
��ͭ�Ͻ�㷺���ں��չ�ҵ�����и�����л��������Ʊ�ͭ������Ʒ�Ĺ������£�

(ע��Al(OH)3��Cu(OH)2��ʼ�ֽ���¶ȷֱ�Ϊ450 ����80 ����)
(1)��⾫����ʱ��������ӦʽΪ________������A��ϡHNO3��Ӧ�������������ڿ�����Ѹ�ٱ�Ϊ����ɫ���������ɫ�Ļ�ѧ��Ӧ����ʽΪ________________��
(2)��������B�����Ϊ__________�������ɹ���B�Ĺ����У������NaOH�ļ���������NaOH�����������������ķ�Ӧ�����ӷ���ʽΪ____________��
(3)������չ�����һ����Ӧ�Ļ�ѧ����ʽ��____CuO��____Al2O3 ____CuAlO2��________����
____CuAlO2��________����
(4)����ͭ�Ͻ���ͭ����������Ϊ63.5%��������5.0 kg�����е�ͭ����ȫת��Ϊ________ mol CuAlO2��������Ҫ1.0 mol��L��1��Al2(SO4)3��Һ________ L��
(5)CuSO4��ҺҲ�������Ʊ������������������________�����ˡ�ϴ�Ӻ��
��(1)Ag����e��=Ag��2NO��O2=2NO2
(2)Al(OH)3��Cu(OH)2��Al(OH)3��OH��=AlO2����2H2O
(3)4��2��4��O2
(4)50��25
(5)����Ũ������ȴ�ᾧ
����������(1)��⾫��Agʱ������������������������������Ag���ĵ������Һ��Ϊ���Һ������������ӦʽΪAg����e��=Ag������Ag��ϡHNO3��Ӧ����NO��NO��O2��������NO2������ʽΪ2NO��O2=2NO2��
(2)NaOH��Һ��Al2(SO4)3��CuSO4��Ӧ����Al(OH)3������Cu(OH)2��������NaOH������Al(OH)3�����ܽ⣬���ӷ���ʽΪAl(OH)3��OH��=AlO2����2H2O��
(3)�����ڷ�Ӧǰ��Cu�Ļ��ϼ۽��ͣ�CuO��������������ʧ���ӵ�ֻ����O2������������O2�����õ�ʧ�����غ���ƽ��
(4)4Cu��4CuO��2Al2O3��2Al2(SO4)3��4CuAlO2
��2Cu��Al2(SO4)3��2CuAlO2
n(Cu)�� ��50 mol
��50 mol
���Կ���ȫת������50 mol CuAlO2��
������ҪAl2(SO4)3�����Ϊ ��25.0 L��
��25.0 L��
(5)���ڵ������нᾧˮ��Ϊ��ֹʧȥ�ᾧˮ��Ӧ��ȡ����Ũ������ȴ�ᾧ�ķ�����



