

| �¶�/������ | 0 | 20 | 30 | 45 | 60 | 80 | 100 |
| Sr(NO3)2 | 28.2 | 40.7 | 47 | 47.2 | 48.3 | 49.2 | 50.7 |
| Ca(NO3)2��4H2O | 102 | 129 | 152 | 230 | 300 | 358 | 408 |
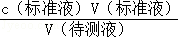 �����ı�Һ�࣬���ⶨ�������ȴ��Ȼ�ƫ�ߣ��ʴ�Ϊ��ƫ�ߡ���֪��������������л��ܼ�A�У�����Ϊ5.39-5.26=0.03g����̼�����Ӧ��ֻ�б����Ӻ������ӣ�Ũ��Ϊ0.107mol/L��̼������Һ22.98mL�����ʵ���Ϊ0.00246mol�������ᱵ�������ȵ����ʵ���Ϊy��x����������ʽΪ212x+261y=5.26 x+y=0.0246�����������ȵ�������������5.39��ȥ����ƺ����ᱵ�������������������Ϊ0.95��
�����ı�Һ�࣬���ⶨ�������ȴ��Ȼ�ƫ�ߣ��ʴ�Ϊ��ƫ�ߡ���֪��������������л��ܼ�A�У�����Ϊ5.39-5.26=0.03g����̼�����Ӧ��ֻ�б����Ӻ������ӣ�Ũ��Ϊ0.107mol/L��̼������Һ22.98mL�����ʵ���Ϊ0.00246mol�������ᱵ�������ȵ����ʵ���Ϊy��x����������ʽΪ212x+261y=5.26 x+y=0.0246�����������ȵ�������������5.39��ȥ����ƺ����ᱵ�������������������Ϊ0.95��

 Сѧ�̲���ȫ���ϵ�д�
Сѧ�̲���ȫ���ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��

| A��ʵ���ҽ��ТٵIJ������õ����������������ƾ��� |
| B������һ��������ת��Ϊ��ѧ�ܵĹ��� |
| C���ۢܢ��漰�ķ�Ӧ��Ϊ������ԭ��Ӧ |
| D�����з�Ӧ�����ӷ���ʽΪSO2+Br2+2H2O��4H++SO42-+2Br- |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A���ýྻ�IJ�˿պȡ������Һ�ھƾ��ƻ��������գ�����ʻ�ɫ������Һ��һ��������K+ |
| B������ɫ��Һ�м��������ữ��BaCl2��Һ�а�ɫ�������֣�����Һ�к���SO42�� |
| C����ij��Һ�еμ�������ˮ���ٵμ�KSCN��Һ������Һ���Ѫ��ɫ����ԭ��Һ��һ����Fe2+ |
| D����ij��ɫ��Һ�м��������Һ������Һ����ɫ����ԭ��Һ�к���I2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| �ɷ� | ����/(mg/L) | �ɷ� | ����/(mg/L) |
| Cl- | 18980 | Ca2+ | 400 |
| Na+ | 10560 | HCO3- | 142 |
| SO42- | 2560 | Mg2+ | 1272 |

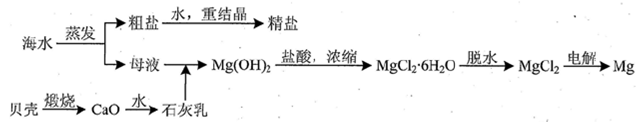
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��HNO3��Һ(H2SO4)������BaCl2��Һ������ |
| B��Fe2�� (Fe3��)����ԭ���ۣ����� |
| C��NaHCO3��Һ(Na2CO3)��ͨ�������CO 2 |
| D��CO2(HCl)��������Һ��ϴ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

| A��BaCl2 | B��BaCO3 |
| C��NaOH | D��Ba(OH)2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
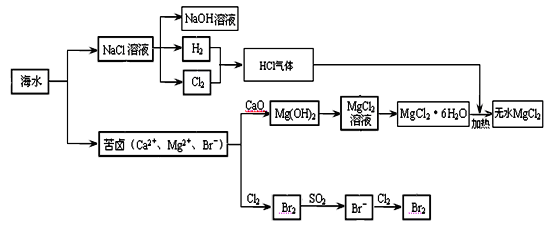
 MgCl2 + 6H2O���÷�ӦҪ��HCl�����н��У�ԭ���� ��
MgCl2 + 6H2O���÷�ӦҪ��HCl�����н��У�ԭ���� ���鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com