
ŅŃÖŖ£ŗ¢Ł 2H2(g)£«O2(g)===2H2O(g)””¦¤H£½£483.6 kJ”¤mol£1””¢ŚH2(g)£«S(g)=== H2S(g)””¦¤H£½£20.1 kJ”¤mol£1”£
ĻĀĮŠÅŠ¶ĻÕżČ·µÄŹĒ(””””)
A£®1 molĒāĘųĶźČ«Č¼ÉÕÉś³ÉŅŗĢ¬Ė®ĪüŹÕČČĮæ241.8 kJ
B£®1 mol H2O(g)ŗĶ1 mol H2S(g)µÄÄÜĮæĻą²ī221.7 kJ
C£®ÓÉ¢Ł¢ŚÖŖ£¬Ė®µÄČČĪČ¶ØŠŌŠ”ÓŚĮņ»ÆĒā
D£®Čō·“Ó¦¢ŚÖŠøÄÓĆ¹ĢĢ¬Įņ£¬1 mol S(s)ĶźČ«·“Ó¦£¬·Å³öµÄČČĮæŠ”ÓŚ20.1 kJ

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
Ņ»¶ØÄÜŌŚĻĀĮŠČÜŅŗÖŠ“óĮæ¹²“ęµÄĄė×Ó×éŹĒ(””””)
¢Łŗ¬ÓŠ“óĮæAl3£«µÄČÜŅŗÖŠ£ŗNa£«”¢NH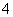 ”¢SO
”¢SO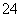 ”¢Cl£
”¢Cl£
¢Ś¼ÓČėAlÄܷųöH2µÄČÜŅŗÖŠ£ŗCl£”¢HCO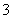 ”¢SO
ӢSO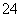 ӢNH
ӢNH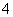
¢Ūŗ¬ÓŠ“óĮæFe3£«µÄČÜŅŗÖŠ£ŗNa£«”¢Mg2£«”¢NO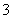 ”¢SCN£
”¢SCN£
¢ÜŌŚŗ¬ÓŠ“óĮæAlO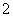 µÄČÜŅŗÖŠ£ŗNH
µÄČÜŅŗÖŠ£ŗNH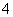 ”¢Na£«”¢Cl£”¢H£«
”¢Na£«”¢Cl£”¢H£«
¢ŻÓÉĖ®µēĄė³öµÄc(H£«)£½1”Į10£14mol”¤L£1µÄČÜŅŗÖŠ£ŗCa 2£«”¢K£«”¢Cl£”¢HCO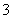
A£®¢Ł¢Ś B£®¢Ł¢Ū¢Ż
C£®¢Ł D£®¢Ł¢Ü¢Ż
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
¶ĢÖÜĘŚŌŖĖŲX”¢Y”¢Z”¢WµÄŌ×ÓŠņŹżŅĄ“ĪŌö“ó£¬ĒŅŌ×Ó×īĶā²ćµē×ÓŹżÖ®ŗĶĪŖ13”£XµÄŌ×Ó°ė¾¶±ČYµÄŠ”£¬XÓėWĶ¬Ö÷×壬ZŹĒµŲæĒÖŠŗ¬Įæ×īøßµÄŌŖĖŲ”£ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ(””””)
A£®Ō×Ó°ė¾¶µÄ“óŠ”Ė³Šņ£ŗr(Y)>r(Z)>r(W)
B£®ŌŖĖŲZ”¢WµÄ¼ņµ„Ąė×ӵĵē×Ó²ć½į¹¹ĻąĶ¬
