
ΑΔΥΨΤΞΝ÷Ω…”…Υ°―νΥα”κ““ΥατϊΉς”Ο÷ΤΒΟΓΘΤδ÷Τ±Η‘≠άμ»γœ¬ΘΚ


ΑΔΥΨΤΞΝ÷(““θΘΥ°―νΥα)ΒΡΡΤ―Έ“Ή»ή”ΎΥ°ΓΘΑΔΥΨΤΞΝ÷Ω…Α¥»γœ¬≤Ϋ÷η÷Τ»ΓΚΆ¥ΩΜ·ΘΚ
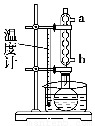
≤Ϋ÷η1ΘΚ‘ΎΗ…‘οΒΡ50 mL‘≤ΒΉ…’ΤΩ÷–Φ”»κ2 gΥ°―νΥαΓΔ5 mL““ΥατϊΚΆ5ΒΈ≈®ΝρΥαΘ§’ώΒ¥ ΙΥ°―νΥα»Ϊ≤Ω»ήΫβΓΘ
≤Ϋ÷η2ΘΚΑ¥ΆΦΥυ ΨΉΑ÷ΟΉΑ≈δΚΟ“«ΤςΘ§Ά®Υ°Θ§‘ΎΥ°‘Γ…œΦ”»»ΜΊΝς5ΓΪ10 minΘ§ΩΊ÷ΤΥ°‘ΓΈ¬Ε»‘Ύ85ΓΪ90 ΓφΓΘ
≤Ϋ÷η3ΘΚΖ¥”ΠΫα χΚσΘ§»Γœ¬Ζ¥”ΠΤΩΘ§ά以ȧ‘ΌΖ≈»κ±υΥ°÷–ά以ΓΔΫαΨßΓΔ≥ι¬ΥΓΔάδΥ°œ¥Β”2ΓΪ3¥ΈΘ§ΦΧ–χ≥ι¬ΥΒΟ¥÷≤ζΈοΓΘ
≤Ϋ÷η4ΘΚΫΪ¥÷≤ζΈοΉΣ“Τ÷Ν150 mL…’±≠÷–Θ§‘ΎΫΝΑηœ¬Φ”»κ25 mL±ΞΚΆΧΦΥα«βΡΤ»ή“ΚΘ§≥δΖ÷ΫΝΑηΘ§»ΜΚσΙΐ¬ΥΓΘ
≤Ϋ÷η5ΘΚΫΪ¬Υ“ΚΒΙ»κ10 mL 4 molΓΛLΘ≠1―ΈΥαΘ§ΫΝΑηΘ§ΫΪ…’±≠÷Ο”Ύ±υ‘Γ÷–ά以ȧ ΙΫαΨßΆξ»ΪΓΘ≥ι¬ΥΘ§‘Ό”ΟάδΥ°œ¥Β”2ΓΪ3¥ΈΓΘ
(1)≤Ϋ÷η1≈®ΝρΥαΒΡΉς”ΟΩ…Ρή «________ΓΘ
(2)≤Ϋ÷η2÷–Θ§άδΡΐΙήΆ®Υ°Θ§Υ°”Π¥”________ΩΎΫχ(ΧνΓΑaΓ±ΜρΓΑbΓ±)ΓΘ
(3)≤Ϋ÷η3≥ι¬Υ ±Θ§”– ±¬Υ÷ΫΜᥩٹȧ±ήΟβ¬Υ÷Ϋ¥©ΩΉΒΡ¥κ © «______________________________________________________________ΓΘ
(4)≤Ϋ÷η4ΖΔ…ζ÷ς“ΣΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΈΣ_____________________ΘΜΙΐ¬ΥΒΟΒΫΒΡΙΧΧεΈΣ________ΓΘ
(5)»ΓΦΗΝΘ≤Ϋ÷η5ΜώΒΟΒΡΨßΧεΦ”»κ Δ”–5 mLΥ°ΒΡ ‘Ιή÷–Θ§Φ”»κ1ΓΪ2ΒΈ1%»ΐ¬»Μ·Χζ»ή“ΚΘ§ΖΔœ÷»ή“Κ±δΉœ…ΪΘ§Ω…≤…”Ο________ΖΫΖ®Θ§Ϋχ“Μ≤Ϋ¥ΩΜ·ΨßΧεΓΘ
ΓΓ(1)¥ΏΜ·ΦΝΓΓ(2)b
(3)Φθ–Γ’φΩ’Ε»Μρ’ΏΦ”“Μ≤ψ(ΜρΕύ≤ψ)¬Υ÷Ϋ
