
Χ�������������ʣ���������A12O3����ϡ���ᣬ��H2SO4����Ba(OH)2���壬�ް�ˮ����A12(SO4)3������Ҫ��ش��������⡣
��1��������ǿ�ᷴӦ��������ǿ�Ӧ���� ������ţ���
��2�����ڵ���ʵ��� ������ţ���
��3��д����������ȡAl(OH)3�����ӷ���ʽ ��
��4�������ʷ�����Ӧ�����ӷ���ʽΪH++OH��=H2O����д���÷�Ӧ�Ļ�ѧ����ʽ ��
��5��34.2g������ˮ���500mL��Һ����Һ��SO42�������ʵ���Ũ��Ϊ ��
��6������۷�����Ӧ�Ļ�ѧ����ʽΪA1+4HNO3=A1(NO3)3+NO��+2H2O���÷�Ӧ�л�ԭ���������������ʵ���֮���� ��
��1���٢ڣ���1�֣����1����0.5�֣����һ�������֣�
��2���ڢܢݢߣ���2�֣����1����0.5�֣����һ�������֣�
��3��Al3����3NH3��H2O=Al(OH)3����3NH4������2�֣�
��4��2HNO3+Ba(OH)2= Ba(NO3)2+2H2O����2�֣�
��5��0.6mol/L����2�֣���λδд��0.5�֣�
��6��1��1����1�֣�
���������������1��Al��Al2O3������ǿ�ᷴӦ��������ǿ�Ӧ����2���������ᡢ��Ρ��������������ڵ���ʣ����ʡ�����Һ������Һ���ǵ���ʣ���3��ʵ���ҳ���������Һ�백ˮ��ȡAl(OH)3��Al3����3NH3��H2O=Al(OH)3����3NH4������4��H++OH��=H2O����ǿ����ǿ�������ɿ������κ�ˮ���кͷ�Ӧ��2HNO3+Ba(OH)2= Ba(NO3)2+2H2O����5����m/M��֪��n[A12(SO4)3]=0.1mol����A12(SO4)3��3SO42����֪��n(SO42��)=0.3mol����n/V��֪��c(SO42��)=0.6mol/L����6����Ԫ��ȫ����0�����ߵ�+3�ۣ�����������Al�ǻ�ԭ����1/4��Ԫ����+5�۽��͵�+2�ۣ�����ԭ����HNO3������������÷�Ӧ�л�ԭ����������֮��Ϊ1��1��
���㣺������Ԫ�ػ��������ʡ����ӷ���ʽ�����ʵ��������ʵ���Ũ�ȡ���ԭ���������������֪ʶ��


 ǧ�������������ĩ�����Ծ�����ϵ�д�
ǧ�������������ĩ�����Ծ�����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪Ca(OH)2��Cl2��Ӧ�������������¶��йأ���һ������ʯ������ͨ��һ����������������ǡ����ȫ��Ӧ(�����ķ�Ӧ��Ϊ���ȷ�Ӧ)���������к���Cl����ClO����ClO3�����ֺ���Ԫ�ص����ӣ�����ClO����ClO3���������ӵ����ʵ���(n)�뷴Ӧʱ��(t)��������ͼ��ʾ��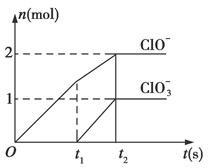
(1)t1ǰ������������______________________(�ѧʽ)��
(2)t2ʱ��Ca(OH)2��Cl2������Ӧ���ܵ����ӷ���ʽ��_____________________________��
(3)��ʯ�����к���Ca(OH)2�����ʵ�����________mol��
(4)NaClO2���ȶ��������Ȼ��û��������ƹ���ʱ������ը���䱬ը��IJ��������________(����ĸ)��
| A��NaCl��Cl2 | B��NaCl��NaClO | C��NaClO3��NaClO4 | D��NaCl��NaClO3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ijУͬѧΪ̽��Br2��I2��Fe3����������ǿ��������������ʵ�顣
ʵ��٣�ȡ����KI��Һ���Թ��У��ȼ�����ˮ�����ټ���CCl4�����ã��۲쵽�²�Һ����Ϻ�ɫ��
ʵ��ڣ�ȡ����FeSO4��Һ���Թ��У��ȼ�����ˮ�����ټ����μ�����KSCN��Һ�����۲쵽��Һ�ʺ�ɫ��
��1��д�����ӷ���ʽ��
ʵ��٣�______________________________________________________��
ʵ��ڣ�______________________________________________________��
��2������������ʵ�飬�����ʵ������Կ��Եó�����ȷ������________��
| A��Br2>I2 | B��Fe3��>Br2 | C��Br2>Fe3�� | D��I��>Br�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ijǿ������ҺX����Ba2+��Al3+��N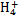 ��Fe2+��Fe3+��C
��Fe2+��Fe3+��C ��S
��S ��S
��S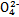 ��Cl-��N
��Cl-��N �е�һ�ֻ���,ȡ����Һ����ʵ��,ʵ����������:
�е�һ�ֻ���,ȡ����Һ����ʵ��,ʵ����������: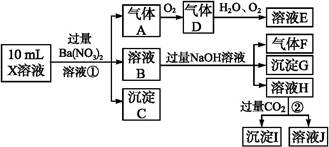
