
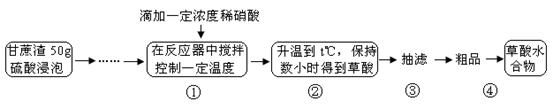
|
|
 �����������Ϣ�ش��������⣺
�����������Ϣ�ش��������⣺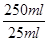 ��0.05000mol/L����������0.05000mol/L��126g/mol��6.3g�����Բ���Ĵ�����
��0.05000mol/L����������0.05000mol/L��126g/mol��6.3g�����Բ���Ĵ�����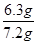 ��100%��87.5%��
��100%��87.5%��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����ij����ͨ����ˮ�У���ˮ��ɫ��ȥ��������һ������ϩ |
| B����п��ϡ���ᷴӦ�������ȴ�п�죬����Ϊ��п�ȴ�п��ԭ��ǿ |
| C��ֻ����ˮ�Ϳ��Խ��������Ȼ�̼���ƾ�����ϩ�������������������� |
| D�����������ˮ������Һ��������������Һˮԡ���ȿɿ������������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��Ũ���ᱣ������ɫ�����Լ�ƿ�� |
| B������������Һ��������Ƥ���IJ����Լ�ƿ�� |
| C���������Ʊ�����ú���� |
| D�����Ƶ���ˮͨ����������ɫ�����Լ�ƿ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����10mL��Ͳ��ȡ8.10mLϡ���� |
| B����pH�Ʋ��ij��Һ��pHΪ2.3 |
| C����pH��ֽ�ⶨ��Һ��pHʱ������������ˮ��ʪ��ֽ |
| D������25mL��ʽ�ζ�����ȡ20.00mL KMnO4��Һ |
�鿴�𰸺ͽ���>>
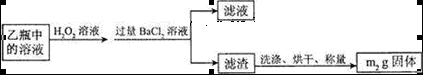
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
 2Fe2O3+8SO2
2Fe2O3+8SO2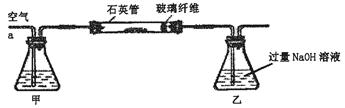
|

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����ƿ | B���ձ� | C���Թ� | D����Ͳ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����2g�����0.5g�����ȡ2.5gʳ�� |
| B���ñ�NaOH��Һ�ζ�δ֪Ũ�����ᣬ��ȥNaOH��Һ20.50mL |
| C���� 10mL ��Ͳ��ȡ8.25mL ���� |
| D����pH��ֽ���ij��ҺpHΪ 3.0 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com