
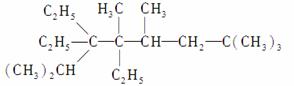
(1)д�����л���������ƻ�ṹ��ʽ��
��
________________________________________________________________________��
��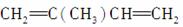 ________________________________________________________________________��
________________________________________________________________________��
��CH2===CHCOOH________________________________________________________________________��
��2,5���� 2,4����ϩ�Ľṹ��ʽ��
________________________________________________________________________
________________________________________________________________________��
(2)������ֳƻƼ���ҹ��ض�����ҩ������������е�һ���������ҹ���ѧ���о�������ṹ���£�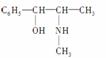
��������к��������ŵ�������________������________��(������ӡ�)��
�����и����ʣ�
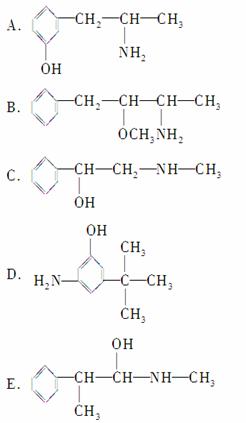
������ػ�Ϊͬ���칹�����________(����ĸ����ͬ)����Ϊͬϵ�����________��
 ǧ�������������ĩ�����Ծ�����ϵ�д�
ǧ�������������ĩ�����Ծ�����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������ˮ��Һ�п��ܴ��ڵ���ƽ�⡢�ε�ˮ��ƽ�������ܽ�ƽ�⣬���Ƕ��ɿ�����ѧƽ���һ�֡��������ѧ��ѧ֪ʶ�ش��������⣺
(1)AΪ0.1 mol��L��1��(NH4)2SO4��Һ���ڸ���Һ������Ũ���ɴ�С��˳��Ϊ
________________________________________________________________________��
(2)BΪ0.1 mol��L��1��NaHCO3��Һ��NaHCO3�ڸ���Һ�д��ڵ�ƽ����(�����ӷ���ʽ��
ʾ)________________________________________________________________________
________________________________________________________________________
________________________________________________________________________��
(3)CΪ0.1 mol��L��1��(NH4)2Fe(SO4)2��Һ����ͬŨ�ȵ�(NH4)2SO4��Һ��Ƚ�________(��
���ʵĻ�ѧʽ)��Һ��NH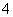 ��Ũ�ȸ�����ԭ����
��Ũ�ȸ�����ԭ����
________________________________________________________________________
________________________________________________________________________��
(4)DΪ��������AgCl����ı�����Һ��AgCl����Һ�д�������ƽ�⣺AgCl(s)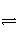
 Ag��(aq)��Cl��(aq)
Ag��(aq)��Cl��(aq)
��25��ʱ��AgCl��Ksp��1.8��10��10���ֽ�����AgCl�ֱ��������Һ���У���100 mL����ˮ����100 mL 0.3 mol��L��1AgNO3��Һ����100 mL 0.1 mol��L��1 MgCl2��Һ��ֽ������ȴ����ͬ�¶ȣ�Ag��Ũ���ɴ�С��˳��Ϊ________(�����)����ʱ��Һ����Cl�����ʵ���Ũ��Ϊ______________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
1.01��105 Pa��150��ʱ����1 L C2H4��2 L C2H6��20 L O2��ϲ���ȼ����ȫ��Ӧ��O2��ʣ�ࡣ����Ӧ��Ļ������ָ���ԭ����ʱ���������Ϊ(����)
A��15 L B��20 L
C��23 L D��24 L
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij�л���Ľṹ��ʽΪ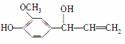 ���������ŷ��࣬��������(����)
