

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
 ijŠ”×éĶ¬Ń§ÉčĻėÓĆČēĶ¼×°ÖƵē½āĮņĖį¼ŲČÜŅŗĄ“ÖĘČ”ŃõĘų”¢ĒāĘų”¢ĮņĖįŗĶĒāŃõ»Æ¼Ų£®
ijŠ”×éĶ¬Ń§ÉčĻėÓĆČēĶ¼×°ÖƵē½āĮņĖį¼ŲČÜŅŗĄ“ÖĘČ”ŃõĘų”¢ĒāĘų”¢ĮņĖįŗĶĒāŃõ»Æ¼Ų£®²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
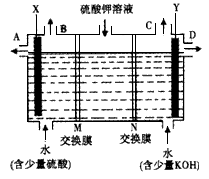
 ijæĪĶā»ī¶ÆŠ”×éĶ¬Ń§ÓĆČēĶ¼×°ÖĆ½ųŠŠŹµŃ飬ŹŌ»Ų“šĻĀĮŠĪŹĢā£ŗ
ijæĪĶā»ī¶ÆŠ”×éĶ¬Ń§ÓĆČēĶ¼×°ÖĆ½ųŠŠŹµŃ飬ŹŌ»Ų“šĻĀĮŠĪŹĢā£ŗ
| ||
| ||
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2013½ģøŹĖąŹ”ÕÅŅ“¶žÖŠøßČż10ŌĀŌĀæ¼»ÆѧŹŌ¾ķ£Ø°Ā°ą£©£Ø“ų½āĪö£© ĢāŠĶ£ŗĢīæÕĢā
(10·Ö)ijæĪĶā»ī¶ÆŠ”×éĶ¬Ń§ÓĆČēĶ¼×°ÖĆ½ųŠŠŹµŃ飬ŹŌ»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ČōæŖŹ¼Ź±æŖ¹ŲKÓėaĮ¬½Ó£¬ŌņA¼«µÄµē¼«·“Ó¦Ź½ĪŖ__________”£
£Ø2£©ČōæŖŹ¼Ź±æŖ¹ŲKÓėbĮ¬½Ó£¬ŌņB¼«µÄµē¼«·“Ó¦Ź½ĪŖ_____________£¬
×Ü·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ_____________ ”£
ÓŠ¹ŲÉĻŹöŹµŃ飬ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ(ĢīŠņŗÅ)________”£
¢ŁČÜŅŗÖŠNa£«ĻņA¼«ŅʶÆ
¢Ś“ÓA¼«“¦ŅŻ³öµÄĘųĢåÄÜŹ¹ŹŖČóKIµķ·ŪŹŌÖ½±äĄ¶
¢Ū·“Ó¦Ņ»¶ĪŹ±¼äŗó¼ÓŹŹĮæŃĪĖįæÉ»Öø“µ½µē½āĒ°µē½āÖŹµÄÅضČ
¢ÜČō±ź×¼×“æöĻĀB¼«²śÉś2.24 LĘųĢ壬ŌņČÜŅŗÖŠ×ŖŅĘ0.2 molµē×Ó
£Ø3£©øĆŠ”×éĶ¬Ń§Ä£Äā¹¤ŅµÉĻÓĆĄė×Ó½»»»Ä¤·ØÖĘÉÕ¼īµÄ·½·Ø£¬ÄĒĆ“æÉŅŌÉčĻėÓĆČēĶ¼×°ÖƵē½āĮņĖį¼ŲČÜŅŗĄ“ÖĘČ”ĒāĘų”¢ŃõĘų”¢ĮņĖįŗĶĒāŃõ»Æ¼Ų”£
¢ŁøƵē½ā²ŪµÄŃō¼«·“Ó¦Ź½ĪŖ______________________”£“ĖŹ±ĶعżŅõĄė×Ó½»»»Ä¤µÄĄė×ÓŹż_______(Ģī”°“óÓŚ”±”¢”°Š”ÓŚ”±»ņ”°µČÓŚ”±)ĶعżŃōĄė×Ó½»»»Ä¤µÄĄė×ÓŹż”£
¢ŚĶصēæŖŹ¼ŗó£¬Ņõ¼«ø½½üČÜŅŗpH»įŌö“ó£¬Ēė¼ņŹöŌŅņ_______________________________”£
¢ŪČō½«ÖʵƵÄĒāĘų”¢ŃõĘųŗĶĒāŃõ»Æ¼ŲČÜŅŗ×éŗĻĪŖĒāŃõČ¼ĮĻµē³Ų£¬Ōņµē³ŲÕż¼«µÄµē¼«·“Ó¦Ź½ĪŖ__________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012-2013ѧğøŹĖąŹ”øßČż10ŌĀŌĀæ¼»ÆѧŹŌ¾ķ£Ø°Ā°ą£©£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
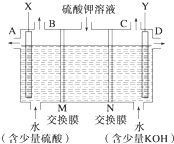
(10·Ö)ijæĪĶā»ī¶ÆŠ”×éĶ¬Ń§ÓĆČēĶ¼×°ÖĆ½ųŠŠŹµŃ飬ŹŌ»Ų“šĻĀĮŠĪŹĢā£ŗ