(4)  ΘΪNaHCO3®DΓζ
ΘΪNaHCO3®DΓζ ΘΪCO2ΓϋΘΪH2OΓΓΨέΥ°―νΥαΓΓ(5)(”Ο Β±»ήΦΝ)÷ΊΫαΨß
ΘΪCO2ΓϋΘΪH2OΓΓΨέΥ°―νΥαΓΓ(5)(”Ο Β±»ήΦΝ)÷ΊΫαΨß
ΓΨΫβΈωΓΩΓΓ(1)”…÷Τ±Η‘≠άμΩ…“‘Ω¥≥ωΘ§≈®ΝρΥαΒΡΉς”Ο «‘ΎΗΟΖ¥”Π÷–ΉςΈΣ¥ΏΜ·ΦΝΓΘ
(2)άδΡΐΥ°ΒΡΝςœρ «ΓΑœ¬Ϋχ…œ≥ωΓ±Θ§Ι ¥”bΙήΫχΥ°Θ§aΙή≥ωΥ°ΓΘ
(3)±ήΟβ¬Υ÷Ϋ¥©ΩΉΒΡ¥κ © «Φθ–Γ’φΩ’Ε»Μρ’ΏΦ”“Μ≤ψ(ΜρΕύ≤ψ)¬Υ÷ΫΓΘ
(4)ΗυΨί≤ζΈοΒΡΫαΙΙΧΊΒψΩ…÷Σ «τ»Μυ(ΓΣCOOH)”κNaHCO3ΖΔ…ζΖ¥”Π ΘΪNaHCO3®DΓζ
ΘΪNaHCO3®DΓζ ΘΪCO2ΓϋΘΪH2OΘΜ¥÷≤ζΤΖΜΙ”–Ω…ΡήΖΔ…ζΗ±Ζ¥”ΠΒΟΒΫΨέΥ°―νΥαΓΘ
ΘΪCO2ΓϋΘΪH2OΘΜ¥÷≤ζΤΖΜΙ”–Ω…ΡήΖΔ…ζΗ±Ζ¥”ΠΒΟΒΫΨέΥ°―νΥαΓΘ
(5)ΖΔœ÷»ή“Κ±δΉœΘ§ΥΒΟςΨßΧε÷–¥φ‘ΎΥ°―νΥαΘ§Ι »‘–η(”Ο Β±»ήΦΝ)÷ΊΫαΨßΘ§Ϋχ“Μ≤Ϋ¥ΩΜ·ΨßΧεΓΘ



| ΡξΦΕ | ΗΏ÷–ΩΈ≥Χ | ΡξΦΕ | ≥θ÷–ΩΈ≥Χ |
| ΗΏ“Μ | ΗΏ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ“Μ | ≥θ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏΕΰ | ΗΏΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θΕΰ | ≥θΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏ»ΐ | ΗΏ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ»ΐ | ≥θ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2013-2014―ßΡξΗΏΩΦΜ·―ßΕΰ¬÷Η¥œΑΫ≠Υ’Ή®”Ο ΒΎ6Ϋ≤Μ·―ßΖ¥”ΠΥΌ¬ ΚΆΜ·―ßΤΫΚβΝΖœΑΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚ―Γ‘ώΧβ
