
��ͼ�ǹ���ҩ�ﻪ��Ƭ(���ص�Ƭ)��ʹ��˵���飬����Ϊ˵����IJ������ݣ�
| ����Ƭ(���ص�Ƭ)ʹ��˵���� ��Ʒ������ɡ� Ʒ�������ص�Ƭ ��ɣ�����Ƭ Ӣ������Cydiodine Tablets ����Ƭ(���ص�Ƭ)����Ҫ���Գɷ��Ƿ��ӵ⣬����1.5 mg/Ƭ���������÷��ӷ�ɢ�����Ƴɷ���̬���ص⣬�������������Ե� �����ء� �ڹ⡢�ܱա����������� ����Ч�ڡ� ���� |

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
������������ȷ����(����)
| A����������ˮ��������ϩ�ͱ� |
| B�������ý������������Ҵ������� |
| C�����������Ƶ�������ͭ�����������Ǻ͵��� |
| D�����������յķ�����������˿���Ϻʹ����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��һƿ�������Һ�����п����� ��K����Na����Mg2����Ba2����Al3����Cl����I����
��K����Na����Mg2����Ba2����Al3����Cl����I���� ��
�� ��S2����
��S2���� ��
�� ��
�� ��ȡ����Һ��������ʵ�飺
��ȡ����Һ��������ʵ�飺
(1)ȡpH��ֽ���飬��Һ��ǿ���ԣ������ų� ���ӵĴ��ڣ�
(2)ȡ��������Һ����������CCl4������������ˮ������CCl4�����ɫ�������ų� ���ӵĴ��ڣ�
(3)��ȡ��������Һ��μ���NaOH��Һ��ʹ��Һ��������Ϊ���ԣ��ڵμӹ����к͵μ���Ϻ���Һ���������������ֿ����ų� ���ӵĴ��ڣ�
(4)ȡ����������������Һ�μ�Na2CO3��Һ���а�ɫ�������ɣ�֤�� ���Ӵ��ڣ��ֿ��ų� ���ӵĴ��ڣ�
(5)��(3)�õ��ļ�����Һ���ȣ�������ų�����������ʹʪ��ĺ�ɫʯ����ֽ��������������ʵ����ʵȷ��������Һ�п϶����ڵ������� ��������ȷ���Ƿ���ڵ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ҵ�ϳ�����ұ��п�����е�п������ZnO��FeO��Fe2O3��CuO��Al2O3�����ʣ�������������Zn(NO3)2��6H2O���壬�乤������Ϊ��
�й��������↑ʼ�����ͳ�����ȫ��pH���±���
| �������� | Al(OH)3 | Fe(OH)3 | Fe(OH)2 | Cu(OH)2 | Zn(OH)2 |
| ��ʼ������pH | 3��3 | 1��5 | 6��5 | 4��2 | 5��4 |
| ������ȫ��pH | 5��2 | 3��7 | 9��7 | 6��7 | 8��0 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ҵ�ϳ��ø�������Ч�ɷ�ΪFeO��Cr2O3����Ҫ����ΪSiO2��Al2O3��Ϊԭ�������ظ���أ�K2Cr2O7����ʵ����ģ�ҵ���ø��������ظ���ص���Ҫ������������ͼ���漰����Ҫ��Ӧ�ǣ�6FeO��Cr2O3��24NaOH��7KClO3=12Na2CrO4��3Fe2O3��7KCl��12H2O���Իش��������⣺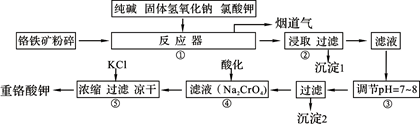
��1��������Һ���������ӵļ��鷽���� ��
��2������۱�����������Ϊ�������ӷ��ţ� ��
��3���ڷ�Ӧ�����У����������봿�Ӧ�Ļ�ѧ����ʽΪ�� ��
��4���̵����е�CO2����H2�ϳɼ״���CH3OH��H2��ȼ���ȷֱ�Ϊ����H=��725.5 kJ/mol����H=��285.8 kJ/mol��д����ҵ����CO2��H2�ϳ�CH3OH���Ȼ�ѧ����ʽ�� ��
��5��2011�����������ĸ���Ⱦ�¼���˵��������������ˮ���������ŷŶ��������滷���м����Σ������ⷨ�Ǵ�������Ⱦ��һ�ַ�������������������ʯī��������⺬Cr2O72-�����Է�ˮ��һ��ʱ������Fe(OH)3��Cr(OH)3������
��д����ⷨ������ˮ���ܷ�Ӧ�����ӷ���ʽ ��
����֪Cr(OH)3��Ksp=6.3��10�C31�����ر�ˮ�����������ֵ��0.1 mg/L��Ҫʹ��Һ��c(Cr3+)�������ϵر�ˮ��ֵ���������Һ��c(OH-)�� mol/L��ֻд�������ʽ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������ƿ������ڸ��Ƶر�ˮ�ʣ��������ؽ������ӷ�ˮ�������ೱ��Ҳ������Ӧ�������ȡ���ҵ�������������Ƶ���Ҫ�������£�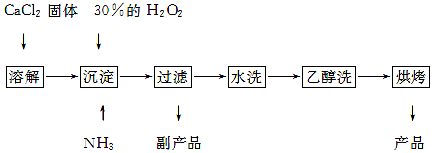
