
¹čŌŚµŲæĒÖŠµÄŗ¬Įæ½Ļøß”£¹č¼°Ęä»ÆŗĻĪļµÄæŖ·¢ÓÉĄ“ŅŃ¾Ć£¬ŌŚĻÖ“śÉś»īÖŠÓŠ¹ć·ŗÓ¦ÓĆ”£»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©1810ğȚµä»Æѧ¼Ņ±“²ÉĄūĪŚĖ¹ŌŚ¼ÓČČŹÆӢɰ”¢Ä¾ĢæŗĶĢśŹ±£¬µĆµ½Ņ»ÖÖ”°½šŹō”±”£ÕāÖÖ”°½šŹō”±æÉÄÜŹĒ ”£
£Ø2£©ĢÕ“É”¢Ė®ÄąŗĶ²£Į§ŹĒ³£ÓĆµÄ¹čĖįŃĪ²ÄĮĻ”£ĘäÖŠ£¬Éś²śĘÕĶز£Į§µÄÖ÷ŅŖŌĮĻÓŠ ”£
£Ø3£©øß“æ¹čŹĒĻÖ“śŠÅĻ¢”¢°ė µ¼ĢåŗĶ¹ā·ü·¢µēµČ²śŅµ¶¼ŠčŅŖµÄ»ł“”²ÄĮĻ”£¹¤ŅµÉĻĢį“æ¹čÓŠ¶ąÖÖĀ·Ļߣ¬ĘäÖŠŅ»ÖÖ¹¤ŅÕĮ÷³ĢŹ¾ŅāĶ¼¼°Ö÷ŅŖ·“Ó¦ČēĻĀ£ŗ
µ¼ĢåŗĶ¹ā·ü·¢µēµČ²śŅµ¶¼ŠčŅŖµÄ»ł“”²ÄĮĻ”£¹¤ŅµÉĻĢį“æ¹čÓŠ¶ąÖÖĀ·Ļߣ¬ĘäÖŠŅ»ÖÖ¹¤ŅÕĮ÷³ĢŹ¾ŅāĶ¼¼°Ö÷ŅŖ·“Ó¦ČēĻĀ£ŗ
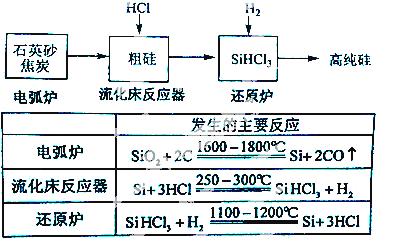
¢ŁÓĆŹÆӢɰŗĶ½¹ĢæŌŚµē»”ĀÆÖŠøßĪĀ¼ÓČČŅ²æÉŅŌÉś²śĢ¼»Æ¹č£¬øĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ £»Ģ¼»Æ¹čÓÖ³Ę £¬Ę侧Ģå½į¹¹Óė ĻąĖĘ”£
¢ŚŌŚĮ÷»Æ“²·“Ó¦µÄ²śĪļÖŠ£¬SiHCl3“óŌ¼Õ¼85%£¬»¹ÓŠSiCl4”¢ SiH2Cl2”¢SiH3ClµČ£¬ÓŠ¹ŲĪļÖŹµÄ·ŠµćŹż¾ŻČēĻĀ±ķ£¬Ģį“æSiHCl3µÄÖ÷ŅŖ¹¤ŅÕ²Ł×÷ŅĄ“ĪŹĒ³Į½µ”¢ĄäÄżŗĶ ”£
SiH2Cl2”¢SiH3ClµČ£¬ÓŠ¹ŲĪļÖŹµÄ·ŠµćŹż¾ŻČēĻĀ±ķ£¬Ģį“æSiHCl3µÄÖ÷ŅŖ¹¤ŅÕ²Ł×÷ŅĄ“ĪŹĒ³Į½µ”¢ĄäÄżŗĶ ”£
| ĪļÖŹ | Si | SiCl4 | SiHCl3 | SiH2Cl2 | SiH3Cl | HCl | Si |
| ·Šµć/”ę | 2355 | 57.6 | 31.8 | 8.2 | -30.4 | -84.9 | -111.9 |
¢ŪSiHCl3¼«Ņ×Ė®½ā£¬ĘäĶźČ«Ė®½āµÄ²śĪļĪŖ ”£
£Ø4£©ĀČ¼ī¹¤ŅµæÉĪŖÉĻŹö¹¤ŅÕÉś²śĢį¹©²æ·ÖŌĮĻ£¬ÕāŠ©ŌĮĻŹĒ ”£
 Ńō¹āæĪĢĆæĪŹ±×÷ŅµĻµĮŠ“š°ø
