
̼��̼�Ļ�����㷺�Ĵ��������ǵ������С�
��1���������з�Ӧ�������仯ʾ��ͼ��2C(s) +O2(g) =2CO(g) ��H= ��
��2�������Ϊ2L���ܱ������У�����1 mol CO2��3mol H��һ�������·�����Ӧ�� CO2(g)+3H2(g) CH3OH(g)+H2O(g) ��H<O���CO2(g)��CH3OH(g)�����ʵ�����ʱ��仯��������ͼ��ʾ��
CH3OH(g)+H2O(g) ��H<O���CO2(g)��CH3OH(g)�����ʵ�����ʱ��仯��������ͼ��ʾ��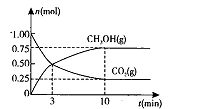
�ٴӷ�Ӧ��ʼ��ƽ�⣬H2O��ƽ����Ӧ����v(H2O)= ��
�����д�ʩ����ʹ��ѧƽ��������Ӧ�����ƶ����� �����ţ���
A�������¶� B����CH3OH��g����ʱҺ���Ƴ�
C��ѡ���Ч���� D���ٳ���l mol CO2��4 mol H2
��3��CO2����ˮ����̼�ᡣ��֪�������ݣ�
| ������� | H2CO3 | NH3��H2O |
| ����ƽ�ⳣ���� 25�棩 | Ka1 = 4��30 �� 10һ7 Ka2= 5��61�� 10һ11 | Kb = 1��77�� 10һ5 |
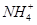 ˮ���ƽ�ⳣ��Kh=Kw/Kb��
ˮ���ƽ�ⳣ��Kh=Kw/Kb��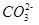
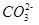 ������һ��ˮ������ӷ���ʽ ��
������һ��ˮ������ӷ���ʽ ��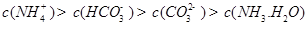

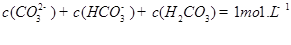
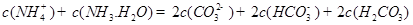
��1��-221.0kJ/mol (2)��0.0375mol/(L��min) �� B D  (3) �ټ� CO32-+H2O
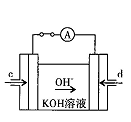
(3) �ټ� CO32-+H2O HCO3-+OH- �� C D (4) d CO-2e-+4OH-=CO32-+2H2O
HCO3-+OH- �� C D (4) d CO-2e-+4OH-=CO32-+2H2O
�������������������һ���ۺ��Խ�ǿ������ѧ������ѧ֪ʶ���ۺ�Ӧ����������1�����������仯��ͼ��ȷ��C(s)+O2(g)=CO2(g) ��H="-393.5kJ/mol," C(s)+CO2(g)="2CO" (g) ��H=+172.5kJ/mol,�ɸ�˹���ɵ�2C(s) +O2(g) =2CO(g) ��H=-221.0kJ/mol����2�����ݻ�ѧ��Ӧ�����ʱ���ʽ����ȷ����Ӧv(H2O)����3�����̼���Ka1 = 4��30 �� 10һ7��NH3��H2O��Kb = 1��77�� 10һ5��֪( NH4)2CO3��Һ��ʾ���ԣ����������غ㣬ѡ��C��D��ȷ����4����ԭ���ԭ������Ϊ�������ҺΪKOH������ȷ���缫��ӦʽΪCO-2e-+4OH-=CO32-+2H2O��
���㣺���黯ѧ��Ӧ�е������仯����ѧ��Ӧ���ʼ���ѧƽ����ƶ��������ˮ�⡢����Ũ�ȵĴ�С�Ƚϡ�


 �·Ƿ��̸����100��ϵ�д�
�·Ƿ��̸����100��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
Ϊ�˷�ֹ����ٻ�����β����ȼú�����������Կ�������Ⱦ�����Dz�ȡ�˺ܶ��ʩ��
��1������β����������Ҫԭ��Ϊ��2NO(g) + 2CO(g) 2CO2(g)+ N2(g) ��H��0��
2CO2(g)+ N2(g) ��H��0��
���÷�Ӧ�ھ��ȡ����ݵ��ܱ���ϵ�н��У�����ʾ��ͼ��ȷ����˵����Ӧ�ڽ��е�t1ʱ�̴ﵽƽ��״̬���� ������ţ���
