ļ¾É»śŠµ±ķĆęÉĻŗģÉ«¹ĢĢåµÄÖ÷ŅŖ³É·Ö£¬B”¢C”¢F¶¼ŹĒµ„ÖŹ£¬D”¢F¶¼ŹĒĪŽÉ«ĪŽĪ¶µÄĘųĢ壬ĖüĆĒÖ®¼äµÄĻą»„×Ŗ»Æ¹ŲĻµČēĻĀĶ¼£ŗ
Ēė»Ų“š£ŗ
(1)Š“³öĻĀĮŠĪļÖŹµÄ»ÆѧŹ½£ŗ
C____________£¬F___________,B__________,GµÄČÜÖŹ____________”£
(2)Š“³öĶ¼ÖŠĘųĢåDŗĶ³ĪĒåŹÆ»ŅĖ®·“Ó¦ĻąÓ¦µÄ»Æѧ·½³ĢŹ½£ŗ______________________

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
| ĪĀ¶Č£Ø”ę£© | 20 | 40 | 60 | 80 | 100 |
| ŹÆøą | 0.32 | 0.26 | 0.15 | 0.11 | 0.07 |
| µØ·Æ | 32 | 44.6 | 61.8 | 83.8 | 114 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
| MgO | MgCl2 | |
| ČŪµć/”ę | 2852 | 714 |
| ·Šµć/”ę | 3600 | 1412 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
AŹĒij¹¤³§·Ļ¾É»śŠµ±ķĆęÉĻŗģÉ«¹ĢĢåµÄÖ÷ŅŖ³É·Ö£¬B”¢C”¢F¶¼ŹĒµ„ÖŹ£¬D”¢F¶¼ŹĒĪŽÉ«ĪŽĪ¶µÄĘųĢ壬ĖüĆĒÖ®¼äµÄĻą»„×Ŗ»Æ¹ŲĻµČēĻĀĶ¼£ŗ
Ēė»Ų“š£ŗ
(1)Š“³öĻĀĮŠĪļÖŹµÄ»ÆѧŹ½£ŗ
C____________£¬F___________,B__________,GµÄČÜÖŹ____________”£
(2)Š“³öĶ¼ÖŠĘųĢåDŗĶ³ĪĒåŹÆ»ŅĖ®·“Ó¦ĻąÓ¦µÄ»Æѧ·½³ĢŹ½£ŗ______________________
²éæ““š°øŗĶ½āĪö>>
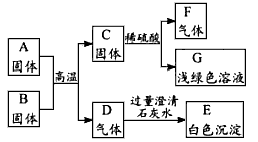
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗĶ¬²½Ģā ĢāŠĶ£ŗĶʶĻĢā
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com