Ńō¹āæĪĢĆæĪŹ±×÷ŅµĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
Éč NAĪŖ°¢·üŁ¤µĀĀŽ³£ŹżµÄÖµ”£ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ
NAĪŖ°¢·üŁ¤µĀĀŽ³£ŹżµÄÖµ”£ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ
A£®1.6gÓÉŃõĘųŗĶ³ōŃõ×é³ÉµÄ»ģŗĻĪļÖŠŗ¬ÓŠŃõŌ×ӵďżÄæĪŖ0.1NA
B£®0.1mol±ūĻ©ĖįÖŠŗ¬ÓŠĖ«¼üµÄŹżÄæĪŖ0.1NA
C£®±ź×¼×“æöĻĀ£¬11.2L±½ÖŠŗ¬ÓŠ·Ö×ӵďżÄæĪŖ0.5NA
D£®ŌŚ ¹żŃõ»ÆÄĘÓėĖ®µÄ·“Ó¦ÖŠ£¬ĆæÉś³É0.1molŃõĘų£¬×ŖŅʵē×ӵďżÄæĪŖ0.4NA
¹żŃõ»ÆÄĘÓėĖ®µÄ·“Ó¦ÖŠ£¬ĆæÉś³É0.1molŃõĘų£¬×ŖŅʵē×ӵďżÄæĪŖ0.4NA
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
»ÆѧÓĆÓļŹĒѧĻ°»ÆѧµÄÖŲŅŖ¹¤¾ß£¬ĻĀĮŠÓĆĄ“±ķŹ¾ĪļÖŹ±ä»ÆµÄ»ÆѧÓĆÓļÖŠ£¬ÕżČ·µÄŹĒ
A .øÖĢśøÆŹ“Ź±æÉÄÜ·¢ÉśµÄ·“Ó¦(Õż¼«)£ŗ2H2O+O2£4e£=4OH£
.øÖĢśøÆŹ“Ź±æÉÄÜ·¢ÉśµÄ·“Ó¦(Õż¼«)£ŗ2H2O+O2£4e£=4OH£
B.1 L”¢0.5 mol/LĻ”ĮņĖįÓė1 L”¢1 mol/LNaOHČÜŅŗ·“Ó¦·Å³ö57.3kJµÄČČ£¬ŌņĘäÖŠŗĶČȵÄČČ»Æѧ·½³ĢŹ½£ŗH2SO4(aq)+2NaOH(aq)=Na2SO4(aq)+2H2O(l)£»”÷H= £114.6 kJ/mol
C.Ģ¼ĖįĒāÄĘČÜŅŗÓė×ćĮæ³ĪĒåŹÆ»ŅĖ®·“Ó¦£ŗHCO3£+Ca2++OH£=CaCO3”ż+H2O
D.ĒāŃõ»ÆĢś³Įµķæɱ»ĒāäåĖįČܽā£ŗ2Fe(OH)3£«6H£«£«2Br££½2Fe2£«£«Br2£«6H2O
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ÓÉ»ĘĶæó(Ö÷ŅŖ³É·ÖŹĒCuFeS2) Į¶ÖĘ¾«ĶµÄ¹¤ŅÕĮ÷³ĢŹ¾ŅāĶ¼ČēĻĀ£ŗ
Į¶ÖĘ¾«ĶµÄ¹¤ŅÕĮ÷³ĢŹ¾ŅāĶ¼ČēĻĀ£ŗ
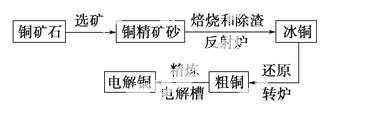