‘ΎΈ“ΙζΤϊ≥ΒΈ≤Τχ“―≥…ΈΣ÷ς“ΣΒΡ¥σΤχΈέ»ΨΈοΘ§ Ι”ΟœΓΆΝΒ»¥ΏΜ·ΦΝΡήΫΪΤϊ≥ΒΈ≤Τχ÷–ΒΡCOΓΔNOxΚΆΧΦ«βΜ·ΚœΈοΉΣΜ·≥…ΈόΕΨΈο÷ Θ§¥”ΕχΦθ…ΌΈέ»ΨΓΘœρ»ίΜΐœύΆ§ΒΡΝΫΗωΟή±’»ίΤςΡΎ(ΉΑ”–Β»ΝΩΒΡΡ≥÷÷¥ΏΜ·ΦΝ)Θ§Ζ÷±π≥δ»κΒ»ΝΩΒΡNOxΦΑC3H6Θ§‘Ύ≤ΜΆ§Έ¬Ε»œ¬Θ§ΖΔ…ζ»γœ¬Ζ¥”ΠΘΚ

ΔΌ18 NO(g)ΘΪ2C3H6(g) 9N2(g)ΘΪ6CO2(g)ΘΪ6H2O(g)ΘΜ
9N2(g)ΘΪ6CO2(g)ΘΪ6H2O(g)ΘΜ
ΔΎ18 NO2(g)ΘΪ4C3H6(g) 9N2(g)ΘΪ12 CO2(g)ΘΪ12 H2O(g)
9N2(g)ΘΪ12 CO2(g)ΘΪ12 H2O(g)
Ζ÷±π≤βΕ®≤ΜΆ§Έ¬Ε» ±NOxΒΡΉΣΜ·¬ Θ§ΥυΒΟΒΡ«ζœΏ»γΆΦΥυ ΨΓΘœ¬Ν–ΥΒΖ®’ΐ»ΖΒΡ «(ΓΓΓΓ)ΓΘ
AΘ°ΗΟΖ¥”ΠΒΡΠΛHΘΨ0
BΘ°Φ”»κ¥ΏΜ·ΦΝΘ§NO(g)ΒΡΉΣΜ·¬ ±»NO2(g)ΒΡΒΆ
CΘ°Ζ÷άκ≥ωH2O(g)Ω…ΧαΗΏΧΦ«βΜ·ΚœΈοΒΡΉΣΜ·¬
DΘ°‘ω¥σC3H6(g)ΒΡ≈®Ε»Ω…ΧαΗΏNOxΒΡΉΣΜ·¬ Θ§ΨΏ”– ΒΦ “β“ε
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2013-2014―ßΡξΗΏΩΦΜ·―ßΕΰ¬÷Η¥œΑΫ≠Υ’Ή®”Ο ΒΎ2Ϋ≤Μ·―ß≥Θ”ΟΦΤΝΩΝΖœΑΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚΧνΩ’Χβ
ΙΛ“Β…œ”ΟΚ§–ΩΈοΝœ(Κ§FeOΓΔCuOΒ»‘”÷ )Ω…÷ΤΒΟΜν–‘ZnOΘ§Νς≥Χ»γœ¬ΘΚ

(1)…œ ωΝς≥Χ÷–Θ§Ϋΰ≥ω”ΟΒΡ «60%H2SO4(1.5 gΓΛcmΘ≠3)Θ§≈δ÷Τ’β÷÷H2SO4 100 mL–η“Σ18.4 molΓΛLΘ≠1ΒΡ≈®H2SO4________ mL(±ΘΝτ“ΜΈΜ–Γ ΐ)ΓΘ
(2)Φ”»κ―θΜ·ΦΝH2O2ΚσΘ§”–Fe(OH)3≥ΝΒμ≥ωœ÷Θ§ΟΜ”–Cu(OH)2≥ΝΒμ≥ωœ÷Θ§»τ»ή“Κ÷–c(Fe3ΘΪ)ΘΫ2.6ΓΝ10Θ≠18 molΓΛLΘ≠1Θ§‘ρ»ή“Κ÷–c(Cu2ΘΪ)ΒΡ»Γ÷ΒΖΕΈß «________molΓΛLΘ≠1ΓΘ(“―÷ΣKsp[Fe(OH)3]ΘΫ2.6ΓΝ10Θ≠39Θ§
Ksp[Cu(OH)2]ΘΫ2.2ΓΝ10Θ≠20)