£Ø1£©ČōæŖŹ¼Ź±æŖ¹ŲKÓėaĮ¬½Ó£¬ŌņA¼«µÄµē¼«·“Ó¦Ź½ĪŖ__________”£
£Ø2£©ČōæŖŹ¼Ź±æŖ¹ŲKÓėbĮ¬½Ó£¬ŌņB¼«µÄµē¼«·“Ó¦Ź½ĪŖ_____________£¬
×Ü·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ_____________ ”£
ÓŠ¹ŲÉĻŹöŹµŃ飬ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ(ĢīŠņŗÅ)________”£
¢ŁČÜŅŗÖŠNa£«ĻņA¼«ŅʶÆ
¢Ś“ÓA¼«“¦ŅŻ³öµÄĘųĢåÄÜŹ¹ŹŖČóKIµķ·ŪŹŌÖ½±äĄ¶
¢Ū·“Ó¦Ņ»¶ĪŹ±¼äŗó¼ÓŹŹĮæŃĪĖįæÉ»Öø“µ½µē½āĒ°µē½āÖŹµÄÅضČ
¢ÜČō±ź×¼×“æöĻĀB¼«²śÉś2.24 LĘųĢ壬ŌņČÜŅŗÖŠ×ŖŅĘ0.2 molµē×Ó
£Ø3£©øĆŠ”×éĶ¬Ń§Ä£Äā¹¤ŅµÉĻÓĆĄė×Ó½»»»Ä¤·ØÖĘÉÕ¼īµÄ·½·Ø£¬ÄĒĆ“æÉŅŌÉčĻėÓĆČēĶ¼×°ÖƵē½āĮņĖį¼ŲČÜŅŗĄ“ÖĘČ”ĒāĘų”¢ŃõĘų”¢ĮņĖįŗĶĒāŃõ»Æ¼Ų”£

¢ŁøƵē½ā²ŪµÄŃō¼«·“Ó¦Ź½ĪŖ______________________”£“ĖŹ±ĶعżŅõĄė×Ó½»»»Ä¤µÄĄė×ÓŹż_______(Ģī”°“óÓŚ”±”¢”°Š”ÓŚ”±»ņ”°µČÓŚ”±)ĶعżŃōĄė×Ó½»»»Ä¤µÄĄė×ÓŹż”£
¢ŚĶصēæŖŹ¼ŗó£¬Ņõ¼«ø½½üČÜŅŗpH»įŌö“ó£¬Ēė¼ņŹöŌŅņ_______________________________”£
¢ŪČō½«ÖʵƵÄĒāĘų”¢ŃõĘųŗĶĒāŃõ»Æ¼ŲČÜŅŗ×éŗĻĪŖĒāŃõČ¼ĮĻµē³Ų£¬Ōņµē³ŲÕż¼«µÄµē¼«·“Ó¦Ź½ĪŖ__________________________”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com