(1)ŌŚ·“ÉäĀÆÖŠ£¬°ŃĶ¾«æóÉ°ŗĶŹÆӢɰ»ģŗĻ¼ÓČȵ½1 000 ”ę×óÓŅ£¬»ĘĶæóÓėæÕĘų·“Ӧɜ³ÉCuŗĶFeµÄµĶ¼ŪĮņ»ÆĪļ£¬ĒŅ²æ·ÖFeµÄĮņ»ÆĪļ×Ŗ±äĪŖµĶ¼ŪŃõ»ÆĪļ”£øĆ¹ż³ĢÖŠĮ½øöÖ÷ŅŖ·“Ó¦µÄ»Æѧ·½³ĢŹ½·Ö±šŹĒ____________________”¢__________________________£¬·“ÉäĀÆÄŚÉś³É ĀÆŌüµÄÖ÷ŅŖ³É·ÖŹĒ________£»
ĀÆŌüµÄÖ÷ŅŖ³É·ÖŹĒ________£»
(2)±łĶ(Cu2SŗĶFeS»„ĻąČŪŗĻ¶ų³É)ŗ¬CuĮæĪŖ20%”«50%”£×ŖĀÆÖŠ£¬½«±łĶ¼ÓČŪ¼Į(ŹÆӢɰ)ŌŚ1 200 ”ę×óÓŅ“µČėæÕĘų½ųŠŠ“µĮ¶”£±łĶÖŠµÄCu2S±»Ńõ»Æ³ÉCu2O£¬Éś³ÉµÄCu2OÓėCu2S·“Ó¦£¬Éś³Éŗ¬CuĮæŌ¼ĪŖ98.5%µÄ“ÖĶ£¬øĆ¹ż³Ģ·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½·Ö±šŹĒ____________£»
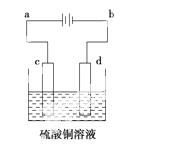
(3)“ÖĶµÄµē½ā¾«Į¶ČēÓŅĶ¼ĖłŹ¾”£ŌŚ“ÖĶµÄµē½ā¹ż³ĢÖŠ£¬“ÖĶ°åÓ¦ŹĒĶ¼ÖŠµē¼«________(ĢīĶ¼ÖŠµÄ×ÖÄø)£»ŌŚµē¼«dÉĻ·¢ÉśµÄµē¼«·“Ó¦Ź½ĪŖ____________________________£»Čō“ÖĶÖŠ»¹ŗ¬ÓŠAu”¢Ag”¢Fe£¬ĖüĆĒŌŚµē½ā²ŪÖŠµÄ“ęŌŚŠĪŹ½ŗĶĪ»ÖĆĪŖ____________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
¹¤ŅµÉĻÓĆÖŲøõĖįÄĘ£ØNa2Cr2O7£©½į¾§ŗóµÄÄøŅŗ£Øŗ¬ÉŁĮæŌÓÖŹFe3+£©Éś²śÖŲøõĖį¼Ų£ØK2Cr2O7£©”£¹¤ŅÕĮ÷³Ģ¼°Ļą¹ŲĪļÖŹČܽā¶ČĒśĻßČēĶ¼
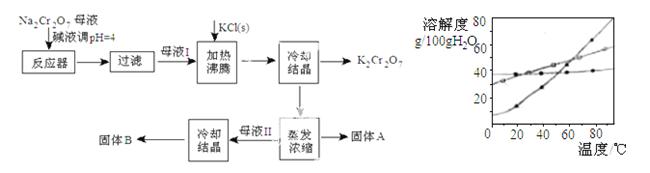 £Ø1£©ÓÉNa2Cr2O7Éś²śK2Cr2O7µÄ»Æѧ·½³ĢŹ½ĪŖ
£Ø1£©ÓÉNa2Cr2O7Éś²śK2Cr2O7µÄ»Æѧ·½³ĢŹ½ĪŖ  ”£ĶعżĄä¹ż½į¾§Īö³ö“óĮæK2Cr2O7µÄŌŅņŹĒ ”£
”£ĶعżĄä¹ż½į¾§Īö³ö“óĮæK2Cr2O7µÄŌŅņŹĒ ”£
£Ø2£©ĻņNa2Cr2O7ÄøŅŗÖŠ¼Ó¼īŅŗµ÷pHµÄÄæµÄŹĒ ”£
£Ø3£©¹ĢĢåAÖ÷ŅŖĪŖ £ØĢī»Æѧ Ź½£©
