
 ijĶ¬Ń§ÓūÓĆŅŃÖŖĪļÖŹµÄĮæÅضČĪŖ0.100 0mol•L-1µÄŃĪĖį²ā¶ØĪ“ÖŖĪļÖŹµÄĮæÅØ¶ČµÄĒāŃõ»ÆÄĘČÜŅŗŹ±£¬Ń”Ōń·ÓĢŖ×÷ÖøŹ¾¼Į£®ĒėĢīŠ“ĻĀĮŠæÕ°×£ŗ
ijĶ¬Ń§ÓūÓĆŅŃÖŖĪļÖŹµÄĮæÅضČĪŖ0.100 0mol•L-1µÄŃĪĖį²ā¶ØĪ“ÖŖĪļÖŹµÄĮæÅØ¶ČµÄĒāŃõ»ÆÄĘČÜŅŗŹ±£¬Ń”Ōń·ÓĢŖ×÷ÖøŹ¾¼Į£®ĒėĢīŠ“ĻĀĮŠæÕ°×£ŗ| µĪ¶Ø | “ż²āČÜŅŗ Ģå»ż/mL | ±ź×¼ĖįĢå»ż | |
| µĪ¶ØĒ°µÄæĢ¶Č/mL | µĪ¶ØŗóµÄæĢ¶Č/mL | ||
| µŚŅ»“Ī | 10.00 | V1 | V2 |
| µŚ¶ž“Ī | 10.00 | 4.10 | 21.10 |
| µŚČż“Ī | 10.00 | 0.40 | 17.60 |
·ÖĪö £Ø1£©Ėį¼īÖŠŗĶµĪ¶ØŹ±£¬ŃŪ¾¦ŅŖ×¢ŹÓ׶ŠĪĘæÄŚČÜŅŗµÄŃÕÉ«±ä»Æ£»µĪ¶ØÖÕµćŹ±ČÜŅŗŃÕÉ«ÓÉŗģÉ«Ķ»±äĪŖĪŽÉ«£»
£Ø2£©øł¾Żc£Ø“ż²ā£©=$\frac{c£Ø±ź×¼£©”ĮV£Ø±ź×¼£©}{V£Ø“ż²ā£©}$·ÖĪö²»µ±²Ł×÷¶ŌV£Ø±ź×¼£©”ĮµÄÓ°Ļģ£¬ŅŌ“ĖÅŠ¶ĻÅØ¶ČµÄĪó²ī£»
£Ø3£©µĪ¶Ø¹ÜµÄŠ”æĢ¶ČŌŚÉĻ·½£¬×¼Č·¶ČĪŖ0.01mL£»
£Ø4£©Īó²ī½Ļ“óµÄŹµŃ鏿¾ŻÓ¦ÉįČ„£¬ŌŁĖć³öŗĻĄķŹµŃéĖłĻūŗıź×¼ŅŗµÄĘ½¾łÖµ£¬øł¾Żc£Ø“ż²ā£©=$\frac{c£Ø±ź×¼£©”ĮV£Ø±ź×¼£©}{V£Ø“ż²ā£©}$¼ĘĖć“ż²āŅŗĒāŃõ»ÆÄʵÄÅØ¶Č£®
½ā“š ½ā£ŗ£Ø1£©Ėį¼īÖŠŗĶµĪ¶ØŹ±£¬ŃŪ¾¦ŅŖ×¢ŹÓ׶ŠĪĘæÄŚČÜŅŗµÄŃÕÉ«±ä»Æ£¬µĪ¶ØÖÕµćŹ±ČÜŅŗŃÕÉ«ÓÉŗģÉ«Ķ»±äĪŖĪŽÉ«£¬ĒŅ°ė·ÖÖÓÄŚ²»ĶŹÉ«£¬
¹Ź“š°øĪŖ£ŗ׶ŠĪĘæÖŠČÜŅŗŃÕÉ«µÄ±ä»Æ£»ŗģ£»ĪŽ£»
£Ø2£©A£®ĖįŹ½µĪ¶Ø¹ÜĪ“ÓƱź×¼ŃĪĖįČÜŅŗČóĻ“¾ĶÖ±½Ó×¢Čė±ź×¼ŃĪĖįČÜŅŗ£¬±ź×¼ŅŗµÄÅضČĘ«Š”£¬Ōģ³ÉV£Ø±ź×¼£©Ę«“ó£¬øł¾Żc£Ø“ż²ā£©=$\frac{c£Ø±ź×¼£©”ĮV£Ø±ź×¼£©}{V£Ø“ż²ā£©}$æÉÖŖ£¬²ā¶Øc£ØNaOH£©Ę«“󣬹ŹA“ķĪó£»