����ͼ�Ц�����K��n��w�ֱ��ʾ����Ӧ���ʡ�ƽ�ⳣ�������ʵ���������������
��2��������β����úȼ�ղ����������������������CH4����ԭNOX�������������������Ⱦ����֪��
CH4(g)+2NO2(g)��N2(g)��CO2(g)+2H2O(g) ��H����867 kJ/mol ��
2NO2(g) N2O4(g) ��H����56.9 kJ/mol ��
N2O4(g) ��H����56.9 kJ/mol ��
H2O(g) �� H2O(l) ��H����44.0 kJ��mol ��
д��CH4����ԭN2O4(g)����N2��H2O(l)���Ȼ�ѧ����ʽ�� ��
��3����NH3����ԭNOXҲ�������������������Ⱦ����ͼ������NH3����ԭ����������һ��������ͨ��������ͨ�������ݳ������е������ﺬ�����Ӷ���ȷ�������ѵ��ʣ���Ӧԭ��Ϊ��NO(g) +NO2(g)+2NH3(g) 2N2(g) + 3H2O(g)��
2N2(g) + 3H2O(g)��
�ٸ÷�Ӧ�ġ�H 0����������������� ����������
�ڶ������巴Ӧ����ij���(B)��ƽ��ѹǿ(pB)�������ʵ���Ũ��(cB)Ҳ���Ա�ʾƽ�ⳣ��������KP����
��������Ӧ��KP�� ��
��4��NO2��O2������NaNO3������ȼ�ϵ�أ���ԭ����ͼ��ʯīIΪ��ص� ���� �õ����ʹ�ù�����ʯīI�缫������������Y����缫��ӦΪ ��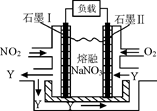
��5�����Ṥҵβ���е������NO��NO2���������ء�CO(NH2)2����Һ��ȥ����Ӧ���ɶԴ�������Ⱦ�����塣1 mol���������չ�ҵβ���е����������NO��NO2�����Ϊ1�U1��������Ϊ g��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�±��е��������ƻ�1 mol�����еĻ�ѧ�������ĵ�����(kJ):
| ���� | Cl2 | Br2 | I2 | HCl | HBr | HI | H2 |
| ����/kJ | 243 | 193 | 151 | 432 | 366 | 298 | 436 |
 2HX(X����Cl��Br��I )�ķ�Ӧ�����ȷ�Ӧ���Ƿ��ȷ�Ӧ?��:��______________��
2HX(X����Cl��Br��I )�ķ�Ӧ�����ȷ�Ӧ���Ƿ��ȷ�Ӧ?��:��______________�� �鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(����Ԥ����)��úת��Ϊˮú������Ҫ��ѧ��ӦΪC(s)��H2O(g) CO(g)��H2(g)��C(s)��CO(g)��H2(g)��ȫȼ�յ��Ȼ�ѧ����ʽΪ��
CO(g)��H2(g)��C(s)��CO(g)��H2(g)��ȫȼ�յ��Ȼ�ѧ����ʽΪ��
C(s)��O2(g)=CO2(g)����H1����393.5 kJ��mol��1
H2(g)�� O2(g)=H2O(g)����H����242.0 kJ��mol��1
O2(g)=H2O(g)����H����242.0 kJ��mol��1
CO(g)�� O2(g)=CO2(g)����H����283.0 kJ��mol��1
O2(g)=CO2(g)����H����283.0 kJ��mol��1
��ش�
(1)�����������ݣ�д��C(s)��ˮ������Ӧ���Ȼ�ѧ��Ӧ����ʽ��_____________________________��
(2)�ȽϷ�Ӧ�����ݿ�֪��1 mol CO(g)��1 mol H2(g)��ȫȼ�շų�������֮�ͱ�1 mol C(s)��ȫȼ�շų��������ࡣ��ͬѧ�ݴ���Ϊ��úת��Ϊˮú������ʹúȼ�շų����������������ͬѧ���ݸ�˹����������ͼѭ��ͼ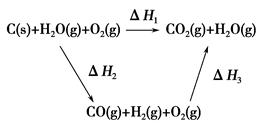
���ݴ���Ϊ��úת��Ϊˮú����ȼ�շų���������úֱ��ȼ�շų���������ȡ���
