

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
 ����CuCO3��Cu(OH)2��������������SiO2�����Ļ����ʵ�����Կ�ȸʯΪԭ���Ʊ�CuSO4��5H2O�IJ������£�
����CuCO3��Cu(OH)2��������������SiO2�����Ļ����ʵ�����Կ�ȸʯΪԭ���Ʊ�CuSO4��5H2O�IJ������£�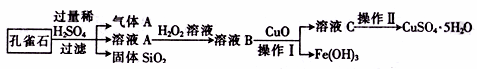
 �����ڴ˲����е���Ҫ������ ��
�����ڴ˲����е���Ҫ������ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
 �ܶ�Ϊ1.84 g��mL-1��_____________mL��
�ܶ�Ϊ1.84 g��mL-1��_____________mL�� ��һ��ʢ����������ˮ����Ͳ��ϡ�ͣ�����ȴ�����£�
��һ��ʢ����������ˮ����Ͳ��ϡ�ͣ�����ȴ�����£� �����ո����Բ��ӣ���
�����ո����Բ��ӣ����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

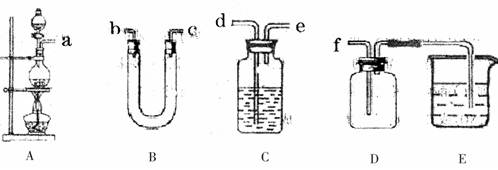
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 ͼ��ʾʵ��װ�ý���ʵ�飨ͼ��a��b��c��ʾֹˮ�У���
ͼ��ʾʵ��װ�ý���ʵ�飨ͼ��a��b��c��ʾֹˮ�У���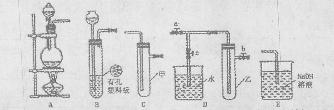
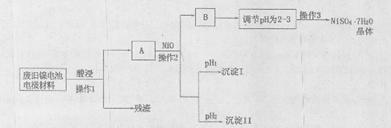
| M��OH��n | Ksp[�� | pH | |
| ��ʼ���� | ������ȫ | ||
| Al(OH)3 | 1.9��10-23 | 3.43 | 4.19 |
| Fe(OH)3 | 3.8��10-38 | 2.53 | 2.94 |
| Ni(OH)2 | 1.6��10-14 | 7.60 | 9.75 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��294.7 L | B��378.0 L | C��2240.0 L | D��1120.0 L |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��O2 H2��CO2���� | B��O2 CO2 | C��CO2���� | D��H2 O2 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com