Ź½£© £¬¹ĢĢåBÖ÷ŅŖĪŖ £ØĢī»ÆѧŹ½£©”£
£¬¹ĢĢåBÖ÷ŅŖĪŖ £ØĢī»ÆѧŹ½£©”£
£Ø4£©ÓĆČČĖ®Ļ“µÓ¹ĢĢåA£¬»ŲŹÕµÄĻ“µÓŅŗ×ŖŅʵ½ÄøŅŗ £ØĢī”°I”±”°II”±»ņ”°III”±£©ÖŠ£¬¼ČÄÜĢįøß²śĀŹÓÖæÉŹ¹ÄÜŗÄ×īµĶ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ŗ£Ė®Õ¼µŲĒņ×Ü“¢Ė®ĮæµÄ97.2%”£Čō°Ńŗ£Ė®µ»ÆŗĶ»Æ¹¤Éś²ś½įŗĻĘšĄ“£¬¼ČæÉŅŌ½ā¾öµĖ®×ŹŌ“ȱ·¦µÄĪŹĢā£¬ÓÖæÉŅŌ³ä·ÖĄūÓĆŗ£Ńó׏Ō“”£Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
(1)ÄæĒ°£¬ŅŌĻĀ”°¼¼Źõ”±ÖŠ£¬æÉÓĆÓŚ”°ŗ£Ė®µ»Æ”±µÄŹĒ___________________________
(ĢīŠņŗÅ)”£
¢ŁÕōĮó·Ø””¢ŚÄ¤·ÖĄė·Ø(µēÉųĪö”¢·“ÉųĶø)””¢ŪĄä¶³·Ø””¢ÜĄė×Ó½»»»·Ø
(2)ŗ£ Ė®É¹ŃĪĄūÓƵĻÆѧŌĄķŹĒ____________________________________________£»
Ė®É¹ŃĪĄūÓƵĻÆѧŌĄķŹĒ____________________________________________£»
·ÖĄėŹ³ŃĪ¾§ĢåŗóµÄÄøŅŗÖŠŗ¬ÓŠ____________£¬¾¹ż·ÖĄė”¢Ģį“æŗó£¬æÉÓĆÓŚ_________”£
(3)¹¤ŅµÉĻĄūÓƵē½ā±„ŗĶŹ³ŃĪĖ®æÉÖʵĆÖŲŅŖ»Æ¹¤²śĘ·£¬·“Ó¦µÄ Ąė×Ó·½³ĢŹ½ĪŖ________________________________________________________________________£»
Ąė×Ó·½³ĢŹ½ĪŖ________________________________________________________________________£»
µē½āĖłÓƵď³ŃĪĖ®ŠčŅŖ¾«ÖĘ£¬ĘäŌŅņŹĒ_____________________________________
________________________________________________________________________£»
¾«ÖĘŹ±ĖłÓĆŹŌ¼Į£ŗŃĪĖį”¢BaCl2ČÜŅŗ”¢NaOHČÜŅŗ”¢Na2CO3ČÜŅŗ£¬¼ÓČėŹŌ¼ĮŹ±£¬Na2CO3ČÜŅŗ±ŲŠėŌŚ¼ÓČė__________________________________________________________Ö®ŗó¼ÓČė”£
