
”¾»ÆѧєŠŽ5-ÓŠ»ś»Æѧ»ł“””æ(15·Ö)ÓŠ»śøß·Ö×Ó»ÆŗĻĪļGµÄŗĻ³ÉĀ·ĻßČēĻĀ£ŗ

ŅŃÖŖ£ŗ
¢ŁA¼ČÄÜÓėNaHCO3ČÜŅŗ·“Ó¦£¬ÓÖÄÜÓėFeCl3ČÜŅŗ·¢ÉśĻŌÉ«·“Ó¦£¬ĘäŗĖ“Ź²ÕńĒāĘ×ÓŠ4øö·å”£
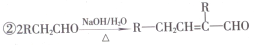
Ēė»Ų“š£ŗ
£Ø1£©BµÄĆū³ĘĪŖ________£¬GÖŠŗ¬ÓŠµÄ¹ŁÄÜĶÅĆū³ĘĪŖ________”£
£Ø2£©B”śC”¢D”śEµÄ·“Ó¦ĄąŠĶ·Ö±šĪŖ________”¢________”£
£Ø3£©A£«E”śFµÄ»Æѧ·½³ĢŹ½ĪŖ________”£
£Ø4£©D·¢ÉśŅų¾µ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ________”£
£Ø5£©·ūŗĻĻĀĮŠĢõ¼žµÄEµÄĶ¬·ÖŅģ¹¹ĢåÓŠ________ÖÖ(²»æ¼ĀĒĮ¢ĢåŅģ¹¹)£¬Š“³öĘäÖŠŅ»ÖֵĽį¹¹¼ņŹ½_______”£¢Łŗ¬ÓŠ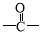 ½į¹¹£¬¢Ś±½»·ÉĻÓŠ2øöČ”“ś»ł
½į¹¹£¬¢Ś±½»·ÉĻÓŠ2øöČ”“ś»ł

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014-2015ѧğŗž±±Ź”µČøßČżĻĀѧʌʌ֊ĖÄŠ£ĮŖæ¼»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ĻĀĮŠŹĒ25 ”ęŹ±Ä³Š©ŃĪµÄČܶȻż³£ŹżŗĶČõĖįµÄµēĄėĘ½ŗā³£Źż£¬ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ
»ÆѧŹ½ | AgCl | Ag2CrO4 | CH3COOH | HClO | H2CO3 |
Ksp»ņKa | Ksp=1.8”Į10£10 | Ksp=9.0”Į10£12 | Ka=1.8”Į10£5 | Ka=3.0”Į10£8 | Ka1=4.1”Į10£7 Ka2=5.6”Į10£11 |
A£®ĻąĶ¬ÅضČCH3COONaŗĶNaClOµÄ»ģŗĻČÜŅŗÖŠ£¬ø÷ÖÖĄė×ÓÅØ¶ČµÄ“óŠ”¹ŲĻµŹĒ£ŗc(Na£«)£¾c(ClO£)£¾c(CH3COO£)£¾c(OH£)£¾c(H£«)
B£®“ĪĀČĖįÄĘČÜŅŗÖŠĶØČėÉŁĮæCO2µÄĄė×Ó·½³ĢŹ½ĪŖ£ŗ2ClO££«CO2£«H2O£½CO32 ££«2HClO
C£®Ļņ0.1 mol”¤L£1 CH3COOHČÜŅŗÖŠµĪ¼ÓNaOHČÜŅŗÖŠÖĮc(CH3COOH):c(CH3COO£)£½5£ŗ9£¬“ĖŹ±ČÜŅŗµÄpH=5
D£®ĻņÅØ¶Č¾łĪŖ1.0”Į10£3 mol”¤L£1µÄKClŗĶK2CrO4»ģŗĻČÜŅŗÖŠµĪ¼Ó1.0”Į10£3 mol”¤L£1µÄAgNO3ČÜŅŗ£¬CrO42ØDĻČŠĪ³É³Įµķ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014-2015ѧğŗžÄĻŹ”øßČż×ŪŗĻÄÜĮ¦²āŹŌ£ØĪ壩Ąķ×Ū»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ÄÜÕżČ·±ķŹ¾ĻĀĮŠ·“Ó¦µÄĄė×Ó·½³ĢŹ½