���������ŷ��࣬��������(����)
A��ϩ�� B������
C������ D������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
һ�ȴ�����2�֣����ȴ�����4�ֵ�����(����)
A������ B��2������
C����ϩ D����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij�л���3.24 gװ��Ԫ�ط���װ�ã�ͨ������������ʹ֮��ȫȼ�գ������ɵ���������ͨ����ˮCaCl2(A)�ܺͼ�ʯ��(B)�ܣ����A������2.16 g��B������9.24 g����֪���л������Է�������С��200������л����ʵ��ʽ�ͻ�ѧʽ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij��ѧ��ȤС�鰴�����з������С��ɺ��������Ʊ����������塱��ʵ�飺
����1��ȡһ����������������������NaOH��Һ����Ӧ��ȫ����ˡ�
����2���߽��������Һ�еμ�ϡ��������Һ��pH=8��9�����á����ˡ�ϴ�ӡ�
����3��������2�еõ��Ĺ�������������ϡ���ᡣ
����4�� ��
��ش��������⣺
��1������ʵ���еĹ��˲�����Ҫ�������� �Ȳ���������
��2������1������Ӧ�����ӷ���ʽΪ
���˵�Ŀ����__________________________________________��
��3��������2�е���ҺpH=8��9ʱ����������Ƿ���ȫ�ķ�����______________��
��4������2����Һ��pH���ƽ��Ѳ������ɸ���____________________��������Ӧ�����ӷ���ʽΪ ��
��5������4�IJ���Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������A��B���⣬�ֱ��Ӧ�ڡ����ʽṹ�����ʡ��͡�ʵ�黯ѧ������ѡ�γ�ģ������ݣ�ÿ��12�֡���ѡ������һ�����������ⶼ���𡣽���A�����֡�
A�������ʽṹ�����ʡ�
���������з�Ӧ�ϳ������谷��CaO��3C CaC2��CO����CaC2��N2
CaC2��CO����CaC2��N2  CN2��C��CaCN2��2H2O===NH2CN��Ca(OH)2��NH2CN��ˮ��Ӧ��������[CO(NH2)2]�����غϳ������谷��
CN2��C��CaCN2��2H2O===NH2CN��Ca(OH)2��NH2CN��ˮ��Ӧ��������[CO(NH2)2]�����غϳ������谷��
��1��д����Ca��ͬһ������������������ͬ���ڲ��������ӵĻ�̬ԭ�ӵĵ����Ų�ʽ�� �� CaCN2��������ΪCN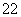 �����ݵȵ���ԭ��������֪CN
�����ݵȵ���ԭ��������֪CN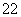 �Ŀռ乹��Ϊ ��
�Ŀռ乹��Ϊ ��
 ��2�����ط�����Cԭ�Ӳ�ȡ �ӻ������ط��ӵĽṹ��ʽ�� ��
��2�����ط�����Cԭ�Ӳ�ȡ �ӻ������ط��ӵĽṹ��ʽ�� ��
��3�������谷( )�׳ơ����������������������谷����������( )
�����������������谷�����֮��ͨ�� ��ϣ������������γɽ�ʯ��
��4��CaO������ͼ��ʾ��CaO������Ca2+����λ��Ϊ ��
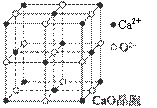

��5��CaO�����NaCl����ľ����ֱܷ�Ϊ��CaO 3401 kJ·mol-1��NaCl 786 kJ·mol-1������CaO��NaCl�����ܴ����Ҫԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����ڵ綯������������ȼ�ϵ�أ�ͨ����NaCl��Һ��NaOH��ҺΪ���Һ�����Ͻ�Ϊ�����������缫Ϊ����������˵����ȷ����(����)
A����NaCl��Һ��NaOH��ҺΪ���Һʱ��������Ӧ��Ϊ��O2��2H2O��4e��===4OH��
B����NaOH��ҺΪ���Һʱ��������ӦΪ��Al��3OH����3e��===Al(OH)3
C����NaOH��ҺΪ���Һʱ������ڹ��������е��Һ��pH���ֲ���
D����ع���ʱ������ͨ�����·����������
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com