B£®µĪ¶ØĒ°Ź¢·ÅĒāŃõ»ÆÄĘČÜŅŗµÄ׶ŠĪĘæÓĆÕōĮóĖ®Ļ“¾»ŗóƻӊøÉŌļ£¬“ż²āŅŗµÄĪļÖŹµÄĮæ²»±ä£¬¶ŌV£Ø±ź×¼£©ĪŽÓ°Ļģ£¬øł¾Żc£Ø“ż²ā£©=$\frac{c£Ø±ź×¼£©”ĮV£Ø±ź×¼£©}{V£Ø“ż²ā£©}$æÉÖŖ£¬²ā¶Øc£ØNaOH£©ĪŽÓ°Ļģ£¬¹ŹB“ķĪó£»
C£®ĖįŹ½µĪ¶Ø¹ÜŌŚµĪ¶ØĒ°ÓŠĘųÅŻ£¬µĪ¶ØŗóĘųÅŻĻūŹ§£¬Ōģ³ÉV£Ø±ź×¼£©Ę«“ó£¬øł¾Żc£Ø“ż²ā£©=$\frac{c£Ø±ź×¼£©”ĮV£Ø±ź×¼£©}{V£Ø“ż²ā£©}$æÉÖŖ£¬²ā¶Øc£ØNaOH£©Ę«“󣬹ŹC“ķĪó£»
D£®¶ĮČ”ŃĪĖįĢå»żŹ±£¬µĪ¶Ø½įŹųŹ±ø©ŹÓ¶ĮŹż£¬Ōģ³ÉV£Ø±ź×¼£©Ę«Š”£¬øł¾Żc£Ø“ż²ā£©=$\frac{c£Ø±ź×¼£©”ĮV£Ø±ź×¼£©}{V£Ø“ż²ā£©}$£¬æÉÖŖc£Ø“ż²ā£©Ę«Š”£¬¹ŹDÕżČ·£»
E£®µĪ¶Ø¹ż³ĢÖŠ£¬×¶ŠĪĘæµÄÕńµ“¹żÓŚ¼¤ĮŅ£¬Ź¹ÉŁĮæČÜŅŗ½¦³ö£¬Ōģ³ÉV£Ø±ź×¼£©Ę«Š”£¬øł¾Żc£Ø“ż²ā£©=$\frac{c£Ø±ź×¼£©”ĮV£Ø±ź×¼£©}{V£Ø“ż²ā£©}$£¬æÉÖŖc£Ø“ż²ā£©Ę«Š”£¬¹ŹE×¼Č·£»
¹Ź“š°øĪŖ£ŗD”¢E£»
£Ø3£©ÓÉĶ¼æÉÖŖ£¬ĘšŹ¼¶ĮŹżĪŖ9.00mL£¬ÖÕµć¶ĮŹżĪŖ26.10mL£¬¹Ź“š°øĪŖ£ŗ9.00£»26.10£»
£Ø4£©µŚ¶ž“ĪŹµŃéĻūŗÄŃĪĖįµÄĢå»żĪŖ17mL£¬µŚČż“ĪŹµŃéĻūŗÄŃĪĖįµÄĢå»żĪŖ17.20mL£¬ŗóĮ½“ĪĻūŗÄŃĪĖįµÄĘ½¾łĢå»żĪŖ£ŗ$\frac{17.00+17.20}{2}$=17.10mL£¬ŃĪĖįÓėĒāŃõ»ÆÄĘČÜŅŗ·¢Éś·“Ó¦£ŗNaOH+HCl=NaCl+H2O£¬
ŌņÓ¦Āś×ćV£ØHCl£©”Įc£ØHCl£©=V£ØNaOH£©”Įc£ØNaOH£©£¬
c£ØNaOH£©=$\frac{0.1000mol/L”Į17.10”Į1{0}^{-3}L}{10.00”Į1{0}^{-3}L}$=0.1710mol/L£¬
¹Ź“š°øĪŖ£ŗ0.1710£®
µćĘĄ ±¾Ģāæ¼²éÖŠŗĶµĪ¶Ø£¬ĪŖøßĘµæ¼µć£¬°ŃĪÕĖį¼īÖŠŗĶµĪ¶ØµÄŹµÖŹ”¢Īó²ī·ÖĪöĪŖ½ā“šµÄ¹Ų¼ü£¬²ąÖŲ·ÖĪöÓėÓ¦ÓĆÄÜĮ¦µÄ漲飬עŅā½įŗĻ¹«Ź½·ÖĪöĪó²ī£¬ĢāÄæÄŃ¶Č²»“ó£®



