
����X��Y��Z��M����Ԫ�ؾ�Ϊ������Ԫ�ء��й���Ϣ���±���
��1��XԪ�������ڱ��е�_________���ڡ���_________�壻
��2��YԪ�ص�Ԫ�ط���Ϊ_________��1�����ʷ����к�_________�����õ��Ӷԣ�
��3��Z����CO2������ȼ�գ��仯ѧ��Ӧ����ʽΪ_________ ��
��4��������X2M3�У���ԭ�ӣ������ӣ��������ﵽ8�����ȶ��ṹ����X2M3�ĵ���ʽΪ _________���û������ˮ��Һ�ڿ����о��ú�ᱻ���������ʣ�����һ�ֵ��ʣ�������Һ��ǿ���ԡ��û�ѧ����ʽ��ʾ����仯����_________��
��1������IA����2��N��3;��3��2Mg+CO2 2MgO+C����4��2Na2S3+O2+2H2O=4NaOH+6S
2MgO+C����4��2Na2S3+O2+2H2O=4NaOH+6S
���������������1�����ں�XԪ�ص����ʵ���ɫ��ӦΪ��ɫ��˵������NaԪ�ء�11��Ԫ��Na�ĺ�������Ų���2��8��1.��������Ԫ�����ڱ���λ�ڵ������ڵ�IA����2����������Ҫ�ɷ���N2��O2���е�N2�͡�����YԪ����NԪ�ء���ÿ��N2������Nԭ��ͨ�����Թ��õ��ӶԽ�ϣ��γ���3�����ۼ�����3��������Ԫ��Z��ԭ��ʧȥ2�����ӵõ�8���ӽṹ�����ӣ���ZΪMgԪ�ء�Mg��CO2������ȼ�յĻ�ѧ��Ӧ����ʽΪ2Mg+CO2 2MgO+C����4������MԪ�ص�ԭ��M��ĵ�������K���3����������Na��ͬһ���ڡ���M�ĺ�������Ų�Ϊ2��8��6������M��16��Ԫ��S����Na2S3�и����Ӷ��ﵽ���������ﵽ8�����ȶ��ṹ�������ʽΪ ���û������ˮ��Һ�ڿ����о��ú�ᱻ���������ʣ�����һ�ֵ��ʣ�������Һ��ǿ���ԡ��û�ѧ����ʽ��ʾ����仯����2Na2S3+O2+2H2O=4NaOH+6S��
2MgO+C����4������MԪ�ص�ԭ��M��ĵ�������K���3����������Na��ͬһ���ڡ���M�ĺ�������Ų�Ϊ2��8��6������M��16��Ԫ��S����Na2S3�и����Ӷ��ﵽ���������ﵽ8�����ȶ��ṹ�������ʽΪ ���û������ˮ��Һ�ڿ����о��ú�ᱻ���������ʣ�����һ�ֵ��ʣ�������Һ��ǿ���ԡ��û�ѧ����ʽ��ʾ����仯����2Na2S3+O2+2H2O=4NaOH+6S��
���㣺����Ԫ�����ڱ���Ԫ�������ɵ�֪ʶ����Ҫ����Ԫ�ص��ƶϡ�λ�á��ṹ�����ʡ�����ʽ����д����ɫ��Ӧ�ȡ�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������߷ֱ��ʾԪ�ص�ij��������˵�����Ĺ�ϵ��ZΪ�˵������YΪԪ�ص��й����ʣ���
��1����������Ԫ���й�������������߱��������Ӧ�Ŀո��У�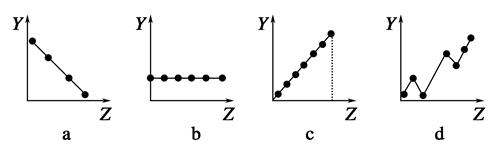
�ٵڢ�A��Ԫ�صļ۵�����________��
�ڵ�������Ԫ�ص�����ϼ�________��
��F����Na����Mg2����Al3�������Ӱ뾶________��
��2��Ԫ��X��Y��Z��M��N��Ϊ����������Ԫ�أ���ԭ����������������֪Yԭ������������������������֮��Ϊ3��4��MԪ��ԭ�ӵ���������������Ӳ���֮��Ϊ4��3��N����Z����X���İ뾶��С��������XN������Ϊ���塣�ݴ˻ش�
��XΪ________�����ƣ���YΪ________��Ԫ�ط��ţ���Zԭ�ӽṹʾ��ͼΪ________��
��N������������ˮ����Ļ�ѧʽΪ__________��
�۹�ҵ����ȡ����M�Ļ�ѧ����ʽΪ______________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
X��Y��Z��M��N��QΪԪ�����ڱ�ǰ�����ڵ�����Ԫ�ء����У�Xԭ�Ӻ����M����ֻ�����ԳɶԵ��ӣ�Yԭ�Ӻ����L���������K���������Z�ǵؿ��ں�����������������ߵ�Ԫ�أ�M���ڲ��������������������9����N��ԭ��������MС1, Q��Ԫ�����ڱ��ĸ�Ԫ���е縺�������ش��������⣺
��1��XԪ�������ڱ��е�λ���� ������ ��Ԫ�أ�������Χ���ӵĵ����Ų�ͼΪ ��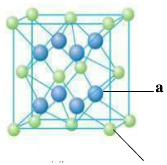
��2��XZ2���ӵ�����ṹ�� ��YZ2������Y���ӻ��������Ϊ ����ͬ������������ˮ�е��ܽ�Ƚϴ���� ��д����ʽ���������� ��
��3������Ԫ��N���ε���ɫ��ӦΪ ɫ����������ζ����Է�����ɫ��Ӧ����ԭ���� ��
��4��Ԫ��M��Ԫ��Q�γɾ���ṹ��ͼ��ʾ�����侧���߳�Ϊa pm����aλ����bλ��֮��ľ���Ϊ_______pm��ֻҪ������ʽ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������Ԫ�����ڱ���һ���֣�����Ԫ�آ٣����ڱ��е�λ�ã����û�ѧ����ش��������⣺
