
|
��30 ml 0.04 mol��L��1��AlCl3��Һ�У���μ���0.2 mol��L��1��NaOH��Һ����ʼ�г������ɣ����ӵ�a mlʱ�����պ��ܽ⣻�����6 ml 0.2 mol��L��1 NaOH��Һ����μ���0.04 mol��L��1��AlCl3��Һ�����ӵ�b mlʱ���ɵij������������a��b����ֵ�ֱ��� | |
| [����] | |
A�� |
12��10 |
B�� |
24��10 |
C�� |
24��5 |
D�� |
12��5 |
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ���¿αꡡ�̲�ȫ�⡡���л�ѧ�����˽̰�(����1) �˽̰� ���ͣ�013
|
��30 mL��0.04 mol/L��A1Cl3��Һ����μ���0.2 mol/L��NaOH��Һ����ʼʱ�г������ɣ����ӵ�a mLʱ�����պ��ܽ⣬�����6 mL��0.2 mol/L��NaOH��Һ����μ���0.04 mol/L��AlCl3��Һ�����ӵ�b mLʱ���ɳ��������������a��b����ֵ�ֱ�Ϊ | |
| [����] | |
A�� |
12��10 |
B�� |
24��10 |
C�� |
24��5 |
D�� |
12��5 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
Ϊ̽��ͭ��Ũ����ķ�Ӧ��ij��ȤС�����������ʵ�顣
��ʵ��1��ͭ��Ũ���ᷴӦ��ʵ��װ����ͼ��ʾ��
(1)��װ��A�з�����Ӧ�Ļ�ѧ����ʽΪ��
(2)��Ϩ��ƾ��ƺ���Ϊ�е��ܴ��ڣ�B�е�Һ�岻�ᵹ������ԭ���ǣ� ��
���װ��ǰ����������Ϊ��ʹװ���еIJ���������ȫ�����գ�Ӧ����ȡ�IJ����ǣ� ��
��ʵ��2��ʵ���з����Թ��ڳ��˲�����ɫ�����⣬��ͭ˿���滹�к�ɫ��������ɣ����п��ܺ�������ͭ��������ͭ����ͭ������ͭ��
�������ϣ�
��������ͭ�����Ի����»ᷢ������������ԭ��Ӧ����Cu2+��ͭ���ʣ��ڿ����г�����գ�����ת��Ϊ����ͭ��
����ͭ������ͭ�����¶�������ϡ���ᣬ�ڿ����г�����գ���ת��Ϊ����ͭ�Ͷ�������
Ϊ���о��ijɷ֣���С��ͬѧ���ռ����㹻���Ĺ������������ʵ�飺
(3)��������м��������Ƿ�ϴ�Ӹɾ���ʵ�鷽���ǣ� ��
(4)���������ڿ���������ʱ��ʹ�õ�ʵ���������˲����������żܡ��ƾ����⣬�������У�
��
(5)��ȷ�����Ƿ�������ȫ�IJ����ǣ� ��
(6)�����չ�����һ�������Ļ�ѧ��Ӧ����ʽΪ�� ��
������泥�NH4ClO4���Ǹ��ϻ���ƽ�������Ҫ�ɷ֣�ʵ���ҿ�ͨ�����з�Ӧ��ȡ��
��������NaClO4(aq)+NH4Cl(aq) NH4ClO4(aq)+NaCl(aq)
(7)���ð�����Ũ�������NH4Cl��������Ӧ����Ҫ��繩�Ⱦ��ܽ��У���ԭ����:
��
(8)����Ӧ�õ��Ļ����Һ��NH4ClO4��NaCl�����������ֱ�Ϊ0.30��0.l5��������ʵ��ܽ����������ͼ�����ӻ����Һ�л�ý϶�NH4ClO4�����ʵ���������Ϊ����������ƣ�����Ũ���� �����ˣ� �����
Ϊ�˲ⶨ��Ʒ��NH4ClO4�ĺ�����װ������ͼ��ʾ������װ�á������̶�װ������ȥ����ʵ�鲽�����£�
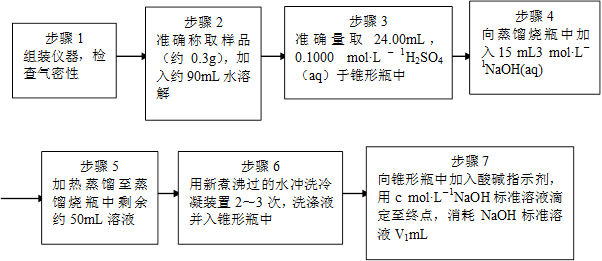
(9)������3�У�ȷȡ��24.00 mL H2SO4(aq)�IJ��������� ��
(10)��ʵ����ȷ�����ɵİ���ϡ������ȫ���յ�ʵ�鲽���� ����д�����ţ���
(11)�������ظ�ʵ��2~3�Σ���ԭ���� ��
(12)�����0.320 g�����к�NH4ClO4��ȷֵΪ0.095g����ʵ��ⶨ�����0.092g ����ʵ��
��������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012���Ϻ���������������ѧ����ĩ������⻯ѧ�Ծ� ���ͣ�ʵ����
Ϊ̽��ͭ��Ũ����ķ�Ӧ��ij��ȤС�����������ʵ�顣
��ʵ��1��ͭ��Ũ���ᷴӦ��ʵ��װ����ͼ��ʾ��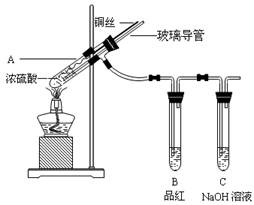
(1)��װ��A�з�����Ӧ�Ļ�ѧ����ʽΪ��
(2)��Ϩ��ƾ��ƺ���Ϊ�е��ܴ��ڣ�B�е�Һ�岻�ᵹ������ԭ���ǣ� ��
���װ��ǰ����������Ϊ��ʹװ���еIJ���������ȫ�����գ�Ӧ����ȡ�IJ����ǣ� ��
��ʵ��2��ʵ���з����Թ��ڳ��˲�����ɫ�����⣬��ͭ˿���滹�к�ɫ��������ɣ����п��ܺ�������ͭ��������ͭ����ͭ������ͭ��
�������ϣ�
��������ͭ�����Ի����»ᷢ������������ԭ��Ӧ����Cu2+��ͭ���ʣ��ڿ����г�����գ�����ת��Ϊ����ͭ��
����ͭ������ͭ�����¶�������ϡ���ᣬ�ڿ����г�����գ���ת��Ϊ����ͭ�Ͷ�������
Ϊ���о��ijɷ֣���С��ͬѧ���ռ����㹻���Ĺ������������ʵ�飺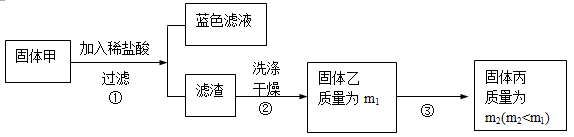
(3)��������м��������Ƿ�ϴ�Ӹɾ���ʵ�鷽���ǣ� ��
(4)���������ڿ���������ʱ��ʹ�õ�ʵ���������˲����������żܡ��ƾ����⣬�������У�
��
(5)��ȷ�����Ƿ�������ȫ�IJ����ǣ� ��
(6)�����չ�����һ�������Ļ�ѧ��Ӧ����ʽΪ�� ��
������泥�NH4ClO4���Ǹ��ϻ���ƽ�������Ҫ�ɷ֣�ʵ���ҿ�ͨ�����з�Ӧ��ȡ��
��������NaClO4(aq)+NH4Cl(aq) NH4ClO4(aq)+NaCl(aq)
NH4ClO4(aq)+NaCl(aq)
(7)���ð�����Ũ�������NH4Cl��������Ӧ����Ҫ��繩�Ⱦ��ܽ��У���ԭ����:
��
(8)����Ӧ�õ��Ļ����Һ��NH4ClO4��NaCl�����������ֱ�Ϊ0.30��0.l5��������ʵ��ܽ����������ͼ�����ӻ����Һ�л�ý϶�NH4ClO4�����ʵ���������Ϊ����������ƣ�����Ũ���� �����ˣ� �����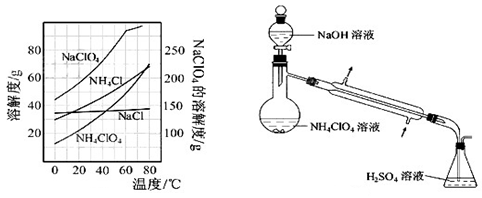
Ϊ�˲ⶨ��Ʒ��NH4ClO4�ĺ�����װ������ͼ��ʾ������װ�á������̶�װ������ȥ����ʵ�鲽�����£�
(9)������3�У�ȷȡ��24.00 mL H2SO4(aq)�IJ��������� ��
(10)��ʵ����ȷ�����ɵİ���ϡ������ȫ���յ�ʵ�鲽���� ����д�����ţ���
(11)�������ظ�ʵ��2~3�Σ���ԭ���� ��
(12)�����0.320 g�����к�NH4ClO4��ȷֵΪ0.095g����ʵ��ⶨ�����0.092g����ʵ��
��������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ���Ϻ���������������ѧ����ĩ������⻯ѧ�Ծ� ���ͣ�ʵ����
Ϊ̽��ͭ��Ũ����ķ�Ӧ��ij��ȤС�����������ʵ�顣
��ʵ��1��ͭ��Ũ���ᷴӦ��ʵ��װ����ͼ��ʾ��

(1)��װ��A�з�����Ӧ�Ļ�ѧ����ʽΪ��
(2)��Ϩ��ƾ��ƺ���Ϊ�е��ܴ��ڣ�B�е�Һ�岻�ᵹ������ԭ���ǣ� ��
���װ��ǰ����������Ϊ��ʹװ���еIJ���������ȫ�����գ�Ӧ����ȡ�IJ����ǣ� ��
��ʵ��2��ʵ���з����Թ��ڳ��˲�����ɫ�����⣬��ͭ˿���滹�к�ɫ��������ɣ����п��ܺ�������ͭ��������ͭ����ͭ������ͭ��
�������ϣ�
��������ͭ�����Ի����»ᷢ������������ԭ��Ӧ����Cu2+��ͭ���ʣ��ڿ����г�����գ�����ת��Ϊ����ͭ��
����ͭ������ͭ�����¶�������ϡ���ᣬ�ڿ����г�����գ���ת��Ϊ����ͭ�Ͷ�������
Ϊ���о��ijɷ֣���С��ͬѧ���ռ����㹻���Ĺ������������ʵ�飺
(3)��������м��������Ƿ�ϴ�Ӹɾ���ʵ�鷽���ǣ� ��
(4)���������ڿ���������ʱ��ʹ�õ�ʵ���������˲����������żܡ��ƾ����⣬�������У�
��
(5)��ȷ�����Ƿ�������ȫ�IJ����ǣ� ��
(6)�����չ�����һ�������Ļ�ѧ��Ӧ����ʽΪ�� ��
������泥�NH4ClO4���Ǹ��ϻ���ƽ�������Ҫ�ɷ֣�ʵ���ҿ�ͨ�����з�Ӧ��ȡ��
��������NaClO4(aq)+NH4Cl(aq) NH4ClO4(aq)+NaCl(aq)
NH4ClO4(aq)+NaCl(aq)
(7)���ð�����Ũ�������NH4Cl��������Ӧ����Ҫ��繩�Ⱦ��ܽ��У���ԭ����:
��
(8)����Ӧ�õ��Ļ����Һ��NH4ClO4��NaCl�����������ֱ�Ϊ0.30��0.l5��������ʵ��ܽ����������ͼ�����ӻ����Һ�л�ý϶�NH4ClO4�����ʵ���������Ϊ����������ƣ�����Ũ���� �����ˣ� �����

Ϊ�˲ⶨ��Ʒ��NH4ClO4�ĺ�����װ������ͼ��ʾ������װ�á������̶�װ������ȥ����ʵ�鲽�����£�
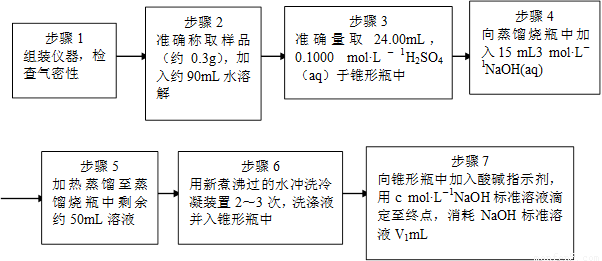
(9)������3�У�ȷȡ��24.00 mL H2SO4(aq)�IJ��������� ��
(10)��ʵ����ȷ�����ɵİ���ϡ������ȫ���յ�ʵ�鲽���� ����д�����ţ���
(11)�������ظ�ʵ��2~3�Σ���ԭ���� ��
(12)�����0.320 g�����к�NH4ClO4��ȷֵΪ0.095g����ʵ��ⶨ�����0.092g ����ʵ��
��������Ϊ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com