
(ÿ��3�ֹ���10�֣����Ṥҵ�з�����Ϊ����������ɷ�ΪSiO2��Fe2O3��Al2O3��MgO��ij̽����ѧϰС���ͬѧ������·����������������н���Ԫ�ص���ȡʵ�顣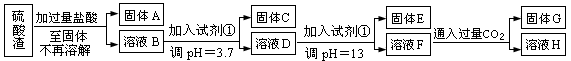
��֪��ҺpH=3.7ʱ��Fe3+�Ѿ�������ȫ��һˮ�ϰ����볣��Kb=1.8��10��5���䱥����Һ��c(OH��)ԼΪ1��10-3mol��L-1����ش�
��1��д��A������������Һ��Ӧ�Ļ�ѧ����ʽ������������������������������������
��2����������������ʹ���Լ��٣��Ʋ��Լ���Ӧ������������������������������������������ĸ��ţ�
| A���������� | B�������� | C����ˮ | D��ˮ |
 ��ѧʵ����ϵ�д�
��ѧʵ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ������ʡ������һ���¿���ѧ�Ծ��������棩 ���ͣ������
(ÿ��3�ֹ���10�֣����Ṥҵ�з�����Ϊ����������ɷ�ΪSiO2��Fe2O3��Al2O3��MgO��ij̽����ѧϰС���ͬѧ������·����������������н���Ԫ�ص���ȡʵ�顣
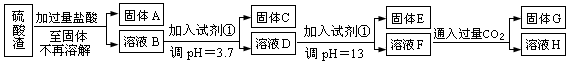
��֪��ҺpH=3.7ʱ��Fe3+�Ѿ�������ȫ��һˮ�ϰ����볣��Kb=1.8��10��5���䱥����Һ��c(OH��)ԼΪ1��10-3mol��L-1����ش�
��1��д��A������������Һ��Ӧ�Ļ�ѧ����ʽ�� ����������������������������������
��2����������������ʹ���Լ��٣��Ʋ��Լ���Ӧ���� ��������������������������������������ĸ��ţ�
A���������ơ����������� B�������������������� ���� C����ˮ������������ ��D��ˮ
��3����ҺD������E��������Ҫ������ҺpH=13�����pH��С�����ܵ��µĺ���������������� ������дһ�㣩
��4��H�����ʵĻ�ѧʽ�������������������������� ��
��5��������ҺF��c(Mg2+)��������������������25��ʱ��������þ��Ksp=5.6��10-12��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com