��1��������ۻ������Ӽ�˷���
���Ӽ�
���Ӽ�
�������������������ۻ������Ӽ�˷���
���ۼ�
���ۼ�
����������������������Ӽ�˷���
���Ӽ�
���Ӽ�
�������������־�����۵��ɸߵ��͵�˳����
SiO2��KClO3��I2
SiO2��KClO3��I2
��
��2���������־��壺��CO
2����NaCl����Na����Si����CS
2�����ʯ�����ǵ��۵�ӵ͵��ߵ�˳��Ϊ
�٢ݢۢڢܢ�
�٢ݢۢڢܢ�
������ţ���
��3����H
2����NH
4��
2SO
4��SiC��CO
2��HF�У��ɼ��Լ��γɵķǼ��Է�����
CO2
CO2
���ɷǼ��Լ��γɵķǼ��Է�����
H2
H2
�����γɷ��Ӿ����������
H2��CO2��HF
H2��CO2��HF
����������ľ���Ļ�ѧʽ��
HF
HF
���������Ӿ������
��NH4��2SO4
��NH4��2SO4
������ԭ�Ӿ������
SiC
SiC
���������ʵ��۵��ɸߵ��͵�˳����
SiC����NH4��2SO4��HF��CO2��H2
SiC����NH4��2SO4��HF��CO2��H2
��
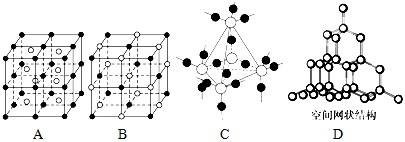
��4��A��B��C��DΪ���־��壬�������£�
A����̬ʱ�ܵ��磬����������
B��������CS
2��������ˮ
C����̬ʱ�����磬Һ̬ʱ�ܵ��磬������ˮ
D����̬��Һ̬ʱ�������磬�۵�Ϊ3 500��
���ƶ����ǵľ������ͣ�A��
��������
��������
��B��
���Ӿ���
���Ӿ���
��C��
���Ӿ���
���Ӿ���
��D��
ԭ�Ӿ���
ԭ�Ӿ���
��
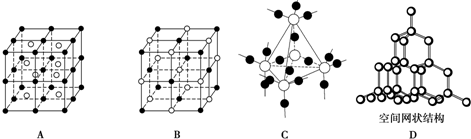
��5��ͼ��A��D����ѧ��ѧ�̿����ϳ����ļ��־���ṹģ�ͣ�����д��Ӧ���ʵ����ƣ�
A��
CsCl
CsCl
��B��
NaCl
NaCl
��C��
SiO2
SiO2
��D��
���ʯ
���ʯ
��



 ������ȫ�̼����ĩ���100��ϵ�д�
������ȫ�̼����ĩ���100��ϵ�д