(3)Φ”»κNH4HCO3Κσ…ζ≥…ΒΡ≥ΝΒμ «–ΈΧ§ΨυΈΣZna(OH)b(CO3)c(aΓΔbΓΔcΈΣ’ΐ’ϊ ΐ)ΒΡΝΫ÷÷Φν ΫΧΦΥα–ΩAΚΆBΒΡΜλΚœΈοΘ§A÷–aΘΫ5ΓΔbΘΫ6Θ§‘ρ…ζ≥…Φν ΫΧΦΥα–ΩAΒΡΜ·―ßΖΫ≥Χ ΫΈΣ_______________________________________________ΓΘ
(4)»Γœ¥Β”ΓΔΚφΗ…ΚσΒΡΦν ΫΧΦΥα–ΩAΚΆBΒΡΜλΚœΈο49.70 gΘ§ΤδΈο÷ ΒΡΝΩΈΣ0.10 molΘ§ΗΏΈ¬±Κ…’Άξ»ΪΖ÷ΫβΒΟΒΫ37.26 g ZnOΓΔ3.584 L CO2(±ξΉΦΉ¥Ωωœ¬)ΚΆΥ°Θ§Ά®ΙΐΦΤΥψ«σ≥ωΦν ΫΧΦΥα–ΩBΒΡΜ·―ß ΫΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2013-2014―ßΡξΗΏΩΦΜ·―ßΕΰ¬÷Η¥œΑΫ≠Υ’Ή®”Ο ΒΎ1Ϋ≤Έο÷ ΒΡΉι≥…–‘÷ ΚΆΖ÷άύΝΖœΑΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚ―Γ‘ώΧβ
Ϋπ τΦΑΤδΜ·ΚœΈοΒΡΉΣΜ·ΙΊœΒ «Μ·―ß―ßœΑΒΡ÷Ί“ΣΡΎ»ί÷°“ΜΓΘœ¬Ν–ΗςΉιΈο÷ ΒΡΉΣΜ·ΙΊœΒ÷–≤Μ»Ϊ≤Ω «Ά®Ιΐ“Μ≤ΫΖ¥”ΠΆξ≥…ΒΡ «(ΓΓΓΓ)ΓΘ

≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2013-2014―ßΡξΗΏΩΦΜ·―ßΕΰ¬÷Η¥œΑΫ≠Υ’Ή®”Ο ΒΎ15Ϋ≤ Β―ιΜ·―ßΝΖœΑΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚ Β―ιΧβ
”…’ΐΕΓ¥Φ(”Οn?C4H9OH±μ Ψ)÷Τ±Η’ΐδεΕΓΆι(Ζ–ΒψΘΚ101.6 Γφ)ΒΡΖ¥”Π»γœ¬ΘΚn?C4H9OHΘΪNaBrΘΪH2SO4®DΓζn?C4H9BrΘΪNaHSO4ΘΪH2O
Η±Ζ¥”ΠΘΚn?C4H9OH CH3CH2CHCH2ΓϋΘΪH2O
CH3CH2CHCH2ΓϋΘΪH2O
2n?C4H9OH (n?C4H9)2OΘΪH2O
(n?C4H9)2OΘΪH2O
3H2SO4(≈®)ΘΪ2NaBr(ΙΧ)=2NaHSO4ΘΪBr2ΓϋΘΪSO2ΓϋΘΪ2H2O
“―÷ΣΖ¥”ΠΈοΦΑΤδ”ΟΝΩΘΚΙΧΧεNaBr 0.24 molΘ§n?C4H9OH 0.20 molΘ§≈®H2SO4 29 mLΘ§H2O 20 mLΓΘ