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2016-2017ѧğĖÄ“ØŹ”øßŅ»ÉĻ10ŌĀŌĀæ¼»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŹµŃéĢā
ij»ÆѧŠĖȤŠ”×éÄāÓĆĻĀĶ¼×°ÖĆ²ā¶Øij“ÖŠæѳʷ֊ŠæµÄÖŹĮæ·ÖŹż”£ĖūĆĒ²éŌÄ׏ĮĻŗó»ńÖŖøĆ“ÖŠæѳʷ֊µÄĘäĖū³É·Ö²»ÄÜÓėĻ”ŃĪĖį·“Ó¦”£ĒėĢīŠ“ŅŌĻĀŹµŃé±Øøę”£
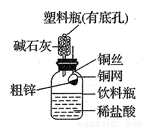
£Ø1£©ŹµŃéÄæµÄ£ŗ ”£
£Ø2£©ŹµŃé²½Öč£ŗ
¢Ł³ĘČ”10.0g“ÖŠæÖĆÓŚĶĶųÖŠ£¬°“Ķ¼Ź¾×°ÖĆ×é×°ŗó£¬³ĘµĆŅĒĘ÷ŗĶŅ©Ę·×ÜÖŹĮæĪŖ120.0g”£
¢Ś½«ĶĶų²åČė×ćĮæµÄĻ”ŃĪĖįÖŠ£¬ÓŠ¹Ų·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ ”£
¢Ū·“Ó¦ĶźČ«ŗó£¬³ĘµĆ×°ÖĆ×ÜÖŹĮæĪŖ119.8g£¬Ōņ“ÖŠæÖŠŠæµÄÖŹĮæ·ÖŹżĪŖ ”£
£Ø3£©ĪŹĢāĢ½¾æ£ŗ£ØŅŃÖŖ¼īŹÆ»ŅŹĒNaOHŗĶCaOµÄ»ģŗĻĪļ£¬³£ÓĆÓŚøÉŌļ”¢ĪüŹÕĘųĢ壩
¢ŁøĆŹµŃéČō²»ÓĆ¼īŹÆ»Ņ£¬ŌņĖł²āµĆµÄ“ÖŠæÖŠŠæµÄÖŹĮæ·ÖŹż½« £ØĢī”°Ę«“ó”±”¢”°Ę«Š””±»ņ”°ĪŽÓ°Ļģ”±£©£»
¢ŚČō½«“ÖŠæ»»³Éijŗ¬ÓŠŌÓÖŹµÄŹÆ»ŅŹÆѳʷ£ØŌÓÖŹŅ²²»ÓėĻ”ŃĪĖį·“Ó¦£©£¬ÅŠ¶ĻøĆŹµŃé·½°øÄܲ»ÄÜÓĆÓŚŹÆ»ŅŹÆѳʷ֊CaCO3ÖŹĮæ·ÖŹżµÄ²ā¶Ø£¬²¢ĖµĆ÷ĄķÓÉ____________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | ŹµŃéץÉĻµÄ¾Ę¾«µĘĒćµ¹ĮĖČ¼ÉÕĘšĄ“£¬ĀķÉĻÓĆŹŖ²¼ĘĖĆš | |
| B£® | ²»É÷½«Ėį»ņ¼īŅŗ½¦ČėŃŪÄŚ£¬Į¢¼“±ÕÉĻŃŪ¾¦£¬ÓĆŹÖČą²Į | |
| C£® | ʤ·ōÉĻ½¦ÓŠ½Ļ¶ąµÄÅØĮņĖį£¬øĻ½ōÓĆĖ®³åĻ“ | |
| D£® | ŹµŃéŹŅ½šŹōÄĘ×Å»š£¬Į¢¼“ÓĆĖ®½½Ćš |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