��֪CaO2��8H2O�ʰ�ɫ������ˮ��������350 �����ҿ�ʼ�ֽ�ų�������
��1��������������ȡCaO2��8H2O�Ļ�ѧ����ʽ�� ��
��2�����顰ˮϴ���Ƿ�ϸ�ķ����� ��
��3������ʱ���ñ�ˮ�����¶���0 �����ң������ԭ����(д������)��
�� ;
�� ��
��4���ⶨ��Ʒ��CaO2�ĺ�����ʵ�鲽�裺
��һ����ȷ��ȡa g��Ʒ��������ƿ�У�������������ˮ������b g KI���壬�ٵ�������2 mol��L��1��H2SO4��Һ����ַ�Ӧ��
�ڶ�������������ƿ�м��뼸�ε�����Һ��
����������μ���Ũ��Ϊc mol��L��1��Na2S2O3��Һ����Ӧ��ȫ������Na2S2O3��ҺV mL��
����֪��I2��2S2O32-��2I����S4O62-��
��CaO2����������Ϊ (����ĸ��ʾ)��
��ijͬѧ��һ���͵ڶ����IJ������ܹ淶������������̫����������õ�CaO2�������������� (�����Ӱ�족����ƫ�͡���ƫ�ߡ�)��ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��1�������ӹ����θ�д����������ʽ��KAlSi3O8�� ��
��2����һ����ɫ��ĩ������K2SO4��NaHCO3��BaCl2��FeCl3��KCl���������е�ij������ɣ��ֽ�������ʵ�飺
�ٽ���ɫ��ĩ��ˮ�ܽ⣬����ɫ��Һ��
����������õ���ɫ��Һ�м���NaOH��Һ���۲쵽�а�ɫ����A���ɣ����˺�����Һ�еμ������ữ��AgNO3��Һ���ֵõ���ɫ����B��
�������������жϣ�
��A�Ļ�ѧʽ�� B�Ļ�ѧʽ�� ��
��ԭ��ɫ��ĩ��һ������ �����ܺ��� ��
�Կ��ܺ��е����ʣ���ͨ�� ����ʵ�����ƣ���һ�����顣������������У���պȡ����Һ�������ھƾ��ƻ��������գ�������ɫ�ܲ����۲죻����ϡ����ϴ����˿��������ȷ�IJ���˳��Ϊ ��
A���٢ڢۢ� B���ܢ٢ڢ� C���ܢڢ٢ڢۢ� D���٢ۢڢ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ʵ����ģ�����ij�Ͼɺ�������(��Ҫ�ɷ�ΪNiO������Fe2O3��CaO��CuO��BaO��)����Ni2O3���乤���������£�
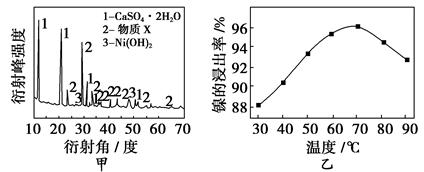
(1)����ͼ����ʾ��X��������ͼ�ף���֪����������������Ҫ�ɷ֣����С�����X��Ϊ________��ͼ�ұ�ʾ���Ľ��������¶ȵĹ�ϵ���������¶ȸ���70 ��ʱ�����Ľ����ʽ��ͣ���������Ni(OH)2����������ԭ����
________________________________________________________________��
(2)���������и���Ʒ�Ļ�ѧʽΪ______________��
(3)��֪�й��������↑ʼ�����ͳ�����ȫ��pH���±���
| �������� | Fe(OH)3 | Fe(OH)2 | Ni(OH)2 |
| ��ʼ������pH | 1.5 | 6.5 | 7.7 |
| ������ȫ��pH | 3.7 | 9.7 | 9.2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���ؾ�ʯ����Ҫ�ɷ�ΪBaSO4��Ϊ��Ҫԭ���Ʊ����·ۣ�ZnS��BaSO4�Ļ����Ĺ����������£�
��1������ʱ����̿���������Ŀ���ǣ���____________________________����____________________________��
��2�����չ����з����ķ�Ӧ֮һ��4CO��g����BaSO4��s�� BaS��s����4CO2��g�����÷�Ӧ��ƽ�ⳣ���ı���ʽΪ__________________________________________________��
BaS��s����4CO2��g�����÷�Ӧ��ƽ�ⳣ���ı���ʽΪ__________________________________________________��
��Ӧ���з�����Ӧ�Ļ�ѧ����ʽΪ____________________________________��
��4���ù�ҵ������п��������FeO��Fe2O3���ʣ���������ȡ����п��Һʱ�����Ⱥ����H2O2��Һ��̼��п���ƣ�����H2O2��Һʱ������Ӧ�����ӷ���ʽΪ
________________________________������̼��п��Ŀ���ǽ���Һ�е�Fe3��ת��Ϊ���ɫ������д���÷�Ӧ�Ļ�ѧ����ʽ��__________________________________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com