÷Τ±ΗΙΐ≥ΧΨ≠άζ»γœ¬ΈεΗω≤Ϋ÷ηΘ§ ‘ΜΊ¥πœ¬Ν–Έ ΧβΘΚ
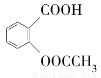
(1)ΆΕΝœΘΚ‘Ύ‘≤ΒΉ…’ΤΩ÷–Φ”»κ20 mLΥ°Θ§‘Ό¬ΐ¬ΐΦ”»κ29 mL≈®ΝρΥαΘ§ΜλΚœΨυ‘»≤Δά以÷Ν “Έ¬ΚσΘ§‘Ό“ά¥ΈΦ”»κ0.20 mol’ΐΕΓ¥Φ(‘Φ18 mL)ΚΆ0.24 molδεΜ·ΡΤΘ§≥δΖ÷’ώΒ¥ΚσΦ”»κΦΗΝΘΖ– ·ΓΘ
ΔΌά以÷Ν “Έ¬Κσ‘ΌΦ”’ΐΕΓ¥ΦΚΆδεΜ·ΡΤΒΡΡΩΒΡ «________ΓΘ
AΘ°Φθ…ΌHBrΒΡΜ”ΖΔ
BΘ°Ζά÷Ι’ΐΕΓ¥ΦΒΡΜ”ΖΔ
CΘ°Ζά÷ΙδεΜ·ΡΤ±Μ―θΜ·
DΘ°Ζά÷ΙΈ¬Ε»ΙΐΗΏΒΦ÷¬ΧΩΜ·ΫαΫΙ
ΔΎ±ΨΖ¥”Π÷–ΝρΥα”κδεΜ·ΡΤΉς”Ο…ζ≥…«βδεΥαΘ§«βδεΥα”κ’ΐΕΓ¥ΦΉς”ΟΖΔ…ζ»Γ¥ζΖ¥”Π…ζ≥…’ΐδεΕΓΆιΓΘΝρΥαΒΡ”ΟΝΩΚΆ≈®Ε»Ιΐ¥σΕΦΕ‘±Ψ÷Τ±Η≤ΜάϊΒΡ÷ς“Σ‘≠“ρ «
_____________________________________________________ΓΘ
(2)Φ”»»ΜΊΝςΘΚΖ¥”ΠΉΑ÷Ο―Γ‘ώΔρΕχ≤Μ―Γ‘ώΔώΒΡ‘≠“ρ «_________________ΓΘ‘ΎΖ¥”ΠΉΑ÷Ο÷–άδΡΐΥ°”ΠΗΟ¥”________(ΧνΓΑAΓ±ΜρΓΑBΓ±)ΕΥΫχΥ°ΓΘ

(3)Ζ÷άκ¥÷≤ζΈοΘΚΖ¥”ΠΫα χ¥ΐΖ¥”Π“Κά以ΚσΘ§”Ο÷±–ΈάδΡΐΙήΜΜœ¬«ρ–ΈάδΡΐΙήΘ§ΫΪΤδ±δΈΣ’τΝσΉΑ÷ΟΫχ––’τΝσΘ§ΒΟΒΫΒΡ¥÷≤ζΈο÷–≥ΐ÷ς≤ζΤΖΆβΘ§ΜΙΚ§œ¬Ν–―Γœν÷–ΒΡ________ΓΘ
ΔΌC4H9OHΓΓΔΎSO2ΓΓΔέ(C4H9)2OΓΓΔήBr2ΓΓΔίNaHSO4 ΔόH2OΓΓΔΏH2SO4
(4)œ¥Β”¥÷≤ζΈοΘΚΫΪΝσ≥ω“Κ“Τ÷ΝΖ÷“Κ¬©ΕΖ÷–Θ§Φ”»κΒ»ΧεΜΐΒΡΥ°œ¥Β”ΚσΘ§Ζ÷“ΚΒΟ”–Μζ≤ψΓΘ»γΙϊ≤ΜΡή≈–ΕœΡΡ≤ψ «”–Μζ≤ψΘ§Ω…“‘”Ο_______________ΖΫΖ®ά¥≈–ΕœΓΘΕ‘”–Μζ≤ψΘ§Ϋχ––œ¥Β”≥ΐ‘”ΓΔ≥ΐΥ°ΚσΩ…ΒΟΒΫ¥÷≤ζΤΖΓΘ
