
ij�ռ���Ʒ�����������������õ����ʣ�Ϊ�˲ⶨ�䴿�ȣ�������������������
��һ��������500 ml �ռ���Ʒ��Һ��
��1���������ƿ�Ƿ�©Һ�ķ����ǣ���ƿ�ڼ���һ����ˮ������ƿ����
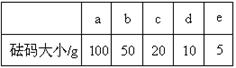
��2��������Ϊ13��1 g�Ŀ��ձ�����Ҫ��ȡ�ռ���Ʒ20 g �������������ƽ�ϳ�ȡʱ������ѡȡ���������Ϊ��������������е���ĸ����������ͼ��ѡ������ȷ��ʾ����λ�õ�ѡ����������������ĸ����
 |
 ������������ ���������
������������ ��������λ�� ��3�� ������Һ�IJ�����������ͼ����ͼ��ʾ�����ͼ����Ӧ����ͼ�е� ����ѡ����ĸ��֮�䡣
A������ڡ� B��������� C������� D�������

 |
�ڶ������к͵ζ�����ȷ���ռ���Ʒ�Ĵ��ȡ�
A���ü�ʽ�ζ�����ȡ25��00 mL�ռ���Һ����ƿ�У������뼸�η�̪��ָʾ��
B����Ũ��Ϊc mol / L���������Һװ����ϴ�õ���ʽ�ζ����У�����Һ��ʹ��ʼ����ΪV1 mL
C������ƿ�µ�һ�Ű�ֽ���ζ����յ�ʱ�����¶���ΪV2 mL
����գ�
��1���ζ����յ���ж����ǣ����������һ��������Һʱ����Һ����ɫ�� ɫ��Ϊ ɫ��
��2��C����IJ����У���ƿ�µ�һ�Ű�ֽ�������� ��
��3�����ռ���Ʒ����Ϊ_________________________��
 �����ҵ��ٿ���������������ϵ�д�
�����ҵ��ٿ���������������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����



�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
����֪�ⶨ�к��ȵ�ʵ�鲽�����£�����ȡ50mL 0.25mol/L���ᵹ��С�ձ��У������¶� ����ȡ50mL 0.55mol/L NaOH��Һ�������¶ȣ� �۽�NaOH��Һ����С�ձ��У���Ͼ��Ⱥ�������Һ�¶ȡ���ش�
��1��NaOH��Һ�Թ�����ԭ�� ��
��2������NaOH��Һ����ȷ������ (����ĸ����
A���ز������������� B��һ��Ѹ�ټ��� C�������μ���
��3��ʹ������NaOH��Һ��Ͼ��ȵ���ȷ������ ��
��4������Һ���ܶȾ�Ϊ1g��cm-3,�кͺ���Һ�ı�����c=4.18J��(g����)-1�������ʵ����������к���Ϊ�������� ���� д���÷�Ӧ���Ȼ�ѧ����ʽ_________
��5��������0.5 mol H2SO4��Ũ�����뺬1 mol NaOH����Һ��ϣ��ų������� ���С�ڡ��������ڡ����ڡ���57.3 kJ��ԭ�������������� ����
��ij�ռ���Ʒ�����������������õ����ʣ�Ϊ�˲ⶨ�䴿�ȣ��������²�����
A����250 mL������ƿ�ж������250 mL�ռ���Һ
B���ü�ʽ�ζ�����ȡ25.00 mL�ռ���Һ����ƿ�У������뼸�η�̪��ָʾ��
C������ƽ��ȷ��ȡ�ռ���ƷW g�����ձ���������ˮ�ܽ�
D�������ʵ���Ũ��ΪM�ı�������Һװ����ϴ�õ���ʽ�ζ����У�����Һ��ʹ��ʼ����ΪV1 mL
E.����ƿ�µ�һ�Ű�ֽ���ζ�����Һǡ���ɺ�ɫ��Ϊ��ɫʱ�����¶���ΪV2 mL
����գ�
��1����ȷ���������˳���� (����ĸ��ʾ����
��2���۲�ζ���Һ��ĸ߶�ʱӦע��
��3��E����IJ�������ƿ�µ�һ�Ű�ֽ�������� ��
��4��ijѧ��ʵ��ʱ����ƿ���ռ���Ʒ��Һϴ�ӣ�ʹ�ⶨ��Ũ��_________���ƫ�ߡ���ƫ�͡�����Ӱ�족����ԭ����
��5�����ռ���Ʒ���ȵļ���ʽΪ_________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��16�֣�ij�ռ���Ʒ�����������������õ����ʣ�Ϊ�˲ⶨ�䴿�ȣ�������������������
��һ��������500 ml �ռ���Ʒ��Һ��
��1���������ƿ�Ƿ�©Һ�ķ����ǣ���ƿ�ڼ���һ����ˮ������ƿ����
��2��������Ϊ13��1 g�Ŀ��ձ�����Ҫ��ȡ�ռ���Ʒ20 g �������������ƽ�ϳ�ȡʱ������ѡȡ���������Ϊ��������������е���ĸ����������ͼ��ѡ������ȷ��ʾ����λ�õ�ѡ����������������ĸ����
 |
 ������������ ���������
������������ ��������λ����3�� ������Һ�IJ�����������ͼ����ͼ��ʾ�����ͼ����Ӧ����ͼ�е� ����ѡ����ĸ��֮�䡣
A������ڡ� B��������� C������� D�������

 |
�ڶ������к͵ζ�����ȷ���ռ���Ʒ�Ĵ��ȡ�
A���ü�ʽ�ζ�����ȡ25��00 mL�ռ���Һ����ƿ�У������뼸�η�̪��ָʾ��
B����Ũ��Ϊc mol / L���������Һװ����ϴ�õ���ʽ�ζ����У�����Һ��ʹ��ʼ����ΪV1 mL
C������ƿ�µ�һ�Ű�ֽ���ζ����յ�ʱ�����¶���ΪV2 mL
����գ�
��1���ζ����յ���ж����ǣ����������һ��������Һʱ����Һ����ɫ�� ɫ��Ϊ ɫ��
��2��C����IJ����У���ƿ�µ�һ�Ű�ֽ�������� ��
��3�����ռ���Ʒ����Ϊ_________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�����ʡ��ˮһ�и߶���ѧ�ڵ�һ�ο��Ի�ѧ�� ���ͣ������
ʵ���⣺�����9�֣�ij�ռ���Ʒ�����������������õ����ʣ�Ϊ�˵ζ��䴿�ȣ��������µζ�������?
| A����250 mL������ƿ������250 mL�ռ���Һ�� | B���ü�ʽ�ζ�����ȡ25.00 mL�ռ���Һ����ƿ�в��μӼ��η�̪��ָʾ����? | C������ƽ��ȷ��ȡ�ռ���ƷW��g�����ձ���������ˮ�ܽ⣻ | D�������ʵ���Ũ��Ϊcmol/L�ı�������Һװ����ʽ�ζ��ܡ�����Һ����¿�ʼ����ΪV1 mL��E.����ƿ�µ�һ�Ű�ֽ���ζ����һ������ɫ�պ���ʧ Ϊֹ������Ӻָ���ɫ�����¶���V2 mL��? Ϊֹ������Ӻָ���ɫ�����¶���V2 mL��? |
 2)�۲�ζ�����Һ��ĸ߶�ʱӦע�⣺ ��?
2)�۲�ζ�����Һ��ĸ߶�ʱӦע�⣺ ��?�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com