A£®Ļņ±½·ÓÄĘČÜŅŗÖŠĶØČėÉŁĮæCO2£ŗ2C6H5O££«CO2£«H2OØD”ś2C6H5OH£«CO32£
B£®£ØNH4£©2Fe£ØSO4£©2ČÜŅŗÓė¹żĮæNaOHČÜŅŗ·“Ó¦ÖĘFe£ØOH£©2£ŗFe2£«£«2OH£=Fe£ØOH£©2”ż
C£®ĻņFeCl3ČÜŅŗÖŠ¼ÓČėMg£ØOH£©2£ŗ3Mg£ØOH£©2 + 2Fe3+ = 2Fe£ØOH£©3 + 3Mg2+
D£®NaHCO3ČÜŅŗÓėĻ”H2SO4·“Ó¦:CO32£+ 2H+ = H2O + CO2”ü
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014-2015ѧğŗÓ±±Ź”øßČżĻĀѧʌȿµ÷æ¼ŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗ¼ņ“šĢā
Ń”æ¼[»Æѧ”Ŗ”ŖĪļÖŹ½į¹¹ÓėŠŌÖŹ]£Ø15·Ö£©ÓÉCu”¢N”¢B”¢NiµČŌŖĖŲ×é³ÉµÄŠĀŠĶ²ÄĮĻӊ׏ć·ŗÓĆĶ¾”£
£Ø1£©»łĢ¬ Cu£«µÄ×īĶā²ćŗĖĶāµē×ÓÅŲ¼Ź½ĪŖ_________.
£Ø2£©ŃŠ¾æÕßŌ¤ĻėŗĻ³ÉŅ»øö“æ“āÓɵŖ×é³ÉµÄŠĀĪļÖÖ N5£«N3££¬ČōN5£« Ąė×ÓÖŠĆæøöµŖŌ×Ó¾łĀś×ć8µē×Ó½į¹¹£¬ŅŌĻĀÓŠ¹ŲN5£« ĶĘ²āÕżČ·µÄŹĒ ( £©
A£®N5£«ÓŠ24øöµē×Ó
B£®N5£«Ąė×ÓÖŠ“ęŌŚČż¶ŌĪ“³É¼üµÄµē×Ó¶Ō
C£®N5£«ŃōĄė×ÓÖŠ“ęŌŚĮ½øöµŖµŖČż¼ü
£Ø3£©»ÆŗĻĪļ 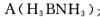 ŹĒŅ»ÖÖĒ±ŌŚµÄ“¢Ēā²ÄĮĻ£¬ĖüæÉÓÉĮłŌŖ»·×“»ÆŗĻĪļ
ŹĒŅ»ÖÖĒ±ŌŚµÄ“¢Ēā²ÄĮĻ£¬ĖüæÉÓÉĮłŌŖ»·×“»ÆŗĻĪļ 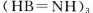 ”£Ķعż
”£Ķعż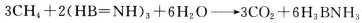 ÖʵƔ£
ÖʵƔ£
¢ŁÓėÉĻŹö»Æѧ·½³ĢŹ½ÓŠ¹ŲµÄŠšŹö²»ÕżČ·µÄŹĒ_________£ØĢī±źŗÅ£©”£
A.·“Ó¦Ē°ŗóĢ¼Ō×ӵĹģµĄŌÓ»ÆĄąŠĶ²»±ä
B£®CH4”¢H2O”¢CO2·Ö×Óæռ乹ŠĶ·Ö±šŹĒ£ŗÕżĖÄĆęĢåŠĪ”¢VŠĪ”¢Ö±ĻߊĪ
C£®µŚŅ»µēĄėÄÜ£ŗN>O>C>B
D£®»ÆŗĻĪļAÖŠ“ęŌŚÅäĪ»¼ü
¢Ś1øö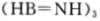 ·Ö×ÓÖŠÓŠ_________øö
·Ö×ÓÖŠÓŠ_________øö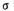 ¼ü”£
