
ŌŚĻĀĮŠÖŠŃ§»Æѧ֊³£¼ūĪļÖŹµÄ×Ŗ»Æ¹ŲĻµĶ¼ÖŠ£¬·“Ó¦Ģõ¼ž¼°²æ·Ö·“Ó¦ĪļŗĶ²śĪļĪ“Č«²æ×¢Ć÷£¬ŅŃÖŖA”¢DĪŖČÕ³£Éś»īÖŠ³£¼ūµÄ½šŹōµ„ÖŹ£¬ĘäĖü¾łĪŖ»ÆŗĻĪļĒŅBĪŖŗģ×ŲÉ«¹ĢĢ唣Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ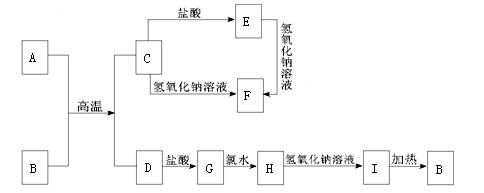
£Ø1£©Š“³öAµÄŌ×Ó½į¹¹Ź¾ŅāĶ¼ ”ų
£Ø2£©Š“³öĻĀĮŠĪļÖŹµÄ»ÆѧŹ½£ŗ B£ŗ ”ų E£ŗ ”ų
£Ø3£©Š“³öĻĀĮŠ·“Ó¦µÄ»Æѧ·½³ĢŹ½»ņĄė×Ó·½³ĢŹ½£ŗ
C”śFµÄ»Æѧ·½³ĢŹ½ ”ų
G”śHµÄĄė×Ó·½³ĢŹ½ ”ų
 æŚĖćŠÄĖćĖŁĖćÓ¦ÓĆĢāĻµĮŠ“š°ø
æŚĖćŠÄĖćĖŁĖćÓ¦ÓĆĢāĻµĮŠ“š°ø Ķ¬²½ĶŲÕ¹ŌĶĮĻµĮŠ“š°ø
Ķ¬²½ĶŲÕ¹ŌĶĮĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
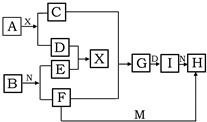 ŌŚĻĀĮŠÖŠŃ§»Æѧ֊³£¼ūĪļÖŹµÄ×Ŗ»Æ¹ŲĻµĶ¼ÖŠ£¬ŅŃÖŖ³£ĪĀĻĀAĪŖµ»ĘÉ«¹ĢĢå»ÆŗĻĪļ”¢BĪŖ½šŹōµ„ÖŹ£¬D”¢E”¢MŹĒ³£¼ūĘųĢåµ„ÖŹ£¬ĘäÖŠIĪŖŗģŗÖÉ«¹ĢĢ壬MĪŖ»ĘĀĢÉ«£¬NŹĒĪøĖįµÄÖ÷ŅŖ³É·Ö£¬¹¤ŅµÉĻÓĆEŗĶMĄ“ÖĘČ”N£¬ŹŌĶʶĻ£ŗ
ŌŚĻĀĮŠÖŠŃ§»Æѧ֊³£¼ūĪļÖŹµÄ×Ŗ»Æ¹ŲĻµĶ¼ÖŠ£¬ŅŃÖŖ³£ĪĀĻĀAĪŖµ»ĘÉ«¹ĢĢå»ÆŗĻĪļ”¢BĪŖ½šŹōµ„ÖŹ£¬D”¢E”¢MŹĒ³£¼ūĘųĢåµ„ÖŹ£¬ĘäÖŠIĪŖŗģŗÖÉ«¹ĢĢ壬MĪŖ»ĘĀĢÉ«£¬NŹĒĪøĖįµÄÖ÷ŅŖ³É·Ö£¬¹¤ŅµÉĻÓĆEŗĶMĄ“ÖĘČ”N£¬ŹŌĶʶĻ£ŗ²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ



²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ŌŚĻĀĮŠÖŠŃ§»Æѧ֊³£¼ūĪļÖŹµÄ×Ŗ»Æ¹ŲĻµĶ¼ÖŠ£¬·“Ó¦Ģõ¼ž¼°²æ·Ö·“Ó¦ĪļŗĶ²śĪļĪ“Č«²æ×¢Ć÷£¬ŅŃÖŖA”¢DĪŖČÕ³£Éś»īÖŠ³£¼ūµÄ½šŹōµ„ÖŹ£¬ĘäĖü¾łĪŖ»ÆŗĻĪļĒŅBĪŖŗģ×ŲÉ«¹ĢĢ唣Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©Š“³öAµÄŌ×Ó½į¹¹Ź¾ŅāĶ¼ ”ų
£Ø2£©Š“³öĻĀĮŠĪļÖŹµÄ»ÆѧŹ½£ŗ B£ŗ ”ų E£ŗ ”ų
£Ø3£©Š“³öĻĀĮŠ·“Ó¦µÄ»Æѧ·½³ĢŹ½»ņĄė×Ó·½³ĢŹ½£ŗ
C”śFµÄ»Æѧ·½³ĢŹ½ ”ų
G”śHµÄĄė×Ó·½³ĢŹ½ ”ų
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2011-2012Äź°²»ÕĪßŗžŅ»ÖŠø߶žĻĀŃ§ĘŚĘŚÖŠæ¼ŹŌĪÄæĘ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
£Ø5·Ö£©ŌŚĻĀĮŠÖŠŃ§»Æѧ֊³£¼ūĪļÖŹµÄ×Ŗ»Æ¹ŲĻµĶ¼ÖŠ£¬ŅŃÖŖ³£ĪĀĻĀAĪŖµ»ĘÉ«¹ĢĢ唢BĪŖ½šŹōµ„ÖŹ£¬D”¢E”¢MŹĒ³£¼ūĘųĢåµ„ÖŹ£¬ĘäÖŠMĪŖ»ĘĀĢÉ«£¬NŹĒĪøĖįÖ÷ŅŖ³É·Ö£¬¹¤ŅµÉĻÓĆEŗĶMĄ“ÖĘČ”N£¬ŹŌĶʶĻ£ŗ
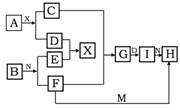
£Ø1£©Š“³öĻĀĮŠ»ÆѧŹ½ A I
£Ø2£©G×Ŗ»ÆĪŖIµÄĻÖĻóĪŖ
£Ø3£©F+M”śHĄė×Ó·½³ĢŹ½ĪŖ
£Ø4£©A+X”śC+DµÄ»Æѧ·½³ĢŹ½ĪŖ________________________
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com