
 CO2 (g)+ H2(g) ��H 2 = ?41 kJ/mol����ʼʱ���ܱ������г���1.00 molCO��1.00 molH2O���ֱ��������ʵ�飬̽��Ӱ��ƽ������أ�����������ͬ�Ҳ������κθ���Ӧ��Ӱ�죩��ʵ���������±���
CO2 (g)+ H2(g) ��H 2 = ?41 kJ/mol����ʼʱ���ܱ������г���1.00 molCO��1.00 molH2O���ֱ��������ʵ�飬̽��Ӱ��ƽ������أ�����������ͬ�Ҳ������κθ���Ӧ��Ӱ�죩��ʵ���������±���| ʵ���� | �������/L | �¶�/��C |
| �� | 2.0 | 1200 |
| �� | 2.0 | 1300 |
| �� | 1.0 | 1200 |


 CO2 (g)+ H2(g) �ķ�Ӧ��һ�����ȷ�Ӧ�������¶����ߣ���Ӧ�ķ�Ӧ���ʼӿ죬��ʹƽ�������ƶ���ʵ������߱�ʵ������߸���ﵽƽ�⣬����ƽ��ʱCO2��Ũ��Ҫ��һЩ�����֮�£�ʵ��۱��������С���������������ѹǿ�����ڷ�Ӧǰ������ϵ�����䣬��������ѹǿֻ������ѧ��Ӧ���ʣ����Dz��ܸı�ƽ�⣬���ʵ��۵����߱�ʵ��ٸ���ﵽƽ�⣬��ƽ��ʱCO2��Ũ����ʵ��ٵ�2������ͼ
CO2 (g)+ H2(g) �ķ�Ӧ��һ�����ȷ�Ӧ�������¶����ߣ���Ӧ�ķ�Ӧ���ʼӿ죬��ʹƽ�������ƶ���ʵ������߱�ʵ������߸���ﵽƽ�⣬����ƽ��ʱCO2��Ũ��Ҫ��һЩ�����֮�£�ʵ��۱��������С���������������ѹǿ�����ڷ�Ӧǰ������ϵ�����䣬��������ѹǿֻ������ѧ��Ӧ���ʣ����Dz��ܸı�ƽ�⣬���ʵ��۵����߱�ʵ��ٸ���ﵽƽ�⣬��ƽ��ʱCO2��Ũ����ʵ��ٵ�2������ͼ
 CO2 (g)+ H2(g)
CO2 (g)+ H2(g)  ="0.44" ��2�֣�
="0.44" ��2�֣� =
= =" 1.0" ��1�֣�
=" 1.0" ��1�֣�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 C6 H5- CH=CH2 (g) +H2(g) ��H1
C6 H5- CH=CH2 (g) +H2(g) ��H1
 C6 H6 (g) ��H2
C6 H6 (g) ��H2 C6 H5- CH2CH3 (g) ��H3
C6 H5- CH2CH3 (g) ��H3 C6 H5- CH=CH2 (g) +H2(g) �Ħ�H= ��
C6 H5- CH=CH2 (g) +H2(g) �Ħ�H= ���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��4a��4b��4c | B��2a��2b��6c |
| C��2a��2b��2c | D��2a��6b��2c |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 CO(g)��H2(g)����C(s)��CO(g)��H2(g)��ȫȼ�յ��Ȼ�ѧ����ʽ�ֱ�Ϊ��
CO(g)��H2(g)����C(s)��CO(g)��H2(g)��ȫȼ�յ��Ȼ�ѧ����ʽ�ֱ�Ϊ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 CH3OH(g)����H1
CH3OH(g)����H1 CH3OH(g)��H2O(g)����H2
CH3OH(g)��H2O(g)����H2| �¶� | 250 �� | 300 �� | 350 �� |
| K | 2.041 | 0.270 | 0.012 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

 Fe3+(aq)+3OH-(aq) �� ��H=" a" kJ?mol-1
Fe3+(aq)+3OH-(aq) �� ��H=" a" kJ?mol-1 H+(aq)+OH-(aq) ����H=" b" kJ?mol-1
H+(aq)+OH-(aq) ����H=" b" kJ?mol-1�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 CH3OCH3��H2O
CH3OCH3��H2O CH3OCH3(g)��CO2(g) ��H����247kJ/mol
CH3OCH3(g)��CO2(g) ��H����247kJ/mol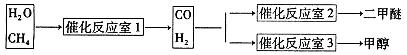
 CH3OCH3(g)��CO2(g)���ܱ������дﵽƽ���Ҫ���CO��ת���ʣ����Բ�ȡ�Ĵ�ʩ��
CH3OCH3(g)��CO2(g)���ܱ������дﵽƽ���Ҫ���CO��ת���ʣ����Բ�ȡ�Ĵ�ʩ��  CH3OH(g)��H2O (g) ��H��0��Ӧ�ﵽƽ��ʱ���ı��¶ȣ�T����ѹǿ��P������Ӧ�����CH3OH�����ʵ����������仯�����ͼ��ʾ�������¶ȣ�T����ѹǿ��P���Ĺ�ϵ�ж���ȷ���� ������ţ�
CH3OH(g)��H2O (g) ��H��0��Ӧ�ﵽƽ��ʱ���ı��¶ȣ�T����ѹǿ��P������Ӧ�����CH3OH�����ʵ����������仯�����ͼ��ʾ�������¶ȣ�T����ѹǿ��P���Ĺ�ϵ�ж���ȷ���� ������ţ�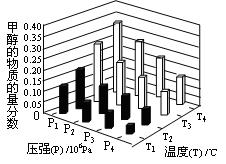
 CO(g)��3H2(g) ��H��0д��ƽ�ⳣ���ı���ʽ��
CO(g)��3H2(g) ��H��0д��ƽ�ⳣ���ı���ʽ�� 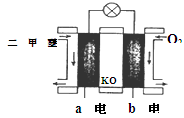
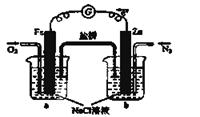
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 2H2��+O2��
2H2��+O2�� 2H2��+O2��
2H2��+O2�� 2H2��+O2��
2H2��+O2�� CO+3H2
CO+3H2 CH3OH��g��+H2O��g������H="-49.0" kJ/mol,���CO2��CH3OH��g����Ũ����ʱ��仯��ͼ��ʾ��
CH3OH��g��+H2O��g������H="-49.0" kJ/mol,���CO2��CH3OH��g����Ũ����ʱ��仯��ͼ��ʾ��
 CH3OH��g����ƽ�ⳣ��:
CH3OH��g����ƽ�ⳣ��:| �¶�/�� | 0 | 100 | 200 | 300 | 400 |
| ƽ�ⳣ�� | 667 | 13 | 1.9��1 | 2.4��1 | 1��1 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com