����������Ϣ,�ش���������:
(1)��ҺX�г�H+��϶����е�������������;����ȷ���Ƿ��е�������������,��Ҫȷ�����������ӵĴ���,��ɿ��Ļ�ѧ����������������������������
(2)����G�Ļ�ѧʽΪ��������
(3)д���й����ӷ���ʽ:
�����������A��������������������
��������ɳ���I��������������������
(4)����ⶨA��F��I��Ϊ0.01 mol,10 mL X��Һ��n(H+)="0.04" mol,������C���ʵ���0.07 mol,��˵��(1)�в���ȷ���������Ӵ��ڵ�����������������������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��Һ����Fe2+��Na+��Al3+��Ba2+��SO42-��NO3-��Cl-�е�4�����ӣ��������ӵ����ʵ�����Ϊ0.1mol���������Һ�м��������ϡ���ᣬ���������������Һ�������������(������ˮ�ĵ�������ӵ�ˮ��)���ش���������:
��1���������Һ�м��������ϡ�����KSCN��Һ��������: ��
��2��д�������Һ�м��������ϡ���ᷢ����Ӧ�����ӷ���ʽΪ:________________________________________________________________________��
��3��������������Һ������4�������ǣ�______________________________________��
��4���ж�����3�����Ӳ��ܴ��ڵ���Ҫ����������:
�������Һ�м��������ϡ���ᣬ���������������Һ������������䣬���������ӵ����ʵ�����Ϊ0.1mol�������__________________________________________��
��5�� �������Һ�м���������NaOH��Һ�������õij������������գ�������ù��������Ϊ______g
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��Ҫ��д�����з���ʽ��
��1��������Ͷ��FeCl3��Һ�з�Ӧ�������ӷ���ʽ ��
��2����������Ͷ������ͭ��Һ�������ӷ���ʽ ��
��3��̼���Ⱶ�������������Ʒ�Ӧ�����ӻ�ѧ����ʽ ��
��4��������̼�����Ʒ�Ӧ�����ӷ���ʽ_____________________________________________��
��5��������������������Һ��Ӧ�����ӷ���ʽ_______________________________________��
��6����NaHSO4��Һ��������������Һ�����ԣ�������Ӧ�����Һ�е����������������ӷ���ʽ
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
A��B��C��D���ֿ������Σ��ֱ���Na+��Ba2+��Cu2+��Ag+��CO32����SO42����Cl����NO3���еIJ�ͬ�����Ӻ������Ӹ�һ����ɡ���������ʵ�飺
�� �������θ�ȡ�������ֱ�����ʢ��5 mL����ˮ����֧�Թ��У�ֻ��B����Һ����ɫ��
�� �ֱ���4֧�Թ��м���2 mLϡ���ᣬ����A����Һ�в�����ɫ������C����Һ���н϶����ݲ�������D����Һ����������
����������ʵ���ƶ��������εĻ�ѧʽ�ֱ�Ϊ��
A B C D
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��1��ijһ��Ӧ��ϵ���з�Ӧ��������ﹲ5�����ʣ�S��H2S ��HNO3��NO ��H2O����÷�Ӧ�л�ԭ������_____������Ӧ������ת����0.3mol���ӣ������������������_____g��
��2����a mol Cl2ͨ�뺬b mol FeBr2����Һ�У���0< a / b��1/2 ʱ����Ӧ�����ӷ���ʽΪ��
2Fe2+ + Cl2 = 2Fe3+ + 2Cl����д����2�����ܷ��������ӷ���ʽ��
�ٵ� a / b =1ʱ ��
�ڵ�a / b��3/2ʱ ��
��3���۲����·�Ӧ���ܽ���ɣ�Ȼ������������⣺
��Al(OH)3��H2O  Al(OH)4��+ H+ ���� Cl2+2OH��= Cl��+ ClO��+H2O
Al(OH)4��+ H+ ���� Cl2+2OH��= Cl��+ ClO��+H2O
������֪B(OH)3��һԪ���ᣬ��д������뷽��ʽ�� ��
��������(CN)2����±�أ���д����������������Һ��Ӧ�����ӷ���ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��Һ�н����±������е�5������(������ˮ�ĵ��뼰���ӵ�ˮ��)���Ҹ����ӵ����ʵ�����Ϊ0.1mol��
| ������ | SO42����NO3����Cl�� |
| ������ | Fe3����Fe2����NH4+��Cu2����Al3�� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com