��������ס�����ͬѧ�۵���ȷ����________(��ס����ҡ�)���жϵ�������
_______________________________________________________��
(3)��úת��Ϊˮú����Ϊȼ�Ϻ�úֱ��ȼ������кܶ��ŵ㣬���о����е������ŵ�______________________________________________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��һֻ�Թ��з��뼸С��þƬ�����Թܷ���ʢ��25 ��ı���ʯ��ˮ���ձ��У��ý�ͷ�ιܵ�5 mL�������Թ��С��Իش��������⣺
��1��ʵ���в�����������________________________________________��
��2���������������ԭ����______________________________________��
��3��д���йص����ӷ���ʽ��____________________________________��
��4����ʵ����֪��MgCl2��Һ��H2��������________�������������������þƬ���������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�о�CO2�����öԴٽ���̼���Ĺ���������Ҫ���塣
��1����CO2�뽹̿��������CO��CO�����������ȡ�
��֪��Fe2O3(s) + 3C(ʯī) =" 2Fe(s)" + 3CO(g) ��H 1 =" +489.0" kJ��mol��1
C(ʯī) +CO2(g) = 2CO(g) ��H 2 =" +172.5" kJ��mol��1
��CO��ԭFe2O3(s)���Ȼ�ѧ����ʽΪ ��
��2��������̼�ϳɼ״���̼���ŵ��·���CO2ת��Ϊ�״����Ȼ�ѧ����ʽ
CO2(g) +3H2(g) CH3OH(g) +H2O(g) ��H
CH3OH(g) +H2O(g) ��H
�ٸ÷�Ӧ��ƽ�ⳣ������ʽΪK= ��
��ȡһ�����CO2��H2�Ļ������(���ʵ���֮��Ϊ1��3)����������ܱ������У�����������Ӧ����Ӧ�����в�ü״������������(CH3OH)�뷴Ӧ�¶�T�Ĺ�ϵ��ͼA��ʾ����÷�Ӧ�Ħ�H 0���>������<����������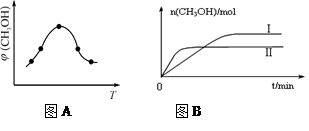
�������ֲ�ͬ�����·�����Ӧ�����CH3OH�����ʵ�����ʱ��仯��ͼB��ʾ������I�����Ӧ��ƽ�ⳣ����С��ϵΪK�� K�����>�� ��<������
��3����CO2Ϊԭ�ϻ����Ժϳɶ������ʡ��ٹ�ҵ������[CO(NH2)2]��CO2��NH3��һ�������ºϳɣ��䷴Ӧ����ʽΪ ������̼��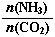 ��3����ƽ��ʱCO2��ת����Ϊ60%����NH3��ƽ��ת����Ϊ ��
��3����ƽ��ʱCO2��ת����Ϊ60%����NH3��ƽ��ת����Ϊ ��
����������Һ������ʽ��е�⣬CO2�ڵ缫�Ͽ�ת��Ϊ���飬�õ缫��Ӧ�ķ���ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�½�����ġ�ǰ�ؿ�ѧ����־�������й�������ѧ�о�Ժ�о�Ա�����ġ�����β����Ⱦ����Σ����������ϵͳ�ز���������β���ŷŶԴ������������彡����ɵ�����Σ����Ŀǰ����β���Ŀ��з������������������ϰ�װ��ת������NO��CO�����Ϊ����β���ijɷ֣������������ڴ�ת�����з�����Ӧ��2CO(g)��2NO(g) N2(g)��2CO2(g) ��H=��a kJ��mol-1��
N2(g)��2CO2(g) ��H=��a kJ��mol-1��
��1��CO2�ĵ���ʽΪ ��
��2����֪2NO(g)��O2(g)=2NO2(g) ��H=��b kJ��mol-1��CO��ȼ�� �ȡ�H=��c kJ��mol-1����д����������β����NO2����Ⱦʱ��NO2��CO�Ŀ��淴Ӧ���Ȼ�ѧ��Ӧ����ʽ ��
��3����һ���¶��£���2.0mol NO��2.4mol����COͨ�뵽�̶��ݻ�Ϊ2L�������У���Ӧ�����в������ʵ�Ũ�ȱ仯��ͼ��ʾ��
���к�����NO��ת����Ϊ ��0~15min NO��ƽ������v(NO)= ��
��20minʱ�����ı䷴Ӧ����������COŨ�ȼ�С����ı������������ ��ѡ����ţ���
a����С������� b������CO���� c�������¶� d�������������
�������ַ�Ӧ��ϵ���¶Ȳ��䣬20minʱ���������г���NO��N2��0.4mol����ѧƽ�⽫ �ƶ���ѡ����������ҡ��������� �ƶ����ڴﵽƽ��ʱ��ƽ�ⳣ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�о�SO2��CO�ȴ�����Ⱦ��Ĵ��������þ����ش����塣
��.�����Ƽ�ѭ�������ѳ�������SO2���÷���Na2SO3��Һ��Ϊ���ռ������չ���pH��n��SO��?n��HSO3-���仯��ϵ���±���
| n��SO32-��?n��HSO3-�� | 91:9 | 1:1 | 9:91 |
| pH | 8.2 | 7.2 | 6.2 |
 CH3OH��g����
CH3OH��g����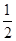 O2��g��=H2O��g������H����241.8 kJ/mol��
O2��g��=H2O��g������H����241.8 kJ/mol��| ��ѧ�� | H��H | H��O | C��H | C��O | C=O |
| ���� | 435 | 463 | 413 | 356 | 745 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�״�ȼ�Ϸ�Ϊ�״����ͺͼ״����͡���ҵ�Ϻϳɼ״��ķ����ܶࡣ
��1��һ�������·�����Ӧ��
��CO2��g�� +3H2��g�� ��CH3OH��g��+H2O��g�� ��H1
��2CO��g�� +O2��g�� ��2CO2��g�� ��H2
��2H2��g��+O2��g�� ��2H2O��g�� ��H3
��CO��g�� + 2H2��g��  CH3OH��g�����ġ�H�� ��
CH3OH��g�����ġ�H�� ��
��2�����ݻ�Ϊ2L���ܱ������н��з�Ӧ�� CO��g��+2H2��g�� CH3OH��g�� �������������䣬��300���500��ʱ�����ʵ���n��CH3OH�� �뷴Ӧʱ��t�ı仯������ͼ��ʾ���÷�Ӧ�ġ�H 0 ����>��<��=����
CH3OH��g�� �������������䣬��300���500��ʱ�����ʵ���n��CH3OH�� �뷴Ӧʱ��t�ı仯������ͼ��ʾ���÷�Ӧ�ġ�H 0 ����>��<��=����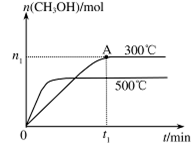
��3����Ҫ��״��IJ��ʣ��ɲ�ȡ�Ĵ�ʩ��____________������ĸ����
| A����������� |
| B�������¶� |
| C�������¶� |
| D��ʹ�ú��ʵĴ��� |
 CO+3H2��T��ʱ����1 L�ܱ�������Ͷ��1 mol CH4��1 mol H2O��g����5Сʱ���÷�Ӧ��ϵ�ﵽƽ��״̬����ʱCH4��ת����Ϊ50% ��������¶��µ�ƽ�ⳣ�� ���������С�������λ���֣���
CO+3H2��T��ʱ����1 L�ܱ�������Ͷ��1 mol CH4��1 mol H2O��g����5Сʱ���÷�Ӧ��ϵ�ﵽƽ��״̬����ʱCH4��ת����Ϊ50% ��������¶��µ�ƽ�ⳣ�� ���������С�������λ���֣���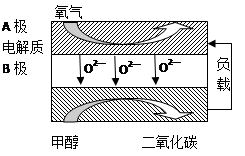
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com