(5) ’Φ·≤ζΈοΘΚΫΪΗ…‘οΚΟΒΡ≤ζΈο“Τ÷Ν–Γ’τΝσΤΩ÷–Θ§Φ”»»’τΝσΘ§ ’Φ·99ΓΪ103 ΓφΒΡΝσΖ÷ΓΘ“Σ÷ΛΟςΉν÷’≤ζΈο «n?C4H9BrΘ§ΖΫΖ® «_____________________________
___________________________________________ΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2013-2014―ßΡξΗΏΩΦΜ·―ßΕΰ¬÷Η¥œΑΫ≠Υ’Ή®”Ο ΒΎ14Ϋ≤Έο÷ ΫαΙΙ”κ–‘÷ ΝΖœΑΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚΧνΩ’Χβ
ΕΧ÷ήΤΎΖ«Ϋπ τ‘ΣΥΊAΓΔBΓΔCΒΡΚΥΒγΚ… ΐ“ά¥Έ‘ω¥σΘ§A‘≠Ή”ΒΡΆβΈßΒγΉ”≈≈≤Φ ΫΈΣns2np2Θ§C «ΒΊΩ«÷–Κ§ΝΩΉνΕύΒΡ‘ΣΥΊΓΘ‘ΣΥΊDΓΔEΕΦΈΜ”ΎΒΎΥΡ÷ήΤΎΘ§Τδ÷–EΒΡΚΥΒγΚ… ΐΈΣ29Θ§D‘≠Ή”ΒΡΚΥΆβΈ¥≥…Ε‘ΒγΉ” ΐ‘ΎΆ§÷ήΤΎ÷– «ΉνΕύΒΡΓΘ«κ”ΟΕ‘”ΠΒΡ‘ΣΥΊΖϊΚ≈ΜρΜ·―ß ΫΧνΩ’ΘΚ
(1)‘ΣΥΊAΓΔBΓΔCΒΡΒΎ“ΜΒγάκΡή”…–ΓΒΫ¥σΒΡΥ≥–ρΈΣ________ΓΘ
(2)“―÷Σ(AB)2Ζ÷Ή”÷–Φϋ”κΦϋ÷°ΦδΒΡΦ–Ϋ«ΈΣ180ΓψΘ§≤Δ”–Ε‘≥Τ–‘Θ§«“ΟΩΗω‘≠Ή”ΒΡΉνΆβ≤ψΒγΉ” ΐΨυ¬ζΉψ8ΒγΉ”ΫαΙΙΘ§‘ρΤδΫαΙΙ ΫΈΣ________Θ§1 molΗΟΖ÷Ή”÷–Κ§”–ΒΡΠ–ΦϋΒΡ ΐΡΩΈΣ________ΓΘ
(3)ΜυΧ§D‘≠Ή”ΒΡΆβΈßΒγΉ”≈≈≤Φ ΫΈΣ________ΓΘΜ·ΚœΈοDO2Cl2ΒΡ»έΒψ «Θ≠96.5 ΓφΘ§Ζ–Βψ «117 ΓφΘ§‘ρΙΧΧ§DO2Cl2 τ”Ύ________ΨßΧεΓΘ

(4)“―÷Σ‘ΣΥΊEΒΡ«βΜ·ΈοΒΡΨßΑϊΫαΙΙ»γ…œΆΦΥυ ΨΘ§‘ρΤδΜ·―ß ΫΈΣ___________
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2013-2014―ßΡξΗΏΩΦΜ·―ßΕΰ¬÷Η¥œΑΫ≠Υ’Ή®”Ο ΒΎ13Ϋ≤ Β―ιΖΫΑΗ…ηΦΤ”κΤάΦέΝΖœΑΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚ Β―ιΧβ