¼ü”£
£Ø4£©ŌŚÅšĖįŃĪÖŠ£¬ŅõĄė×ÓÓŠĮ“ד”¢»·×“µČ¶ąÖÖ½į¹¹ŠĪŹ½”£Ķ¼(a£©ŹĒŅ»ÖÖĮ“ד½į¹¹µÄ¶ąÅšĖįøł£¬Ōņ¶ąÅšĖįøłĄė×Ó·ūŗÅĪŖ_________Ķ¼(b£©ŹĒŚɰ¾§ĢåÖŠŅõĄė×ӵĻ·×“½į¹¹£¬ĘäÖŠÅšŌ×Ó²ÉČ”µÄŌÓ»ÆĄąŠĶĪŖ_________”£
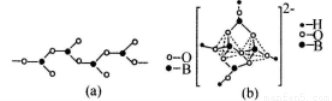
£Ø5£©NiO¾§Ģå½į¹¹ÓėNaCl¾§ĢåĄąĖĘ£¬Ę侧°ūµÄĄā³¤ĪŖacm£¬ŌņøĆ¾§ĢåÖŠ¾ąĄė×ī½üµÄĮ½øöŃōĄė×ÓŗĖ¼äµÄ¾ąĄėĪŖ_________£ØÓĆŗ¬ÓŠaµÄ“śŹżŹ½±ķŹ¾£©”£ŌŚŅ»¶ØĪĀ¶ČĻĀ£¬Ni0¾§ĢåæÉŅŌ×Ō·¢µŲ·ÖÉ¢²¢ŠĪ³É”°µ„·Ö×Ó²ć”±£ØČēĶ¼£©£¬æÉŅŌČĻĪŖŃõĄė×Ó×÷ĆÜÖĀµ„²ćÅÅĮŠ£¬ÄųĄė×ÓĢī³äĘäÖŠ£¬ĮŠŹ½²¢¼ĘĖćĆæĘ½·½Ć×Ć껿ÉĻ·ÖÉ¢µÄøĆ¾§ĢåµÄÖŹĮæĪŖ_________g£ØŃõĄė×ӵİė¾¶ĪŖ1. 40”Į10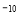 m£¬
m£¬ 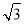 ”Öl. 732£©”£
”Öl. 732£©”£

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014-2015ѧğŗÓ±±Ź”øßČżĻĀѧʌȿµ÷æ¼ŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ijČÜŅŗÖŠæÉÄÜŗ¬ÓŠ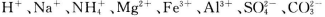 µČĄė×Ó”£µ±ĻņøĆČÜŅŗÖŠ¼ÓČėŅ»¶ØĪļÖŹµÄĮæÅØ¶ČµÄNaOHČÜŅŗŹ±£¬·¢ĻÖÉś³É³ĮµķĪļÖŹµÄĮæĖęNaOHČÜŅŗµÄĢå»ż±ä»ÆµÄĶ¼ĻńČēĶ¼ĖłŹ¾£¬ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ
µČĄė×Ó”£µ±ĻņøĆČÜŅŗÖŠ¼ÓČėŅ»¶ØĪļÖŹµÄĮæÅØ¶ČµÄNaOHČÜŅŗŹ±£¬·¢ĻÖÉś³É³ĮµķĪļÖŹµÄĮæĖęNaOHČÜŅŗµÄĢå»ż±ä»ÆµÄĶ¼ĻńČēĶ¼ĖłŹ¾£¬ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ

A£®ŌČÜŅŗÖŠŅ»¶Øŗ¬ÓŠµÄŃōĄė×ÓŹĒ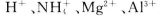
B£®ŌČÜŅŗÖŠŅ»¶Øŗ¬ÓŠSO42£ŗĶNa£«
C£®ŌČÜŅŗÖŠŗ¬ÓŠµÄFe3£«ŗĶAl3£«µÄĪļÖŹµÄĮæÖ®±ČĪŖ1:1
D£®·“Ó¦×īŗóŠĪ³ÉµÄČÜŅŗÖŠŗ¬ÓŠµÄČÜÖŹ½öĪŖNa2SO4
