
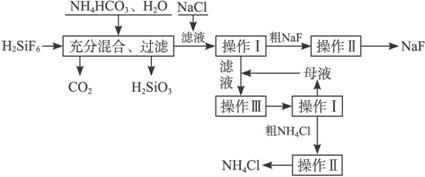
�й�������ˮ���ܽ�ȷֱ�Ϊ��
�Ȼ�泥�
��ش��������⣺
��1�����������з���������ѧ��Ӧ����һ����Ӧ�Ļ�ѧ����ʽΪ________________________���ڶ�����Ӧ�ܽ�������Ϊ_____________________________________________________��
��2����������Ҫ�õ��IJ����������������⣬����______________________________��
��3���������������___________________________________________________��
��4��������ľ��������________________________________________________��
��5��������NH4HCO3�����������ԭ����_____________________________________��
��1��H2SiF6+6NH4HCO3+H2O====6NH
��2���ձ���©��
��3����ȥNaF��NH4Cl���������
��4�����������ܼ������������������ȴ
��5�����뱣֤H2SiF6��ȫ����Ӧ����ֹ�ڽ��з�Ӧ��ʱH2SiF6����NaCl��Ӧ������Na2SiF6����������NaF��Ӱ��NaF����
������������Ҫ�����˻�ѧ�빤ҵ���������֪ʶ����һ����Ӧ�ķ���ʽ��������֪��Ӧ������������֪�����������ƽ���÷�Ӧ����ʽ���ڶ�����Ӧ�ǽ���һ�����ɵ�NH



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
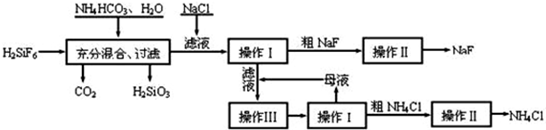
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| �¶� | 10�� | 20�� | 30�� | �ܽ�ȣ�20��NaF-4g��0��NH4F-100g�� ����Na2SiF6-����ˮ |
| NH4Cl�ܽ�� | 33.3g | 37.2g | 41.4g |
�鿴�𰸺ͽ���>>
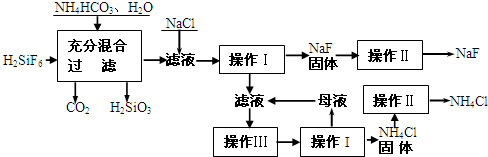
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(15��)��������һ����Ҫ�ķ��Σ���Ҫ����ũҵɱ������ɱ�����ľ�ķ�������������������ȡ�ʵ���ҿ�ͨ����ͼ��ʾ�������Է����ᣨH2SiF6��������Ϊԭ����ȡ�����ƣ����õ�����Ʒ�Ȼ�泥�
�й�������ˮ���ܽ��(g/100gH2O)���£�
| �¶� | 10�� | 20�� | 30�� | �ܽ�ȣ�20��NaF��4 0��NH4F��100�� ����Na2SiF6����ˮ |
| NH4Cl�ܽ�� | 33.3 | 37.2 | 41.4 |
��ش��������⣺
��1����������Ҫ�õ��IJ��������� ��
��2�����������з���������Ӧ����ѧ����ʽΪ��
��
��3������II�������� ��
������ľ��������
��4��������NH4HCO3�����������ԭ���� ������
_______________________________________________________________________
�鿴�𰸺ͽ���>>
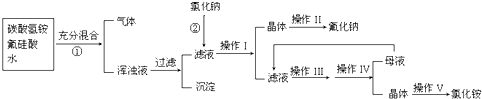
��Ŀ�����л�ѧ ��Դ��2012��ӱ�ʡ������ѧ���������ο��Ի�ѧ�Ծ� ���ͣ������
(15��)��������һ����Ҫ�ķ��Σ���Ҫ����ũҵɱ������ɱ�����ľ�ķ�������������������ȡ�ʵ���ҿ�ͨ����ͼ��ʾ�������Է����ᣨH2SiF6��������Ϊԭ����ȡ�����ƣ����õ�����Ʒ�Ȼ�泥�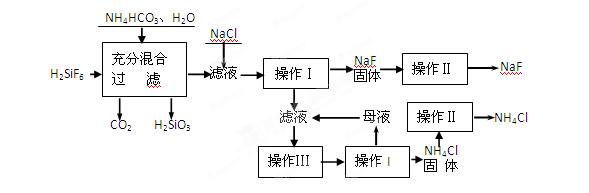
�й�������ˮ���ܽ��(g/100gH2O)���£�
| �¶� | 10�� | 20�� | 30�� | �ܽ�ȣ�20��NaF��4 0��NH4F��100�� ����Na2SiF6����ˮ |
| NH4Cl�ܽ�� | 33 .3 .3 | 37.2 | 41.4 |

�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com