 ŹµŃéŹŅŠčŅŖ0.1mol/LNaOHČÜŅŗ450mlŗĶ0.51mol/LĮņĖįČÜŅŗ500ml£¬øł¾ŻÕāĮ½ÖÖČÜŅŗµÄÅäÖĘĒéæö»Ų“šĻĀĮŠĪŹĢā£ŗ
ŹµŃéŹŅŠčŅŖ0.1mol/LNaOHČÜŅŗ450mlŗĶ0.51mol/LĮņĖįČÜŅŗ500ml£¬øł¾ŻÕāĮ½ÖÖČÜŅŗµÄÅäÖĘĒéæö»Ų“šĻĀĮŠĪŹĢā£ŗ²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | 2øö¼×»ł£¬æÉÄÜÉś³É4ÖÖŅ»ĀČ“śĪļ | B£® | 3øö¼×»ł£¬æÉÄÜÉś³É3ÖÖŅ»ĀČ“śĪļ | ||
| C£® | 3øö¼×»ł£¬æÉÄÜÉś³É5ÖÖŅ»ĀČ“śĪļ | D£® | 4øö¼×»ł£¬æÉÄÜÉś³É4ÖÖŅ»ĀČ“śĪļ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | S$\stackrel{O_{2}/µćČ¼}{”ś}$SO3$\stackrel{H_{2}O}{”ś}$H2SO4 | |
| B£® | NH3$”ś_{“߻ƼĮ£¬”÷}^{O_{2}}$N2$\stackrel{O_{2}£¬H_{2}O}{”ś}$HNO3 | |
| C£® | SiO2$\stackrel{C/øßĪĀ}{”ś}$Si$\stackrel{Cl_{2}”¢øßĪĀ}{”ś}$SiCl4$\stackrel{H_{2}”¢øßĪĀ}{”ś}$Si | |
| D£® | ŗ£Ė®$\stackrel{ŹģŹÆ»Ņ}{”ś}$Mg£ØOH£©2 $\stackrel{µē½ā}{”ś}$Mg |
²éæ““š°øŗĶ½āĪö>>
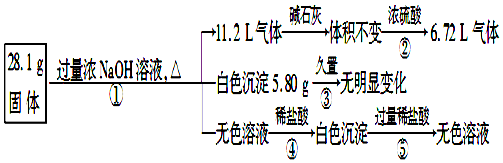
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ½ā“šĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
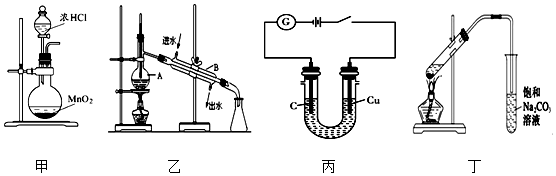
| A£® | ĄūÓĆ×°ÖĆ¼×ÖĘČ”ĀČĘų | |
| B£® | ĄūÓĆ×°ÖĆŅŅ·ÖĄėŅŅĖį£Ø·Šµć£ŗ118”ę£©ÓėŅŅĖįŅŅõ„£Ø·Šµć£ŗ77.1”ę£© | |
| C£® | ĄūÓĆ×°ÖƱūæɵē½āŹ³ŃĪĖ®ÖĘĀČĘųŗĶĒāĘų | |
| D£® | ĄūÓĆ×°Öƶ”ÖĘČ”ŅŅĖįŅŅõ„ |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com