(4)½üÄźĄ“ÓŠČĖĢį³öĮĖŅ»ÖÖĄūÓĆĀČ¼ī¹¤Ņµ²śĘ·¼°ĀČ»ÆÄĘŃ»·ÖĪĄķŗ¬¶žŃõ»ÆĮņµÄ·ĻĘųĶ¬Ź±»ŲŹÕ¶žŃõ»ÆĮņµÄ·½·Ø£¬øĆ·½·ØµÄĮ÷³ĢČēĻĀ£ŗ
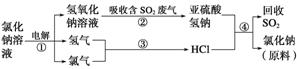
Š“³ö·“Ó¦¢Ś”¢¢ÜµÄ»Æѧ·½³ĢŹ½£ŗ_____________________________________________”¢
________________________________________________________________________ӣ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ŗĻ³É°±¹¤Ņµ”¢ĮņĖį¹¤ŅµµÄÉś²ś¹¤ŅÕ Į÷³ĢČēĶ¼ĖłŹ¾ ”£
ӣ

ŗĻ³ÉĖžŗĶ½Ó“„ŹŅÖŠµÄ·“Ó¦·Ö±šĪŖ
N2(g)£«3H2(g) 2NH3(g)””¦¤H£¼0£»
2SO2(g)£«O2(g) 2SO3(g)””¦¤H£¼0”£
(1)Š“³öĮ÷³ĢÖŠÉč±øµÄĆū³Ę£ŗB________£¬X________”£
(2)½ųČėŗĻ³ÉĖžŗĶ½Ó“„ŹŅÖŠµÄĘųĢ嶼ŅŖ½ųŠŠČČ“¦Ąķ£¬×īĄķĻėµÄČČ“¦Ąķ·½·ØŹĒ___________________”£
(3)²ÉÓĆŃ»·²Ł×÷æÉĢįøßŌĮĻµÄĄūÓĆĀŹ£¬ĻĀĮŠÉś²śÖŠ£¬²ÉÓĆŃ»·²Ł×÷µÄŹĒ________(ĢīŠņŗÅ)”£
¢ŁĮņĖį¹¤Ņµ””¢ŚŗĻ³É°±¹¤Ņµ””¢ŪĻõĖį¹¤Ņµ
(4)¹¤ŅµÉĻ³£ÓĆ98.3%µÄÅØĮņĖįĪüŹÕSO3¶ų²»ÓĆĻ”ĮņĖį»ņĖ®µÄŌŅņŹĒ_______________”£
(5)¹¤ŅµÉś²śÖŠ³£ÓĆ°±£Ėį·Ø½ųŠŠĪ²ĘųĶŃĮņ£¬ŅŌ“ļµ½Ļū ³żĪŪČ¾”¢
³żĪŪČ¾”¢ ·ĻĪļĄūÓƵÄÄæµÄ”£ĮņĖį¹¤ŅµĪ²ĘųÖŠµÄSO2¾“¦ĄķæÉŅŌµĆµ½Ņ»ÖֻƷŹ£¬øĆ·ŹĮĻµÄ»ÆѧŹ½ŹĒ___________________”£
·ĻĪļĄūÓƵÄÄæµÄ”£ĮņĖį¹¤ŅµĪ²ĘųÖŠµÄSO2¾“¦ĄķæÉŅŌµĆµ½Ņ»ÖֻƷŹ£¬øĆ·ŹĮĻµÄ»ÆѧŹ½ŹĒ___________________”£
(6)øł¾Ż»Æѧ Ę½ŗāŅʶÆŌĄķ£¬ĻĀĮŠĢõ¼ž»ņ“ėŹ©ŗĻĄķµÄŹĒ________(ĢīŠņŗÅ)”£
