
 »γΆΦ «Ρ≥―ß–Θ Β―ι “¥”Μ·―ß ‘ΦΝ…ΧΒξ¬ρΜΊΒΡ≈®ΝρΥα ‘ΦΝ±ξ«©…œΒΡ≤ΩΖ÷ΡΎ»ίΘ°œ÷”ΟΗΟ≈®ΝρΥα≈δ÷Τ100mL 1mol?L-1ΒΡœΓΝρΥαΘ°
»γΆΦ «Ρ≥―ß–Θ Β―ι “¥”Μ·―ß ‘ΦΝ…ΧΒξ¬ρΜΊΒΡ≈®ΝρΥα ‘ΦΝ±ξ«©…œΒΡ≤ΩΖ÷ΡΎ»ίΘ°œ÷”ΟΗΟ≈®ΝρΥα≈δ÷Τ100mL 1mol?L-1ΒΡœΓΝρΥαΘ°
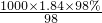 mol/L=18.4mol/LΘ°ΗυΨίœΓ ΆΕ®¬…Θ§œΓ Ά«ΑΚσ»ή÷ ΒΡΈο÷ ΒΡΝΩ≤Μ±δΘ§ά¥ΦΤΥψ≈®ΝρΥαΒΡΧεΜΐΘ§…η≈®ΝρΥαΒΡΧεΜΐΈΣxmLΘ§
mol/L=18.4mol/LΘ°ΗυΨίœΓ ΆΕ®¬…Θ§œΓ Ά«ΑΚσ»ή÷ ΒΡΈο÷ ΒΡΝΩ≤Μ±δΘ§ά¥ΦΤΥψ≈®ΝρΥαΒΡΧεΜΐΘ§…η≈®ΝρΥαΒΡΧεΜΐΈΣxmLΘ§ ΦΤΥψ≈®H2SO4ΒΡΈο÷ ΒΡΝΩ≈®Ε»Θ§‘ΌΗυΨί»ή“ΚœΓ Ά«ΑΚσΈο÷ ΒΡΝΩ≤Μ±δΦΤΥψΥυ–η≈®ΝρΥαΒΡΧεΜΐΘ°
ΦΤΥψ≈®H2SO4ΒΡΈο÷ ΒΡΝΩ≈®Ε»Θ§‘ΌΗυΨί»ή“ΚœΓ Ά«ΑΚσΈο÷ ΒΡΝΩ≤Μ±δΦΤΥψΥυ–η≈®ΝρΥαΒΡΧεΜΐΘ° Ζ÷Έω≈–ΕœΘ°
Ζ÷Έω≈–ΕœΘ° άμΫβ≈δ÷Τ‘≠άμΘ°ΉΔ“β≈®ΝρΥαΒΡœΓ ΆΘ°
άμΫβ≈δ÷Τ‘≠άμΘ°ΉΔ“β≈®ΝρΥαΒΡœΓ ΆΘ° 


| ΡξΦΕ | ΗΏ÷–ΩΈ≥Χ | ΡξΦΕ | ≥θ÷–ΩΈ≥Χ |
| ΗΏ“Μ | ΗΏ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ“Μ | ≥θ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏΕΰ | ΗΏΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θΕΰ | ≥θΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏ»ΐ | ΗΏ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ»ΐ | ≥θ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚ
 »γΆΦ «Ρ≥―ß–Θ Β―ι “¥”Μ·―ß ‘ΦΝ…ΧΒξ¬ρΜΊΒΡΝρΥα ‘ΦΝ±ξ«©…œΒΡ≤ΩΖ÷ΡΎ»ίΘ°Ψί¥ΥΘ§œ¬Ν–ΥΒΖ®’ΐ»ΖΒΡ «Θ®ΓΓΓΓΘ©
»γΆΦ «Ρ≥―ß–Θ Β―ι “¥”Μ·―ß ‘ΦΝ…ΧΒξ¬ρΜΊΒΡΝρΥα ‘ΦΝ±ξ«©…œΒΡ≤ΩΖ÷ΡΎ»ίΘ°Ψί¥ΥΘ§œ¬Ν–ΥΒΖ®’ΐ»ΖΒΡ «Θ®ΓΓΓΓΘ©≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚ

≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚ
 »γΆΦ «Ρ≥―ß–Θ Β―ι “¥”Μ·―ß ‘ΦΝ…ΧΒξ¬ρΜΊΒΡΝρΥα ‘ΦΝ±ξ«©…œΒΡ≤ΩΖ÷ΡΎ»ίΘ°Ψί¥Υœ¬Ν–ΥΒΖ®≤Μ’ΐ»ΖΒΡ «Θ®ΓΓΓΓΘ©
»γΆΦ «Ρ≥―ß–Θ Β―ι “¥”Μ·―ß ‘ΦΝ…ΧΒξ¬ρΜΊΒΡΝρΥα ‘ΦΝ±ξ«©…œΒΡ≤ΩΖ÷ΡΎ»ίΘ°Ψί¥Υœ¬Ν–ΥΒΖ®≤Μ’ΐ»ΖΒΡ «Θ®ΓΓΓΓΘ©≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚ‘ΡΕΝάμΫβ
 »γΆΦ «Ρ≥―ß–Θ Β―ι “¥”Μ·―ß ‘ΦΝ…ΧΒξ¬ρΜΊΒΡ≈®ΝρΥα ‘ΦΝ±ξ«©…œΒΡ≤ΩΖ÷ΡΎ»ίΘ°œ÷”ΟΗΟ≈®ΝρΥα≈δ÷Τ100mL 1mol?L-1ΒΡœΓΝρΥαΘ°
»γΆΦ «Ρ≥―ß–Θ Β―ι “¥”Μ·―ß ‘ΦΝ…ΧΒξ¬ρΜΊΒΡ≈®ΝρΥα ‘ΦΝ±ξ«©…œΒΡ≤ΩΖ÷ΡΎ»ίΘ°œ÷”ΟΗΟ≈®ΝρΥα≈δ÷Τ100mL 1mol?L-1ΒΡœΓΝρΥαΘ°
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚ
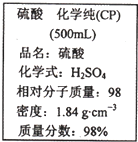 »γΆΦ «Ρ≥―ß–Θ Β―ι “¥”Μ·―ß ‘ΦΝ…ΧΒξ¬ρΜΊΒΡΝρΥα ‘ΦΝ±ξ«©…œΒΡ≤ΩΖ÷ΡΎ»ίΘ°Ψί¥Υœ¬Ν–ΥΒΖ®¥μΈσΒΡ «Θ®ΓΓΓΓΘ©
»γΆΦ «Ρ≥―ß–Θ Β―ι “¥”Μ·―ß ‘ΦΝ…ΧΒξ¬ρΜΊΒΡΝρΥα ‘ΦΝ±ξ«©…œΒΡ≤ΩΖ÷ΡΎ»ίΘ°Ψί¥Υœ¬Ν–ΥΒΖ®¥μΈσΒΡ «Θ®ΓΓΓΓΘ©| AΓΔΗΟΝρΥαΨΏ”–«ΩΝ“ΒΡΗ· ¥–‘Θ§”ΠΖ≈”ΎΈΘœ’Μ·―ß”ΟΤΖΙώ÷–ΆΉ…Τ±ΘΙή | BΓΔ»Γ10 mLΗΟΝρΥα”Ύ…’±≠÷–Θ§‘ΌΦ”Β»ΧεΜΐΒΡΥ°Θ§Ω…≈δΒΟ49%ΒΡΝρΥα | CΓΔ≈δ÷Τ200mL4.6 mol?L-1ΒΡœΓΝρΥα–η»ΓΗΟΝρΥα50mL | DΓΔΗΟΝρΥα”κΒ»÷ ΝΩΒΡΥ°ΜλΚœΥυΒΟ»ή“ΚΒΡΈο÷ ΒΡΝΩ≈®Ε»–Γ”Ύ9.2 mol?L-1 |
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΑΌΕ»÷¬–≈ - ΝΖœΑ≤αΝ–±μ - ‘ΧβΝ–±μ
Κΰ±± ΓΜΞΝΣΆχΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΤΫΧ® | Άχ…œ”–ΚΠ–≈œΔΨΌ±®Ή®«χ | Βγ–≈’©Τ≠ΨΌ±®Ή®«χ | …φάζ Ζ–ιΈό÷ς“ε”–ΚΠ–≈œΔΨΌ±®Ή®«χ | …φΤσ«÷»®ΨΌ±®Ή®«χ
ΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΒγΜΑΘΚ027-86699610 ΨΌ±®” œδΘΚ58377363@163.com