
A”¢B”¢C”¢D”¢E¾łĪŖÓŠ»ś»ÆŗĻĪļ£¬ĖüĆĒÖ®¼äµÄ¹ŲĻµČēĶ¼ĖłŹ¾(ĢįŹ¾£ŗRCH=CHR'
ŌŚĖįŠŌøßĆĢĖį¼ŲČÜŅŗÖŠ·“Ӧɜ³ÉRCOOHŗĶR'COOH£¬ĘäÖŠRŗĶR'ĪŖĶ黳)”£
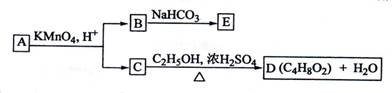
»Ų“šĻĀĮŠĪŹĢā£ŗ
(1)Ö±Į“»ÆŗĻĪļAµÄĻą¶Ō·Ö×ÓÖŹĮæŠ”ÓŚ90£¬A·Ö×ÓÖŠĢ¼”¢ĒāŌŖĖŲµÄ×ÜÖŹĮæ·ÖŹżĪŖ0.814£¬ĘäÓąĪŖŃõŌŖĖŲ£¬ŌņAµÄ·Ö×ÓŹ½ĪŖ_____________£»
(2)ŅŃÖŖBÓėNaHCO3ČÜŅŗĶźČ«·“Ó¦£¬ĘäĪļÖŹµÄĮæÖ®±ČĪŖ1£ŗ2£¬ŌņŌŚÅØĮņĖįµÄ“ß»ÆĻĀ£¬BÓė×ćĮæµÄC2H5OH·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ____________________________£¬·“Ó¦ĄąŠĶĪŖ____________________”£
(3)AæÉŅŌÓė½šŹōÄĘ×÷ÓĆ·Å³öĒāĘų£¬ÄÜŹ¹äåµÄĖÄĀČ»ÆĢ¼ČÜŅŗĶŹÉ«£¬ŌņAµÄ½į¹¹¼ņŹ½ŹĒ__________________
(4)DµÄĶ¬·ÖŅģ¹¹ĢåÖŠ£¬ÄÜÓėNaHCO3ČÜŅŗ·“Ó¦·Å³öCO2µÄÓŠ__________ÖÖ£¬ĘäĻąÓ¦µÄ½į¹¹¼ņŹ½ŹĒ__________________”£
£Ø1£©C5H10O”£
(2£©HOOC”ŖCH2”ŖCOOH£»
HOOC”ŖCH2”ŖCOOH + 2C2H5OH![]() C2H5OOC”ŖCH2”ŖCOOC2H5 +2H2O£»õ„»Æ·“Ó¦£Ø»ņČ”“ś·“Ó¦£©”£
C2H5OOC”ŖCH2”ŖCOOC2H5 +2H2O£»õ„»Æ·“Ó¦£Ø»ņČ”“ś·“Ó¦£©”£
(3£©HO”ŖCH2”ŖCH2”ŖCH=CH”ŖCH3”£
(4£©2£»CH3CH2CH2COOH”¢CH3CH(CH3)COOH”£
£Ø1£©ŃõµÄÖŹĮæ·ÖŹżĪŖ1£0.814£½0.186£¬¼Ł¶ØAµÄĻą¶Ō·Ö×ÓÖŹĮæĪŖ90£¬ŌņN(O)£½=1.0463£¬ĖłŅŌŃõµÄŌ×ÓøöŹżĪŖ1£¬ŌņAµÄĻą¶Ō·Ö×ÓÖŹĮæĪŖ£ŗ£½86£¬ÓÉÉĢÓą·ØµĆ86£16/12=5””10£¬¼“AµĆ·Ö×ÓŹ½ĪŖC5H10O”£
(2£©ÓÉÓŚBÓėNaHCO3ČÜŅŗĶźČ«·“Ó¦£¬ĘäĪļÖŹµÄĮæÖ®±ČĪŖ1£ŗ2£¬ĖłŅŌBÖŠŗ¬ÓŠ2øö£COOH£¬½įŗĻC£«C2H5OH![]() C2H4O2£«H2OæÉÖŖ£¬CĪŖCH3COOH£¬¼“BÖŠŗ¬ÓŠ3øöCŌ×Ó£¬ĻÖŅŃÖŖBÖŠŗ¬ÓŠ2øö£COOH£¬¼“BÖŠ»¹ŗ¬ÓŠŅ»øöCH2£¬ĖłŅŌBµÄ½į¹¹¼ņŹ½ĪŖHOOC£CH2£COOH£¬BÓė×ćĮæµÄC2H5OH·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ£ŗHOOC”ŖCH2”ŖCOOH + 2C2H5OH
C2H4O2£«H2OæÉÖŖ£¬CĪŖCH3COOH£¬¼“BÖŠŗ¬ÓŠ3øöCŌ×Ó£¬ĻÖŅŃÖŖBÖŠŗ¬ÓŠ2øö£COOH£¬¼“BÖŠ»¹ŗ¬ÓŠŅ»øöCH2£¬ĖłŅŌBµÄ½į¹¹¼ņŹ½ĪŖHOOC£CH2£COOH£¬BÓė×ćĮæµÄC2H5OH·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ£ŗHOOC”ŖCH2”ŖCOOH + 2C2H5OH![]() C2H5OOC”ŖCH2”ŖCOOC2H5 +2H2O£¬·“Ó¦ĄąŠĶĪŖõ„»Æ·“Ó¦”£
C2H5OOC”ŖCH2”ŖCOOC2H5 +2H2O£¬·“Ó¦ĄąŠĶĪŖõ„»Æ·“Ó¦”£
(3£©AæÉŅŌÓė½šŹōÄĘ×÷ÓĆ·Å³öĒāĘų£¬ĖµĆ÷AÖŠŗ¬ÓŠµÄ¹ŁÄÜĶÅĪŖ£OH£¬ÄÜŹ¹äåµÄĖÄĀČ»ÆĢ¼ČÜŅŗĶŹÉ«£¬ĖµĆ÷AÖŠŗ¬ÓŠC=C£¬ĒŅAĪŖÖ±Į“»ÆŗĻĪļ£¬½įŗĻĢāÉĻµÄĢįŹ¾æɵĆAµÄ½į¹¹¼ņŹ½ĪŖ£ŗ
HO”ŖCH2”ŖCH2”ŖCH=CH”ŖCH3”£
(4£©ÄÜÓėNaHCO3ČÜŅŗ·“Ó¦·Å³öCO2£¬ĖµĆ÷ĪŖōČĖį£¬Ęä½į¹¹¼ņŹ½ĪŖ£ŗCH3CH2CH2COOH”¢CH3CH(CH3)COOH”£