C£®ŌŖĖŲYµÄ¼ņµ„ĘųĢ¬Ēā»ÆĪļµÄČČĪČ¶ØŠŌ±ČZµÄĒæ
D£®Ö»ŗ¬X”¢Y”¢ZČżÖÖŌŖĖŲµÄ»ÆŗĻĪļ£¬æÉÄÜŹĒĄė×Ó»ÆŗĻĪļ£¬Ņ²æÉÄÜŹĒ¹²¼Ū»ÆŗĻĪļ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ĻĀĮŠĖµ·Ø²»ÕżČ·µÄŹĒ(””””)
A£®·“Ó¦ŹĒ·ÅČČ»¹ŹĒĪüČȱŲŠėæ“Éś³ÉĪļŗĶ·“Ó¦ĪļĖł¾ßÓŠµÄ×ÜÄÜĮæµÄĻą¶Ō“óŠ”
B£®ĀĢÉ«Ö²Īļ½ųŠŠ¹āŗĻ×÷ÓĆŹ±£¬½«Ģ«ŃōÄÜ×Ŗ»ÆĪŖ»ÆѧÄÜ“¢“ęĘšĄ“
C£®ĪüČČ·“Ó¦ÖŠÓÉÓŚ·“Ó¦Īļ×ÜÄÜĮæŠ”ÓŚÉś³ÉĪļ×ÜÄÜĮ棬Ņņ¶ųĪŽĄūÓĆ¼ŪÖµ
D£®ĪļÖŹµÄ»ÆѧÄÜæÉŅŌŌŚŅ»¶ØĢõ¼žĻĀ×Ŗ»ÆĪŖČČÄÜ”¢µēÄÜĪŖČĖĄąĖłĄūÓĆ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
(1)2012ÄźĀ׶Ų°ĀŌĖ»į»š¾ę²ÉÓƱūĶéĪŖČ¼ĮĻ”£±ūĶéČČÖµ½Ļøߣ¬ĪŪČ¾½ĻŠ”£¬ŹĒŅ»ÖÖÓÅĮ¼µÄČ¼ĮĻ”£ŹŌ»Ų“šĻĀĮŠĪŹĢā£ŗ
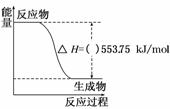
¢ŁČēĶ¼ŹĒŅ»¶ØĮæ±ūĶéĶźČ«Č¼ÉÕÉś³ÉCO2ŗĶ1 mol H2O(l)¹ż³ĢÖŠµÄÄÜĮæ±ä»ÆĶ¼£¬ĒėŌŚĶ¼ÖŠµÄĄØŗÅÄŚĢīČė”°£«”±»ņ”°£”±”£
¢ŚŠ“³ö±ķŹ¾±ūĶéČ¼ÉÕČȵÄČČ»Æѧ·½³ĢŹ½£ŗ___________________________________”£
¢Ū¶ž¼×ĆŃ(CH3OCH3)ŹĒŅ»ÖÖŠĀŠĶČ¼ĮĻ£¬Ó¦ÓĆĒ°¾°¹ćĄ«”£1 mol¶ž¼×ĆŃĶźČ«Č¼ÉÕÉś³ÉCO2ŗĶŅŗĢ¬Ė®·Å³ö1 455 kJČČĮ攣Čō1 mol±ūĶéŗĶ¶ž¼×ĆѵĻģŗĻĘųĢåĶźČ«Č¼ÉÕÉś³ÉCO2ŗĶŅŗĢ¬Ė®¹²·Å³ö1 645 kJČČĮ棬Ōņ»ģŗĻĘųĢåÖŠ£¬±ūĶéŗĶ¶ž¼×ĆѵÄĪļÖŹµÄĮæÖ®±ČĪŖ________”£
(2)øĒĖ¹¶ØĀÉČĻĪŖ£ŗ²»¹Ü»Æѧ¹ż³ĢŹĒŅ»²½Ķź³É»ņ·Ö¼ø²½Ķź³É£¬Õūøö¹ż³ĢµÄ×ÜČČŠ§Ó¦ĻąĶ¬”£ŹŌŌĖÓĆøĒĖ¹¶ØĀÉ»Ų“šĻĀĮŠĪŹĢā£ŗ
¢ŁŅŃÖŖ£ŗH2O(g)===H2O(l)””¦¤H1£½£Q1 kJ/mol C2H5OH(g)===C2H5OH(l)””¦¤H£½£Q2 kJ/mol
C2H5OH(g)£«3O2(g)===2CO2(g)£«3H2O(g)””¦¤H3£½£Q3 kJ/mol
ČōŹ¹46 gŅŗĢ¬ĪŽĖ®¾Ę¾«ĶźČ«Č¼ÉÕ£¬²¢»Öø“µ½ŹŅĪĀ£¬ŌņÕūøö¹ż³ĢÖŠ·Å³öµÄČČĮæĪŖ________kJ”£
¢ŚĢ¼(s)ŌŚŃõĘų¹©Ó¦²»³ä×揱£¬Éś³ÉCOĶ¬Ź±»¹²æ·ÖÉś³ÉCO2£¬Ņņ“ĖĪŽ·ØĶعżŹµŃéÖ±½Ó²āµĆ·“Ó¦£ŗC(s)£«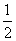 O2(g)===CO(g)µÄ¦¤H”£µ«æÉÉč¼ĘŹµŃ锢ĄūÓĆøĒĖ¹¶ØĀɼĘĖć³öøĆ·“Ó¦µÄ¦¤H£¬¼ĘĖ揱ŠčŅŖ²āµĆµÄŹµŃ鏿¾ŻÓŠ________”£