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ����ר�� ��3��������Ҫ�Ļ�ѧ��Ӧ��ϰ���������棩 ���ͣ�ѡ����
��SO2ͨ������Fe2(SO4)3��Һ�У���ȫ��Ӧ���ټ���K2Cr2O7��Һ��������������ѧ��ӦΪ��SO2��2Fe3����2H2O=SO42����2Fe2����W����
Cr2O72����aFe2����bH���D��Cr3����Fe3����H2O������
�����й�˵����ȷ����(����)��
A����ԭ�ԣ�Cr3��>SO2
B������ʽ���У�a��6��b��7
C��Cr2O72���ܽ�Na2SO3������Na2SO4
D������ʽ����WΪOH��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ����ר�� ��13��ʵ�鷽�������������ϰ���������棩 ���ͣ�ʵ����
̼��������������Ӻ���(aNa2CO3��bH2O2)����Ư�ס�ɱ�����á�ʵ�����������������Ʊ������ʵ�ʵ�鲽�����£�
��1����ȡ����̼�����ܽ���һ����ˮ�������ƿ�У��ټ��������ȶ���(MgCl2��Na2SiO3)��������ȡ�
��2����������30%��H2O2��Һ�ڽ���״̬�µ�����ƿ�У���15 �����ҷ�Ӧ1 h��
��3������Ӧ��Ϻ��ټ���������ˮ�Ҵ������á��ᾧ�����ˡ�����ò�Ʒ��
(1)��1���У��ȶ�����ˮ��Ӧ�������ֳ�����������仯ѧ����ʽΪ___________________________________________________________��
(2)��2���У���Ӧ����Ϊ15 �����ҿɲ�ȡ�Ĵ�ʩ��_____________________
___________________________________________________��
(3)��3���У���ˮ�Ҵ���������____________________________________��
(4)H2O2�ĺ����ɺ�����Ʒ�����ӡ��ֳ�ȡm g(Լ0.5 g)��Ʒ��������й�������ˮ���Ƴ�250 mL��Һ��ȡ25.0 mL����ƿ�У�����ϡ�����ữ������c mol��L��1 KMnO4��Һ�ζ����յ㡣
������250 mL��Һ����IJ����������ձ�������������Ͳ________��________��
���ζ��յ�۲쵽��������______________________________________��
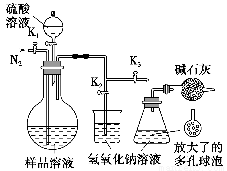
(5)��ģ�������ⶨ��Ʒ��̼���Ƶĺ�����װ������ͼ��ʾ(���Ⱥ̶�װ������ȥ)��ʵ�鲽�����£�
����1������ͼ��ʾ��װ���������װ�������ԡ�
����2��ȷ��ȡ(4)��������Һ50 mL����ƿ�С�
����3��ȷ��ȡ40.00 mLԼ0.2 mol��L��1 NaOH��Һ���ݣ��ֱ�ע���ձ�����ƿ�С�
����4������K1��K2���رջ���K3����ͨ�뵪��һ��ʱ��ر�K1��K2����K3������Һ©������ƿ�м���10 mL 3 mol��L��1������Һ��
����5����������ƿ�е�Һ����ڣ���������һ��ʱ�䡣
����6����K1�ٻ���ͨ�뵪��һ��ʱ�䡣
����7������ƿ�м������ָʾ������c1 mol��L��1 H2SO4����Һ�ζ����յ㣬����H2SO4����ҺV1 mL��
����8����ʵ�鲽��1��7�ظ����Ρ�
������3�У�ȷ��ȡ40.00 mL NaOH��Һ����Ҫʹ�õ�������________��
������1��7�У�ȷ�����ɵĶ�����̼������������Һ��ȫ���յ�ʵ�鲽����________(�����)��
��Ϊ�����Ʒ��̼���Ƶĺ��������貹���ʵ����______________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ����ר�� ��11���л���ѧ������ϰ���������棩 ���ͣ������
���ƽ�����ھ��������������ĺϳ�·��������

(1)д��C13H9NO4S�����к��������ŵ����ƣ�________��
(2)A������������Է���������54��д��A�Ľṹ��ʽ��____________��
(3)��Ӧ������������ȡ����Ӧ����________(�����)��
д����Ӧ���Ļ�ѧ����ʽ��__________________________________________��
(4)��������Ʒ�Ӧ��������Ŀ����___________________________________��
(5)����C��ͬ���칹���ж��֣����мȺ����ǻ����ֺ���ȩ����ͬ���칹����________�֡�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ����ר�� ��11���л���ѧ������ϰ���������棩 ���ͣ�ѡ����
���ܼ�DCHP�����ڱ����������뻷������Ӧ�Ƶá�

����˵����ȷ����(����)��
A���ڱ����������Ķ��ȴ�����3��
B�����������������е�ԭ�ӿ��ܹ�ƽ��
C��DCHP�ܷ����ӳɡ�ȡ������ȥ��Ӧ
D��1 mol DCHP�����뺬2 mol NaOH����Һ��Ӧ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ����ר�� ��10������Ԫ�ؼ��仯������ϰ���������棩 ���ͣ�ѡ����
���з�Ӧ�����ӷ���ʽ��ȷ����(����)��
A��ϡ����������Ӧ��2Fe��6H��=2Fe3����3H2��
B��2 mol��L��1��AlCl3��Һ��7 mol��L��1��NaOH��Һ��������Ȼ�ϣ�2Al3����7OH��=Al(OH)3����AlO2����2H2O
C��Ba(OH)2��Һ�м���������NaHSO4��Һ��Ba2����2OH����2H����SO42��=BaSO4����2H2O
D��NaHCO3��ˮ��������
HCO3����H2O CO32����H3O��
CO32����H3O��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ���ѵ��ר��12���ʽṹ�����ʣ�ѡ�ޣ���ϰ���������棩 ���ͣ������
ͼʾ����ͼ���dz��õĿ�ѧ�о�������
��1����ѧ��ͨ��X�����Ʋ���мȺ�����λ���ֺ����������ṹʾ��ͼ�ɼ�ʾ���£�������λ����������������߱�ʾ��

��д����̬Cuԭ�ӵĺ�������Ų�ʽ____________________��
��д������������ˮ��ͭ���ӵĽṹ��ʽ�����뽫��λ����ʾ������________��
��2����ͼ���о�����Ԫ���⻯��ķе�仯���ɵ�ͼ������c���Ա������________��Ԫ���⻯��ķе�仯���ɡ���λͬѧ��ij����Ԫ���⻯��ķе�ı仯���ƻ���������������������a������b������Ϊ��ȷ����________������a������b������������______________________________________________________________��

��3����������Ԫ�ص���̬�����Ի�̬ԭ��ʧȥ�����һ������ת��Ϊ��̬��̬������������������������һ�����ܣ���ΪE������ͼ��ʾ��

��ͬ�����ڣ�����ԭ������������Eֵ�仯����������________��
������ͼ���ṩ����Ϣ�����ƶ�E��________E��������������������������������ͬ����
�����ݵ�һ�����ܵĺ����Ԫ�������ɣ����ƶ�Eþ________E����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ���ѵ�� ר��9�ǽ���Ԫ�ؼ��仯������ϰ���������棩 ���ͣ������
�������շ�������������к��̳�����������������Ȼ�����к����ʣ�ij�����������£�

��1����ʯϴ���������ö���դ��������ô������������С���������ö����������ˮ�죬���������������룬Ŀ����_____________________________________________��
��2����NO2��NO���������ʵ���֮��1��1���ͨ��ʯ��ˮ�У�����������ԭ��Ӧ����һ�����κ�ˮ����д���÷�Ӧ�Ļ�ѧ����ʽ��_________________________________________��
��֪�����ε�ˮ��Һ�ʼ��ԣ������ӷ���ʽ��ʾ��__________________________________��
��3����Һ�ڳ�����������������������Ҫ��________���ڻ����������м���NaOH�����������___________________________________________��
��4��������ⲿ�Ų�������ǰ���������̳��ĺ������±���
�Ŀ | ����� | ����� | |
����ǰ | ������ | GWKB3��2000 | |
�̳���mg��m��3�� | 13 929.0 | 61.0 | 80 |
�������ü�����е�����ʹ�õIJⶨ������__________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ��߿���ѧ���ָ�ϰ���ѵ�� ר��4���ʽṹԪ����������ϰ���������棩 ���ͣ�ѡ����
W��X��Y��Z���ֶ�����Ԫ����Ԫ�����ڱ��е����λ����ͼ��ʾ��W����̬�⻯���������ۺ����ᷴӦ�������ӻ��������˵������ȷ���ǣ���������
W | X |
|
| Y | Z |
A��X��Y��Z������⻯���ȶ�����������Y
B��ZԪ���������Ӧˮ���������һ��ǿ��Y
C��XԪ���γɵĵ��������ӻ�ԭ������Y
D��YZ2�����и�ԭ�ӵ�����������8e���ȶ��ṹ
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com