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
| ÅØĮņĖį |
| ”÷ |
| ÅØĮņĖį |
| ”÷ |



²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
| ÅØĮņĖį |
| ¼ÓČČ |
| ÅØĮņĖį |
| ¼ÓČČ |


 Š“³öĖÄÕßÖ®Ņ»¼“æÉ
Š“³öĖÄÕßÖ®Ņ»¼“æÉ Š“³öĖÄÕßÖ®Ņ»¼“æÉ
Š“³öĖÄÕßÖ®Ņ»¼“æÉ| ŹµŃ鱹ŗÅ | CĪļÖŹµÄĮæÅØ¶Č£Ømol?L-1£© | NaOHĪļÖŹµÄĮæÅØ¶Č£Ømol?L-1£© | »ģŗĻČÜŅŗµÄpH |
| m | 0.1 | 0.1 | pH=9 |
| n | 0.2 | 0.1 | pH£¼7 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ







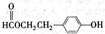
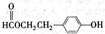
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ

| ÅØĮņĖį |
| ”÷ |
| ÅØĮņĖį |
| ”÷ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
| ĪļÖŹ±ąŗÅ | ĪļÖŹ×Ŗ»Æ¹ŲĻµ | A | D | E |
| ¢Ł |  |
Si | SiO2 | H2SiO3 |
| ¢Ś | N2 | NO2 | HNO3 | |
| ¢Ū | S | SO3 | H2SO4 | |
| ¢Ü | Na | Na2O2 | NaOH |
| A”¢¢Ś¢Ū | B”¢¢Ś¢Ü |
| C”¢¢Ł¢Ū¢Ü | D”¢¢Ł¢Ś¢Ū¢Ü |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com