
K2CO3+6H2O![]() 2CH3OH+3O2+4KOH������գ�
2CH3OH+3O2+4KOH������գ�

ͼ5
(1)���ʱ����ԭ��صĸ������Դ____________���������������ĵ缫��ӦΪ________��
(2)�ŵ�ʱ�������ĵ缫��ӦʽΪ____________��
(3)�ڴ˹���������ȫ��Ӧ���ҳ���A������������648 g����׳�������������O2________L(��״����)��
(4)���ڳ��³�ѹ�£�1 g CH3OHȼ������CO2��Һ̬H2Oʱ����22.68 kJ����ʾ�÷�Ӧ���Ȼ�ѧ����ʽΪ_______________��
������(1)�ٸ� ��4OH--4e-====2H2O+O2
(2)CH3OH-6e-+8OH-====![]() +6H2O
+6H2O
(3)���ݵ��ӵ�ʧ�غ㣬�ã�
4Ag �� O2
![]() V(O2)
V(O2)
V(O2)=33.6 L
(4)CH3OH(l)+![]() O2(g)====CO2(g)+2H2O(l)����H=725.76 kJ��mol-1
O2(g)====CO2(g)+2H2O(l)����H=725.76 kJ��mol-1
�𰸣�(1)�� 4OH--4e-====2H2O+O2
(2)CH3OH-6e-+8OH-====![]() +6H2O
+6H2O
(3)33.6
(4)CH3OH(l)+![]() O2(g)====CO2(g)+2H2O(l)����H=725.76 kJ��mol-1
O2(g)====CO2(g)+2H2O(l)����H=725.76 kJ��mol-1



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| �ŵ� |
| ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
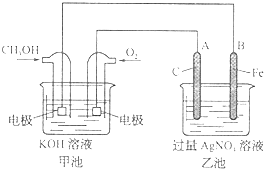 �ݱ�����Ħ��������˾������һ���Լ״�Ϊԭ�ϣ���KOHΪ����ʵ������ֻ��Ŀɳ��ĸ�Чȼ�ϵ�أ���һ�ε������ʹ��һ���£���ͼ��һ���绯ѧ���̵�ʾ��ͼ����֪�׳ص��ܷ�ӦʽΪ��2CH3OH+3O2+4KOH
�ݱ�����Ħ��������˾������һ���Լ״�Ϊԭ�ϣ���KOHΪ����ʵ������ֻ��Ŀɳ��ĸ�Чȼ�ϵ�أ���һ�ε������ʹ��һ���£���ͼ��һ���绯ѧ���̵�ʾ��ͼ����֪�׳ص��ܷ�ӦʽΪ��2CH3OH+3O2+4KOH| �ŵ� | ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

����գ�
(1)���ʱ����ԭ��صĸ������Դ________���������������ĵ缫��ӦΪ_____________��
(2)�ŵ�ʱ�������ĵ缫��ӦʽΪ__________________________________________��
(3)�ڴ˹���������ȫ��Ӧ���ҳ���A������������648 g����׳�������������O2_______L��(��״����)
(4)���ڳ��³�ѹ�£�1 g CH3OHȼ������CO2��Һ̬H2Oʱ����22.68 kJ,��ʾ�÷�Ӧ���Ȼ�ѧ����ʽΪ ________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�걱�����и߶���ѧ����ĩ���Ի�ѧ���� ���ͣ������
(7��)�ݱ�����Ħ��������˾������һ���Լ״�Ϊԭ�ϣ��� Ϊ����ʵ������ֻ��Ŀɳ��ĸ�Чȼ�ϵ�أ���һ�ε������ʹ��һ���¡���ͼ��һ���绯ѧ���̵�ʾ��ͼ����֪�׳ص��ܷ�ӦʽΪ��2CH3OH+3O2+4KOH
Ϊ����ʵ������ֻ��Ŀɳ��ĸ�Чȼ�ϵ�أ���һ�ε������ʹ��һ���¡���ͼ��һ���绯ѧ���̵�ʾ��ͼ����֪�׳ص��ܷ�ӦʽΪ��2CH3OH+3O2+4KOH 2K2CO3+6H2O
2K2CO3+6H2O
����գ�
��1���ŵ�ʱ�������ĵ缫��ӦʽΪ____ _____��
��2�����ʱ����ԭ��صĸ������Դ_________��������
�������ĵ缫��ӦΪ__________________��
��3���ڴ˹���������ȫ��Ӧ���ҳ���B������������648g����׳������������� _________L����״���£���
_________L����״���£���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�걱�����и߶���ѧ����ĩ���Ի�ѧ���� ���ͣ������
(7��)�ݱ�����Ħ��������˾������һ���Լ״�Ϊԭ�ϣ��� Ϊ����ʵ������ֻ��Ŀɳ��ĸ�Чȼ�ϵ�أ���һ�ε������ʹ��һ���¡���ͼ��һ���绯ѧ���̵�ʾ��ͼ����֪�׳ص��ܷ�ӦʽΪ��2CH3OH+3O2+4KOH
Ϊ����ʵ������ֻ��Ŀɳ��ĸ�Чȼ�ϵ�أ���һ�ε������ʹ��һ���¡���ͼ��һ���绯ѧ���̵�ʾ��ͼ����֪�׳ص��ܷ�ӦʽΪ��2CH3OH+3O2+4KOH 2K2CO3+6H2O
2K2CO3+6H2O

����գ�
��1���ŵ�ʱ�������ĵ缫��ӦʽΪ____ _____��
��2�����ʱ����ԭ��صĸ������Դ_________��������
�������ĵ缫��ӦΪ__________________��
��3���ڴ˹���������ȫ��Ӧ���ҳ���B������������648g����׳������������� _________L����״���£���
_________L����״���£���
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com