ΙΧΧεœθΥα―ΈΦ”»»“ΉΖ÷Ϋβ«“≤ζΈοΫœΗ¥‘”ΓΘΡ≥―ßœΑ–ΓΉι“‘Mg(NO3)2ΈΣ―–ΨΩΕ‘œσΘ§ΡβΆ®Ιΐ Β―ιΧΫΨΩΤδ»»Ζ÷ΫβΒΡ≤ζΈοΘ§Χα≥ω»γœ¬4÷÷≤¬œκΘΚ
ΦΉΘΚMg(NO2)2ΓΔNO2ΓΔO2
““ΘΚMgOΓΔNO2ΓΔO2
±ϊΘΚMg3N2ΓΔO2
ΕΓΘΚMgOΓΔNO2ΓΔN2
(1) Β―ι–ΓΉι≥…‘±Ψ≠Χ÷¬έ»œΕ®≤¬œκΕΓ≤Μ≥…ΝΔΘ§άμ”… «_______________________ΓΘ
≤ι‘ΡΉ ΝœΒΟ÷ΣΘΚ2NO2ΘΪ2NaOH=NaNO3ΘΪNaNO2ΘΪH2O
’κΕ‘ΦΉΓΔ““ΓΔ±ϊ≤¬œκΘ§…ηΦΤ»γœ¬ΆΦΥυ ΨΒΡ Β―ιΉΑ÷Ο(ΆΦ÷–Φ”»»ΓΔΦ–≥÷“«ΤςΒ»Ψυ Γ¬‘)ΘΚ

(2) Β―ιΙΐ≥Χ
ΔΌ“«ΤςΝ§Ϋ”ΚσΘ§Ζ≈»κΙΧΧε ‘ΦΝ÷°«ΑΘ§ΙΊ±’kΘ§ΈΔ»»”≤÷ ≤ΘΝßΙή(A)Θ§Ιέ≤λΒΫE÷–”–Τχ≈ίΝ§–χΖ≈≥ωΘ§±μΟς__________ΓΘ
ΔΎ≥Τ»ΓMg(NO3)2ΙΧΧε3.7 g÷Ο”ΎA÷–Θ§Φ”»»«ΑΆ®»κN2“‘«ΐΨΓΉΑ÷ΟΡΎΒΡΩ’ΤχΘ§ΤδΡΩΒΡ «________ΘΜΙΊ±’kΘ§”ΟΨΤΨΪΒΤΦ”»» ±Θ§’ΐ»Ζ≤ΌΉς «œ»________Θ§»ΜΚσΙΧΕ®‘ΎΙή÷–ΙΧΧε≤ΩΈΜœ¬Φ”»»ΓΘ
ΔέΙέ≤λΒΫA÷–”–ΚλΉΊ…ΪΤχΧε≥ωœ÷Θ§CΓΔD÷–Έ¥ΦϊΟςœ‘±δΜ·ΓΘ
Δή¥ΐ―υΤΖΆξ»ΪΖ÷ΫβΘ§AΉΑ÷Οά以÷Ν “Έ¬ΓΔ≥ΤΝΩΘ§≤βΒΟ Θ”ύΙΧΧεΒΡ÷ ΝΩΈΣ1.0 gΓΘ
Δί»Γ…ΌΝΩ Θ”ύΙΧΧε”Ύ ‘Ιή÷–Θ§Φ”»κ ΝΩΥ°Θ§Έ¥ΦϊΟςœ‘œ÷œσΓΘ
(3) Β―ιΫαΙϊΖ÷ΈωΧ÷¬έ
ΔΌΗυΨί Β―ιœ÷œσΚΆ Θ”ύΙΧΧεΒΡ÷ ΝΩΨ≠Ζ÷ΈωΩ…≥θ≤Ϋ»Ζ»œ≤¬œκ______ «’ΐ»ΖΒΡΓΘ
ΔΎΗυΨίD÷–ΈόΟςœ‘œ÷œσΘ§“ΜΈΜΆ§―ß»œΈΣ≤ΜΡή»Ζ»œΖ÷Ϋβ≤ζΈο÷–”–O2Θ§“ρΈΣ»τ”–O2Θ§D÷–ΫΪΖΔ…ζ―θΜ·ΜΙ‘≠Ζ¥”ΠΘΚ______________(Χν–¥Μ·―ßΖΫ≥Χ Ϋ)Θ§»ή“Κ―’…ΪΜαΆ »ΞΘΜ–ΓΉιΧ÷¬έ»œΕ®Ζ÷Ϋβ≤ζΈο÷–”–O2¥φ‘ΎΘ§Έ¥Φλ≤βΒΫΒΡ‘≠“ρ «__________