Ę½ŗāŅʶÆŌĄķ£¬ĻĀĮŠĢõ¼ž»ņ“ėŹ©ŗĻĄķµÄŹĒ________(ĢīŠņŗÅ)”£
¢ŁŗĻ³É°±¹¤ŅµŌŚøßŃ¹ĻĀ½ųŠŠ
¢ŚŗĻ³É°±¹¤ŅµŗĶĮņĖį¹¤Ņµ¶¼Ź¹ÓĆ“ß»Æ¼Į
¢Ū¼°Ź±½«°±Ņŗ»Æ¶ų·ÖĄė
¢ÜĮņĖį¹¤ŅµÖŠ£¬¾»»ÆŗóµÄĀÆĘųÖŠŅŖÓŠ¹żĮææÕĘų
¢ŻŗĻ³É°±¹¤ŅµŗĶĮņĖį¹¤Ņµ¶¼²ÉÓĆŹŹŅĖµÄĪĀ¶Č
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ŹĄ½ē»·±£ĮŖĆĖ½ØŅéČ«Ćę½ūÖ¹Ź¹ÓĆĀČĘųÓĆÓŚŅūÓĆĖ®µÄĻū¶¾£¬¶ų½ØŅé²ÉÓĆøߊ§”°ĀĢÉ«”±Ļū¶¾¼Į¶žŃõ»ÆĀČ”£¶žŃõ»ÆĀČŹĒŅ»ÖÖ¼«Ņ×±¬ÕصÄĒæŃõ»ÆŠŌĘųĢ壬Ņ×ČÜÓŚĖ®”¢²»ĪČ¶Ø”¢³Ź»ĘĀĢÉ«£¬ŌŚÉś²śŗĶŹ¹ÓĆŹ±±ŲŠė¾”ĮæÓĆĻ”ÓŠĘųĢå½ųŠŠĻ”ŹĶ£¬Ķ¬Ź±ŠčŅŖ±ÜĆā¹āÕÕ”¢Õš¶Æ»ņ¼ÓČČ”£ŹµŃéŹŅŅŌµē½ā·ØÖʱøClO2µÄĮ÷³ĢČēĻĀ£ŗ
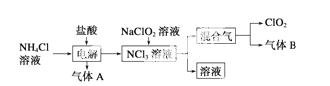
(1)ClO2ÖŠĖłÓŠŌ×Ó________(Ģī”°ŹĒ”±»ņ”°²»ŹĒ”±)¶¼Āś×ć8µē×Ó½į¹¹”£ÉĻĶ¼ĖłŹ¾µē½ā·ØÖĘµĆµÄ²śĪļÖŠŌÓÖŹĘųĢåBÄÜŹ¹ŹÆČļŹŌŅŗĻŌĄ¶É«£¬³żČ„ŌÓÖŹĘųĢåæÉŃ”ÓĆ________”£
A£®±„ŗĶŹ³ŃĪĖ® B£®¼īŹÆ»Ņ
C£®ÅØĮņĖį D£®ÕōĮóĖ®
(2)ĪČ¶ØŠŌ¶žŃõ»ÆĀČŹĒĪŖĶĘ¹ć¶žŃõ»ÆĀȶųæŖ·¢µÄŠĀŠĶ²śĘ·£¬ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ________”£
A£®¶žŃõ»ÆĀČæɹć·ŗÓĆÓŚ¹¤ŅµŗĶŅūÓĆĖ®“¦Ąķ
B£®Ó¦ÓĆŌŚŹ³Ę·¹¤ŅµÖŠÄÜÓŠŠ§µŲŃÓ³¤Ź³Ę·Öü²ŲĘŚ
C£®ĪČ¶ØŠŌ¶žŃõ»ÆĀČµÄ³öĻÖ“ó“óŌö¼ÓĮĖ¶žŃõ»ÆĀȵď¹ÓĆ·¶Ī§
D£®ŌŚ¹¤×÷ĒųŗĶ³ÉĘ·“¢²ŲŹŅÄŚ£¬ŅŖÓŠĶØ·ē×°ÖĆŗĶ¼ą²ā¼°¾Æ±Ø×°ÖĆ
(3)Å·ÖŽ¹ś¼ŅÖ÷ŅŖ²ÉÓĆĀČĖįÄĘŃõ»ÆÅØŃĪĖįÖʱø”£»Æѧ·“Ó¦·½³ĢŹ½ĪŖ________________________________________________________________”£
ȱµćÖ÷ŅŖŹĒ²śĀŹµĶ”¢²śĘ·ÄŃŅŌ·ÖĄė£¬»¹æÉÄÜĪŪČ¾»·¾³”£