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014-2015ѧğŗÓ±±Ź”øßČżĻĀŃ§ĘŚĘŚÖŠæ¼ŹŌĄķ×Ū»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŹµŃéĢā
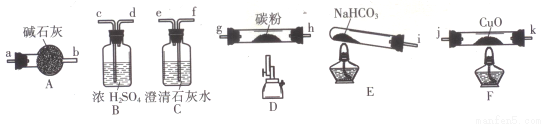
(14·Ö)ij»ÆѧŠ”×éÓĆĻĀĶ¼ĖłĮŠ×°ÖĆ(¹Ģ¶Ø×°ÖĆĀŌ)ŃéÖ¤COÄÜ»¹ŌCuO£¬²¢Ģ½¾æĘ仹Ō²śĪļµÄ×é³É”£
Ēė»Ų“š£ŗ
£Ø1£©°“ĘųĮ÷·½Ļņ£¬ø÷×°ÖĆ“Ó×óµ½ÓŅµÄĮ¬½ÓĖ³ŠņĪŖ£ŗ
i”ś( )( )”ś( )( )”ś( )( )”ś( )( )”ś( )( )Ī²Ęų“¦Ąķ(ĢīŅĒĘ÷½ÓæŚµÄ×ÖÄø±ąŗÅ)”£
£Ø2£©ĖµĆ÷COÄÜ»¹ŌCuOµÄŹµŃéĻÖĻóĪŖ___________________________”£
£Ø3£©øĆŹµŃ铦ĄķĪ²ĘųµÄ·½·ØĪŖ___________________________”£
£Ø4£©ČōČ„µōB×°ÖĆ£¬æÉÄܲśÉśµÄÓ°ĻģĪŖ___________________________”£
£Ø5£©½«ø÷×°ÖĆ°“ÕżČ·µÄĖ³ŠņĮ¬½Óŗó½ųŠŠŹµŃ锣µ±·“Ó¦½įŹųŗó£¬F“¦Ó²ÖŹ²£Į§¹ÜÖŠµÄ¹ĢĢåČ«²æ±äĪŖŗģÉ«”£
[²éŌÄ׏ĮĻ]CuŗĶCu2O¾łĪŖŗģÉ«£»Cu2OŌŚĖįŠŌČÜŅŗÖŠÄÜÉś³ÉCuŗĶCu2£«”£
[Ģį³ö²ĀĻė]ŗģÉ«¹ĢĢåµÄ³É·ÖæÉÄÜĪŖ£ŗ¢ŁCu£»¢ŚCu2O£»¢Ū________”£
[ŹµŃéŃéÖ¤]øĆŠ”×éĪŖŃéÖ¤ÉĻŹö²ĀĻė£¬·Ö±šČ”ÉŁĮæŗģÉ«¹ĢĢå·ÅČĖŹŌ¹ÜÖŠ£¬½ųŠŠČēĻĀŹµŃ锣
ŹµŃ鱹ŗÅ | ²Ł×÷ | ĻÖĻó |
a | ¼ÓČė×ćĮæĻ”ĻõĖį£¬Õńµ“ | ŹŌ¹ÜÄŚŗģÉ«¹ĢĢåĶźČ«Čܽā£¬ČÜŅŗ±äĪŖĄ¶É«£¬²¢ÓŠĪŽÉ«ĘųĢå²śÉś |
b | ¼ÓČė×ćĮæĻ”ĮņĖį£¬Õńµ“ | ŹŌ¹ÜÄŚÓŠŗģÉ«¹ĢĢ壬ČÜŅŗĪŖĪŽÉ« |
[ŹµŃéĘĄ¼Ū]øł¾ŻÉĻŹöŹµŃ飬²»ÄÜČ·¶ØŗģÉ«¹ĢĢå³É·ÖµÄŹµŃéŹĒ________(ĢīŹµŃ鱹ŗÅ)£¬ĄķÓÉĪŖ_________(ÓĆ»Æѧ·½³ĢŹ½±ķŹ¾)”£
[ŹµŃé½įĀŪ]øł¾ŻÉĻŹöŹµŃé½į¹ū£¬æÉÖŖŗģÉ«¹ĢĢåµÄ³É·ÖĪŖ________(Ģī»ÆѧŹ½)”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014-2015ѧğŗÓ±±Ź”øßČżĻĀŃ§ĘŚĘŚÖŠæ¼ŹŌĄķ×Ū»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ĻĀĮŠĄė×Ó·½³ĢŹ½²»ÕżČ·µÄŹĒ
A£®“æ¼īČÜŅŗÖŠµĪ¼Ó·ÓĢŖČÜŅŗĻŌŗģÉ«£ŗCO32 ££«H2O HCO3£ £«OH£
HCO3£ £«OH£
B£®ÉÕ¼īČÜŅŗÓėŠ”ĖÕ“ņČÜŅŗ»ģŗĻ£ŗOH££«HCO3£ =H2O£«CO32 £