| �� ���� | IA | | 0 | |||||
| 1 | �� | ��A | ��A | ��A | ��A | ��A | ��A | |
| 2 | | | | �� | �� | �� | | |
| 3 | �� | | �� | �� | | | �� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�ڻ�ҩ���й��Ŵ��Ĵ���֮һ�����ı�ը��ӦΪ��
2KNO3+3C+S A+N2��+3CO2��������ƽ����
A+N2��+3CO2��������ƽ����
��1���������г��˵����������̼�⣬����һ������A����A�ĵ���ʽΪ ��A����________�����
��2���ڻ�ҩ��λ��Ԫ�����ڱ������ڵ�Ԫ���� �֡�����һ��Ԫ�صĵ��ʿ�������ҩ���������������Ư��֯���������������Ԫ�������ڱ��е�λ���� ��
��3���ڻ�ҩ������Ԫ�ص�ԭ�Ӱ뾶�Ӵ�С��˳���� ����Ԫ�ط��ű�ʾ����
��4��������ʵ��˵���ڻ�ҩ��̼������Ԫ�طǽ��������ǿ������ ��
A��ͬ����ͬŨ����Һ��pH��Na2CO3��Na2SO4
B�����ԣ�H2SO3��H2CO3
C��CS2��̼Ԫ��Ϊ+4�ۣ���Ԫ��Ϊ-2��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ԭ���������������A��B��C��D��E����Ԫ�أ�A�Ƕ������������������������ķǽ���Ԫ�أ�BԪ�ص�ԭ�ӼȲ���ʧȥҲ���õ����ӣ����̬ԭ��ÿ�ֹ���е�������ͬ��CԪ�صļ۵��ӹ���Ϊnsnnpn��1��D����������������Ӳ���֮��Ϊ3��1��E�ǵؿ��к������������Ľ���Ԫ�أ���Ͻ���;��㣬�������
(1)B��D�γɵķǼ��Է���������ԭ�ӵŶԵ�������________������ԭ�ӵ��ӻ��������Ϊ________��
(2)A�ֱ���B��C��D���γɵ�����Ϊ10�Ļ�������ǵķе��ɸߵ��͵�˳����____________(д����ʽ)�����ǵ��ȶ���������ǿ��˳����_______________________________________________________(д����ʽ)��
(3)����ABC��BA2D�Ŀռ乹�ͷֱ���________��________��
(4)B��C��D����Ԫ�صĵ縺���ɴ�С��˳����________(��Ԫ�ط��ű�ʾ)����һ�������ɴ�С��˳����________(��Ԫ�ط��ű�ʾ)��
(5)C�ĵ��ʷ����д���________���м���________���Ҽ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
X��Y��Z��Ϊ������Ԫ�أ�ԭ��������������X�ĵ���Ϊ�ܶ���С�����壬Yԭ������������������������������Z��Xԭ��������������ͬ���ش��������⣺
(1)X��Y��Z��Ԫ�ط��ŷֱ�Ϊ______��______��______��
(2)������Ԫ����ɵĻ������У��Ⱥ��й��ۼ��ֺ������Ӽ�����________��________��
(3)X��Y��ɵĻ������У��Ⱥ��м��Թ��ۼ��ֺ��зǼ��Թ��ۼ�����________��
�˻������������������������ط�Ӧ�����ӷ���ʽΪ__________________________��
�˻����ﻹ�ɽ����Թ�ҵ��ˮ�е�CN������Ϊ̼���κͰ�����Ӧ�����ӷ���ʽΪ______________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
A��B��C��D��ԭ��������С��20������Ԫ�ء�A��Bͬ���壬�����γ�BA3�ͷ��ӡ�B��C��D���γɵļ����ӵ��Ӳ�ṹ��ͬ����B��C��D���Ӱ뾶���μ�С���ݴ˻ش��������⣺
(1)DԪ����Ԫ�����ڱ��е�λ���� ��
(2)B��D���γɵĻ��������ʽΪ ��
(3)��C���ʵ�ˮ��Һ�μӵ�B��D���γɻ������ˮ��Һ�У���������ɫ�������䷴Ӧ�Ļ�ѧ����ʽΪ ��
(4)ʵ�������ȥBA2���壬���������Լ��е� (����ĸ)��
A.���Ը��������Һ B.Ũ����
C.NaOH��Һ D.Ʒ����Һ
(5)��A��B��D����Ԫ������ɵ�ij����ˮ��Һ�Լ��ԣ���ԭ���� (�����ӷ���ʽ��ʾ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�±���Ԫ�����ڱ���һ���֣�����������ĸ�ֱ����ijһ��ѧԪ�ء��û�ѧ����ش��������⡣
| a | | | |||||||||||||||
| | | | | | | | | | | | | b | c | d | | | |
| e | | f | g | | h | i | | ||||||||||
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com