____________________________________________________ΓΘ
Δέ–ΓΉιΧ÷¬έΚσ¥ο≥…ΒΡΙ≤ Ε «…œ ω Β―ι…ηΦΤ»‘≤ΜΆξ…ΤΘ§–ηΗΡΫχΉΑ÷ΟΫχ“Μ≤ΫΧΫΨΩΓΘ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2013-2014―ßΡξΗΏΩΦΜ·―ßΕΰ¬÷Η¥œΑΫ≠Υ’Ή®”Ο ΒΎ11Ϋ≤”–ΜζΜ·―ßΜυ¥ΓΝΖœΑΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚ―Γ‘ώΧβ
XΓΔYΖ÷±π «ΝΫ÷÷“©ΈοΘ§ΤδΫαΙΙ»γΆΦΓΘ

œ¬Ν–ΥΒΖ®’ΐ»ΖΒΡ «(ΓΓΓΓ)ΓΘ
AΘ°1 mol X”κNaOH»ή“ΚΖ¥”ΠΘ§ΉνΕύœϊΚΡ3 mol NaOH
BΘ°XΓΔYΕΦ”–Ϋœ«ΩΒΡΥ°»ή–‘
CΘ°XΓΔYΕΦΡή ΙKMnO4»ή“ΚΆ …Ϊ
DΘ°XΓΔYΒΡΥ°Ϋβ≤ζΈο”–Ω…ΡήΖΔ…ζΨέΚœΖ¥”Π
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ2013-2014―ßΡξΗΏΩΦΜ·―ßΕΰ¬÷Η¥œΑΧαΖ÷―ΒΝΖ Ή®Χβ9Ζ«Ϋπ τ‘ΣΥΊΦΑΤδΜ·ΚœΈοΝΖœΑΨμΘ®ΫβΈωΑφΘ© Χβ–ΆΘΚ―Γ‘ώΧβ
œ¬Ν–Μ·―ß Β―ι ¬ ΒΦΑΤδΫα¬έΕΦ’ΐ»ΖΒΡ «Θ®ΓΓΓΓΘ©ΓΘ
―Γœν Β―ι ¬ ΒΫα¬έ
AΫΪSO2Ά®»κΚ§HClOΒΡ»ή“Κ÷–…ζ≥…H2SO4HClOΒΡΥα–‘±»H2SO4«Ω
B¬Ν≤≠‘ΎΨΤΨΪΒΤΜπ―φ…œΦ”»»»έΜ·ΒΪ≤ΜΒΈ¬δ¬Ν≤≠±μΟφ―θΜ·¬Ν»έΒψΗΏ”Ύ¬Ν
CSiO2Ω…“‘ΚΆNaOH»ή“ΚΦΑHF»ή“ΚΖ¥”ΠSiO2 τ”ΎΝΫ–‘―θΜ·–‘
DΫΪSO2Ά®»κδεΥ°÷–Θ§δεΥ°Ά …ΪSO2ΨΏ”–Τ·ΑΉ–‘
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΑΌΕ»÷¬–≈ - ΝΖœΑ≤αΝ–±μ - ‘ΧβΝ–±μ
Κΰ±± ΓΜΞΝΣΆχΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΤΫΧ® | Άχ…œ”–ΚΠ–≈œΔΨΌ±®Ή®«χ | Βγ–≈’©Τ≠ΨΌ±®Ή®«χ | …φάζ Ζ–ιΈό÷ς“ε”–ΚΠ–≈œΔΨΌ±®Ή®«χ | …φΤσ«÷»®ΨΌ±®Ή®«χ
ΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΒγΜΑΘΚ027-86699610 ΨΌ±®” œδΘΚ58377363@163.com