
�п�Ժ��������ѧ����2013��11��22�������������Ŀ�����Ա�ڹ������״Ρ��ġ�������ġ���Ƭ����ʵ���������ʵ�ռ����Ϊ������ı��ʡ���һ��ѧ��������80����������ṩ��ֱ��֤�ݡ��ⲻ������������������ʶ��ǰ�ƽ���һ��ҲΪ�ڷ��ӡ�ԭ�ӳ߶��ϵ��о��ṩ�˸���ȷ�ķ���������˵������ȷ����(����)
A����������Ĵ��ڣ����ܸ���ˮ����
B���������Ȼ��������Ҫ��������㷺�Ļ�ѧ��֮һ
C����������Ĵ��ڣ��е㣺HCl>HBr>HI>HF
D����������Ĵ��ڣ�ˮ����������������104.5��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ʳ�����ճ�����ı���Ʒ��Ҳ����Ҫ�Ļ���ԭ�ϡ���ʳ�γ���������K����Ca2����Mg2����Fe3����SO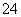 ���������ӣ�ʵ�����ᴿNaCl���������£�
���������ӣ�ʵ�����ᴿNaCl���������£�
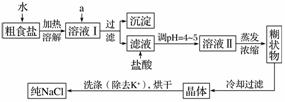
�ṩ���Լ�������Na2CO3��Һ������K2CO3��Һ��NaOH��Һ��BaCl2��Һ��Ba(NO3)2��Һ��75%�Ҵ������Ȼ�̼
(1)����ȥ��Һ���е�Ca2����Mg2����Fe3����SO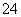 ��ѡ��a���������Լ������μ�˳������Ϊ____________(ֻ�ѧʽ)��
��ѡ��a���������Լ������μ�˳������Ϊ____________(ֻ�ѧʽ)��
(2)���ڳ������Լ���������жϳ�ȥSO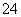 ���Լ������ķ�����________________________________________________________________________��
���Լ������ķ�����________________________________________________________________________��
(3)ϴ�ӳ�ȥNaCl������渽��������KCl��ѡ�õ��Լ�Ϊ________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����ϵ������¶ȣ�ʵ�֡�ѩ����ˮ��ˮ�������������������ı仯���ڱ仯�ĸ��α��ƻ������Ӽ����Ҫ�����������(����)
A����������Ӽ������������Լ�
B�������������Ǽ��Լ�
C����������Լ������Ӽ�������
D�����Ӽ���������������Ǽ��Լ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ά����B1����Ϊ��ø�����ǵĴ�л�����б�����ϵͳ�����á������ʵĽṹʽΪ
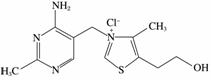
���¹���ά����B1��˵����ȷ����________��
A��ֻ���Ҽ��ͦм�
B�����й��ۼ��������Ӽ�
C�������ʵ��۵���ܸ���NaCl
D������������������
(3)ά����B1��������ˮ�Ĺ�����Ҫ�˷���������������________��
A�����Ӽ������ۼ� B�����Ӽ�����������ۼ�
C����������»��� D�����Ӽ�����������»���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��֪X�Ļ�̬ԭ��L���������K���������2����Y�Ļ�̬ԭ�����������Ų�ʽΪnsnnpn��2����X��Y����̬�⻯���У����ȶ�����________(�ѧʽ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����йئҼ���˵��������� (����)
A�����������ͼ����������s�����ص��γɵģ����γ�ss �Ҽ�
B��s������p�����γ�sp �Ҽ�
C��p��p�����γɦҼ�
D��HCl�����ﺬ��һ��sp �Ҽ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ԭ�ӽṹ��Ԫ�����ڱ�������������ϵ��������ѧ���ʽṹ֪ʶ������ش��������⣺
(1)�밴Ҫ������дһ����Ӧ���ʣ�(�ѧʽ)
���зǼ��Լ������ӻ�����________���Ⱥ��зǼ��Լ��ֺ����Լ��ķǼ��Է���________���Ⱥ��зǼ��Լ��ֺ����Լ��ļ��Է���________��ȫ���ɷǽ���Ԫ����ɵ����ӻ�����________���ɽ���Ԫ�غͷǽ���Ԫ����ɵĹ��ۻ�����________��
(2)�յ�����ɫ���ޡ��۸����������һЩ��ҵ�Ƿ���ΪʳƷ�ͻ�ױƷ�ȵ�Ⱦɫ��������Σ�����ǽ������յ��쳣���Т�4�����ͣ��յ����ķ��ӽṹ��ͼ1��ʾ��

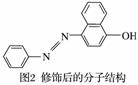
�յ������ˮ�е��ܽ�Ⱥ�С�������Ҵ������˰��ǻ�ȡ���ڶ�λ�γ�ͼ2��ʾ�Ľṹ��������ˮ�е��ܽ�Ȼ�________(�����С��)��ԭ����______________��
(3)��֪Ti3�����γ���λ��Ϊ6����ɫ��ͬ����������ᄃ�壬һ��Ϊ��ɫ����һ��Ϊ��ɫ�����־������ɽ�ΪTiCl3·6H2O��Ϊ�ⶨ�����־���Ļ�ѧʽ�����������ʵ�飺
a���ֱ�ȡ����������������ᄃ�����Ʒ��ɴ�����Һ��
b���ֱ���������Һ�е���AgNO3��Һ����������ɫ������
c��������ȫ��ֱ���˵����ݳ�������ϴ�Ӹ�������������ԭ��ɫ�����ˮ��Һ�õ��İ�ɫ��������Ϊԭ��ɫ�����ˮ��Һ�õ��ij���������2/3������ɫ���������Ļ�ѧʽΪ________________����Cl�����γɵĻ�ѧ��������________��
(4)��ͼ��A��B��C��D�������߷ֱ��ʾ�ڢ�A����A����A����A��Ԫ�ص��⻯��ķе㣬���б�ʾ��A��Ԫ���⻯��е��������______����ʾ��A��Ԫ���⻯��е��������______��ͬһ���е�3��4��5����Ԫ�ص��⻯��е��������ߣ���ԭ����________________________________________________________________________��
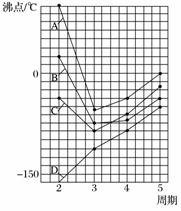
A��B��C�����е�2����Ԫ�ص��⻯��ķе��������ڵ�3����Ԫ�ص��⻯��ķе㣬��ԭ����________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�ж�������ȷ�Ļ����̡�������Ļ�������
(1)ͬ����Ԫ�أ������ң�ԭ�Ӱ뾶��С�����Ӱ뾶Ҳ��С(����)
(2)���Ӳ���Խ�࣬�뾶Խ��(����)
(3)������Ԫ���У���������ϼ۾�������������(����)
(4)Ԫ�ص�ԭ�ӵõ���Խ�࣬�ǽ�����Խǿ��ʧ����Խ�࣬������Խǿ(����)
(5)Ԫ�ص��������Ӧ��ˮ��������Խǿ���ǽ�����Խǿ������Խǿ��������Խǿ(����)
(6)Ԫ�ص���̬�⻯��Խ�ȶ����ǽ�����Խǿ����ˮ��Һ������Խǿ����ԭ��Խ��(����)
(7)��Mg��Al��NaOH��Һ���ɵ�ԭ����У���ΪAl��������Mg������������Al�Ľ����Դ���Mg(����)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
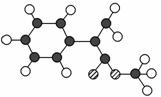 A������ij���������ϵĻ���ԭ��֮һ������ʽΪC10H10O2������ӽṹģ����ͼ��ʾ(ͼ��������֮�����ߴ���������˫��)��
A������ij���������ϵĻ���ԭ��֮һ������ʽΪC10H10O2������ӽṹģ����ͼ��ʾ(ͼ��������֮�����ߴ���������˫��)��
��.���ݷ��ӽṹģ��д��A�Ľṹ��ʽ______________________________________________________��
��.��ӷ�����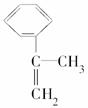 �������ϳ�A����ϳ�·�����£�
�������ϳ�A����ϳ�·�����£�
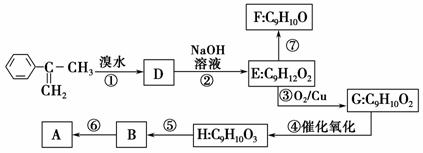
��֪��A������������ˮ�������л���B�ͼ״���
(1)д���ݵķ�Ӧ����_______________________________________��
(2)д��H�Ľṹ��ʽ_______________________________��
(3)��֪F�����к��С�—CH2OH����ͨ��F������Ч��˳���ػ��B����ԭ����___________________________________________________��
(4)д����Ӧ����ʽ(ע����Ҫ�ķ�Ӧ����)��
��___________________________________________________________��
��________________________________________________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com