C£®Įņ“śĮņĖįÄĘČÜŅŗÓėĻ”ĮņĖį»ģŗĻ£ŗ3S2O32 ££«6H£«=4S”ż£«3H2O£«2SO3
D£®ĒāŃõ»ÆĢśČÜÓŚĒāµāĖį£ŗ2Fe(OH)3£«6H£«£«2I£=2Fe2£«£«I2£«6H2O
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014-2015ѧğÕć½Ź”½š»ŖŹ®Š£øßČżĻĀѧʌøßæ¼Ä£Äā£Ø4ŌĀ£©Ąķ×Ū»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ŅŅČ©Ėį£ØHOOC£CHO£©ŹĒÓŠ»śŗĻ³ÉµÄÖŲŅŖÖŠ¼äĢ唣¹¤ŅµÉĻÓĆ”°Ė«¼«ŹŅ³É¶Ōµē½ā·Ø”±Éś²śŅŅČ©Ėį£¬ŌĄķČēĶ¼ĖłŹ¾”£øĆ×°ÖĆÖŠŅõ”¢ŃōĮ½¼«ĪŖ¶čŠŌµē¼«£¬Į½¼«ŹŅ¾łæɲśÉśŅŅČ©Ėį£¬ĘäÖŠŅŅ¶žČ©ÓėMµē¼«µÄ²śĪļ·“Ӧɜ³ÉŅŅČ©Ėį”£ĻĀĮŠĖµ·Ø²»ÕżČ·µÄŹĒ
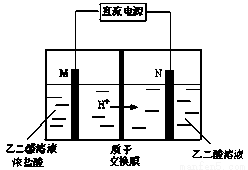
A£®Nµē¼«ÉĻµÄµē¼«·“Ó¦Ź½£ŗHOOC£COOH+2e£+2H+=HOOC£CHO+H2O
B£®ČōÓŠ2 mol H+ ĶعżÖŹ×Ó½»»»Ä¤²¢ĶźČ«²ĪÓė·“Ó¦£¬ŌņøĆ×°ÖĆÖŠÉś³ÉµÄŅŅČ©ĖįĪŖ1 mol
C£®Mµē¼«ÉĻµÄµē¼«·“Ó¦Ź½ĪŖ£ŗ2Cl££2e£=Cl2”ü
D£®ŅŅ¶žČ©ÓėMµē¼«µÄ²śĪļ·“Ӧɜ³ÉŅŅČ©ĖįµÄ»Æѧ·½³ĢŹ½£ŗ Cl2+OHC£CHO+ H2O = HOOC£CHO+2HCl
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ŌŚŅ»¶ØĢå»żµÄĆܱÕČŻĘ÷ÖŠ£¬½ųŠŠČēĻĀ»Æѧ·“Ó¦£ŗ
CO2£Øg£©+H2£Øg£©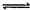 CO£Øg£©+H2O£Øg£©£¬Ęä»ÆŃ§Ę½ŗā³£ŹżKŗĶĪĀ¶ČtµÄ¹ŲĻµČēĻĀ±ķ£ŗ
CO£Øg£©+H2O£Øg£©£¬Ęä»ÆŃ§Ę½ŗā³£ŹżKŗĶĪĀ¶ČtµÄ¹ŲĻµČēĻĀ±ķ£ŗ
| t”ę | 700 | 800 | 830 | 1000 | 1200 |
| K | 0.6 | 0.9 | 1.0 | 1.7 | 2.6 |
»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©øĆ·“Ó¦µÄ»ÆŃ§Ę½ŗā³£Źż±ķ“ļŹ½ĪŖK=””””£»
£Ø2£©øĆ·“Ó¦ĪŖ””””·“Ó¦£ØŃ”ĢīĪüČČ”¢·ÅČČ£©£»
£Ø3£©Ä³ĪĀ¶ČĻĀ£¬Ę½ŗāÅØ¶Č·ūŗĻĻĀŹ½£ŗ3c£ØCO2£©•c£ØH2£©=5c£ØCO£©•c£ØH2O£©£¬ŹŌÅŠ¶Ļ“ĖŹ±µÄĪĀ¶ČĪŖ”””””ę£»
£Ø4£©830”ꏱ£¬ĻņøĆČŻĘ÷ÖŠ¼ÓČė1L CO2Óė1L H2£¬Ę½ŗāŹ±CO2µÄĢå»ż·ÖŹżŹĒ””£®
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com