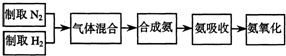
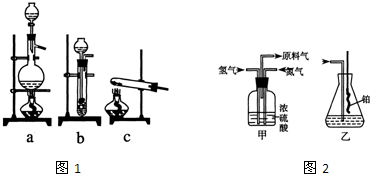
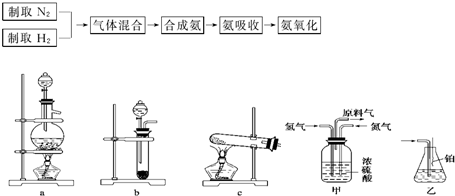
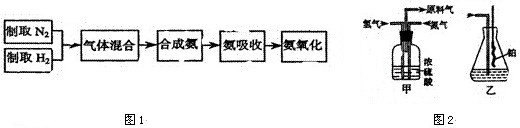
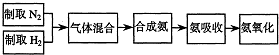
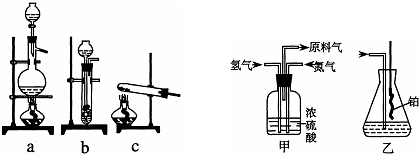
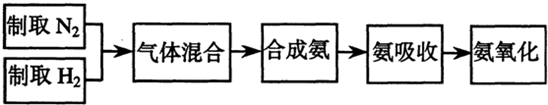
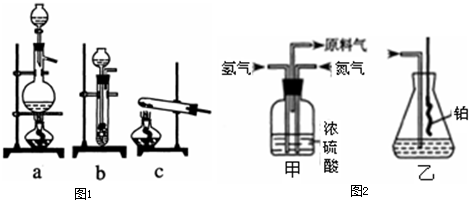
ʵ����ģ��ϳɰ��Ͱ����������������£�

��֪ʵ���ҿ��ñ����������ƣ�NaNO
2����Һ�뱥���Ȼ����Һ�����Ⱥ�Ӧ��ȡ������
��1������ͼa��b��c����װ����ѡ����ȡ����ĺ���װ�ã�

�Ƶ���ѡ��______װ�á�������ѡ��______װ�ã���װ�ñ�ţ���
��2��������������ͨ����ͼ��װ�ã���װ�õ����ó��˽��������⣬���ܹ���______��______�����ã�
��3�����ϳ�����������ȴ����������ⳣ���д����ĵ�����������������������ͨ����װ�õ�ˮ�����հ������ᷢ����������ԭ����______��
��4������װ������һ��ʱ�䰱����ͨ�������ͬʱ�����ȵIJ�˿������װ�õ���ƿ�ڣ���ʹ��˿���ֺ��ȵ�ԭ���ǣ�______����ƿ�л��п������ɵ������ǣ�______���ɶ�ѡ����
A H
2B NO
2 C HNO
3 D NH
4NO
3��5��д����װ���а��������Ļ�ѧ����ʽ��______��4NO+6H
2O



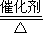 4NO+6H2O��
4NO+6H2O�� 4NO+6H2O��
4NO+6H2O��