O2(g)===CO(g)µÄ¦¤H”£µ«æÉÉč¼ĘŹµŃ锢ĄūÓĆøĒĖ¹¶ØĀɼĘĖć³öøĆ·“Ó¦µÄ¦¤H£¬¼ĘĖ揱ŠčŅŖ²āµĆµÄŹµŃ鏿¾ŻÓŠ________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ÓŠ»śĪļ(CH3CH2)2CHCH3µÄĆū³ĘŹĒ
(”””” )
A£®3£¼×»łĪģĶé”””””””””””””” B£®2£¼×»łĪģĶé
C£®2£ŅŅ»łĪģĶé D£®3£ŅŅ»ł¶”Ķé
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
¶ŌijĖįŠŌČÜŅŗ£ØæÉÄÜŗ¬ÓŠBrØD£¬SO42”Ŗ£¬H2SO3£¬NH4+£©·Ö±š½ųŠŠČēĻĀŹµŃé£ŗ¢Ł¼ÓČČŹ±·Å³öµÄĘųĢåæÉŅŌŹ¹Ę·ŗģČÜŅŗĶŹÉ«£»¢Ś¼Ó¼īµ÷ÖĮ¼īŠŌŗ󣬼ÓČČŹ±·Å³öµÄĘųĢåæÉŅŌŹ¹ČóŹŖµÄŗģÉ«ŹÆČļŹŌÖ½±äĄ¶£»¢Ū¼ÓČėĀČĖ®Ź±£¬ČÜŅŗĀŌĻŌ»ĘÉ«£¬ŌŁ¼ÓČėBaCl2ČÜŅŗŹ±£¬²śÉśµÄ°×É«³Įµķ²»ČÜÓŚĻ”ĻõĖį”£¶ŌÓŚĻĀĮŠĪļÖŹ²»ÄÜČ·ČĻĘäŌŚČÜŅŗÖŠŹĒ·ń“ęŌŚµÄŹĒ £Ø £©
A£® BrØD B£® SO42”Ŗ C£® H2SO3 D£® NH4+
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
ŌŚ10L ŗćČŻĆܱÕČŻĘ÷ÖŠ³äČėX£Øg£©ŗĶY(g)£¬·¢Éś·“Ó¦X£Øg£©+Y£Øg£© M£Øg£©+N£Øg£©£¬ĖłµĆŹµŃ鏿¾ŻČēĻĀ±ķ£ŗ
M£Øg£©+N£Øg£©£¬ĖłµĆŹµŃ鏿¾ŻČēĻĀ±ķ£ŗ
| ŹµŃé | ĪĀ¶Č/”ę | ĘšŹ¼Ź±ĪļÖŹµÄĮæ/mol | Ę½ŗāŹ±ĪļÖŹµÄĮæ/mol | |
| n(X) | n(Y) | n(M) | ||
| ¢Ł | 700 | 0.40 | 0.10 | 0.090 |
| ¢Ś | 800 | 0.10 | 0.40 | 0.080 |
| ¢Ū | 800 | 0.20 | 0.30 | a |
| ¢Ü | 900 | 0.10 | 0.15 | b |
ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ£Ø £©
A£®ŹµŃé¢ŁÖŠ£¬Čō5minŹ±²āµĆn(M)=0.050mol£¬Ōņ0ÖĮ5minŹ±¼äÄŚ£¬ÓĆN±ķŹ¾µÄĘ½¾ł·“Ó¦ĖŁĀŹv£ØN£©=1.0”Į10-2mol/(L·min)
B£®ŹµŃé¢ŚÖŠ£¬øĆ·“Ó¦µÄĘ½ŗā³£ŹżK=2.0
C£®ŹµŃé¢ŪÖŠ£¬“ļµ½Ę½ŗāŹ±£¬XµÄ×Ŗ»ÆĀŹĪŖ60%
D£®ŹµŃé¢ÜÖŠ£¬“ļµ½Ę½ŗāŹ±£¬b>0.060
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com