(4)ĪŅ¹ś¹ć·ŗ²ÉÓĆ¾øÉŌļæÕĘųĻ”ŹĶµÄĀČĘųÓė¹ĢĢåŃĒĀČĖįÄĘ(NaClO2)·“Ó¦Öʱø£¬»Æѧ·½³ĢŹ½ŹĒ________________________________________________________________________
________________________________________________________________________£¬
“Ė·ØĻą±ČÅ·ÖŽ·½·ØµÄÓŵćŹĒ______________________________________________”£
(5)æĘѧ¼ŅÓÖŃŠ¾æ³öĮĖŅ»ÖÖŠĀµÄÖʱø·½·Ø£¬ĄūÓĆĮņĖįĖį»ÆµÄ²ŻĖį(H2C2O4)ČÜŅŗ»¹ŌĀČĖįÄĘ£¬»Æѧ·“Ó¦·½³ĢŹ½ĪŖ_________________________________________________________
________________________________________________________________________ӣ
“Ė·ØĢįøßĮĖÉś²ś¼°“¢“ę”¢ŌĖŹäµÄ°²Č«ŠŌ£¬ŌŅņŹĒ________________________________
________________________________________________________________________ӣ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
Ńõ»Æ»¹Ō·“Ó¦ÖŠŹµ¼ŹÉĻ°üŗ¬Ńõ»ÆŗĶ»¹ŌĮ½øö¹ż³Ģ”£ĻĀĆęŹĒŅ»øö»¹Ō¹ż³Ģ
µÄ·“Ó¦Ź½£ŗ NO3££«4H£«£«3e”śNO£«2H2O
KMnO4”¢Na2CO3”¢Cu2O”¢Fe2(SO4)3ĖÄÖÖĪļÖŹÖŠµÄŅ»ÖÖĪļÖŹ£Ø¼×£©ÄÜŹ¹ÉĻŹö»¹Ō¹ż³Ģ·¢Éś”£
£Ø1£©Š“³ö²¢ÅäĘ½øĆŃõ»Æ»¹Ō·“Ó¦µÄ·½³ĢŹ½£ŗ
£Ø2£©·“Ó¦ÖŠĻõĖįĢåĻÖĮĖ ”¢ ŠŌÖŹ”£
£Ø3£©·“Ó¦ÖŠČō ²śÉś0.2molĘųĢ壬Ōņ×ŖŅʵē×ÓµÄĪļÖŹµÄĮæŹĒ mol”£
²śÉś0.2molĘųĢ壬Ōņ×ŖŅʵē×ÓµÄĪļÖŹµÄĮæŹĒ mol”£
£Ø4£©Čō1mol¼×ÓėijÅضČĻõĖį·“Ó¦Ź±£¬±»»¹ŌĻõĖįµÄĪļÖŹµÄĮæŌö¼Ó£¬ŌŅņŹĒ£ŗ
ӣ
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com