
”¾ĢāÄæ”æĻĀ±ķŹĒÉś»īÉś²śÖŠ³£¼ūµÄĪļÖŹ£¬±ķÖŠĮŠ³öĮĖĖüĆĒµÄ(Ö÷ŅŖ£©³É·Ö”£
±ąŗÅ | ¢Ł | ¢Ś | ¢Ū | ¢Ü | ¢Ż | ¢Ž | ¢ß |
Ćū³Ę | ¾Ę¾« | “×Ėį | »š¼ī | Ź³ŃĪ | Ķµ¼Ļß | ŃĒĮņĖįōū | ĖÕ“ņ |
Ö÷ŅŖ³É·Ö | CH3CH2OH | CH3COOH | NaOH | NaCl | Cu | SO2 | Na2CO3 |
£Ø1£©ĒėÄć¶Ō±ķÖŠ¢Ł”«¢ßµÄÖ÷ŅŖ³É·Ö½ųŠŠ·ÖĄą(Ģī±ąŗÅ£©£ŗŹōÓŚµē½āÖŹµÄŹĒ______£¬ŹōÓŚ·Ēµē½āÖŹ_______”£
£Ø2£©¹żĮæ¢ŚµÄĖ®ČÜŅŗÓė¢ß·“Ó¦µÄĄė×Ó·½³ĢŹ½______________________”£
£Ø3£©Ä³Ķ¬Ń§ÓĆ¢ŻŗĶÅØĮņĖį¹²ČČĄ“Öʱø¢Ž£¬»Æѧ·½³ĢŹ½ĪŖ£ŗCu+2H2SO4(ÅØ£©![]() CuSO4+SO2”ü+2H2O
CuSO4+SO2ӟ+2H2O
¢ŁĒėÓƵ„ĻßĒűź³öµē×Ó×ŖŅʵÄĒéæö____________£»
¢ŚÅØH2SO4±ķĻÖ³öĄ“µÄŠŌÖŹŹĒ£ŗ_______£»µ±µē×Ó×ŖŅĘ0.1molŹ±£¬ĻūŗÄŃõ»Æ¼ĮµÄĪļÖŹµÄĮæĪŖ_______”£
£Ø4£©ČēĶ¼±ķŹ¾Ä³Ķ¬Ń§ÅäÖĘ480mL 0.5mol/L µÄNaOHČÜŅŗµÄ²æ·Ö²Ł×÷Ź¾ŅāĶ¼£¬ĘäÖŠÓŠ“ķĪóµÄŹĒ_______£¬ÕāŃł²Ł×÷ĖłÅäÖʵÄČÜŅŗ±ČŅŖĒóµÄÅضČŅŖ_________ (Ģī”°Ę«øß”±”¢”°Ę«µĶ”±”¢”°²»Ó°Ļģ”±£©”£ÅäÖĘÓ¦³ĘČ”NaOH________g”£

”¾“š°ø”æ¢Ś¢Ū¢Ü¢ß ¢Ł¢Ž 2CH3COOH+CO32-=2CH3COO-+CO2”ü+H2O ![]() ĖįŠŌ”¢Ńõ»ÆŠŌ 0.05moL C Ę«µĶ 10.0
ĖįŠŌ”¢Ńõ»ÆŠŌ 0.05moL C Ę«µĶ 10.0
”¾½āĪö”æ
£Ø1£©ŹōÓŚµē½āÖŹµÄÓŠ“×Ėį”¢»š¼ī”¢Ź³ŃĪ”¢ĖÕ“ņ£¬“š°øŃ”¢Ś¢Ū¢Ü¢ß£»ŹōÓŚ·Ēµē½āÖŹµÄÓŠ¾Ę¾«”¢ŃĒĮņĖįōū£¬“š°øŃ”¢Ł¢Ž£»£Ø2£©“×ĖįÓėĢ¼ĖįÄĘ·“Ó¦µÄ·½³ĢŹ½ĪŖ£ŗ2CH3COOH+CO32-=2CH3COO-+CO2”ü+H2O£»£Ø3£©¢ŁŃõ»Æ»¹Ō·“Ó¦ÖŠµē×Ó×ŖŅĘĒéæöµÄ±ķŹ¾æÉÓƵ„ĻßĒűķŹ¾£¬ĒéæöČēĻĀ£ŗ![]() £»¢ŚÅØH2SO4±ķĻÖ³öĄ“µÄŠŌÖŹŹĒ£ŗĖįŠŌ”¢Ńõ»ÆŠŌ£»øł¾Ż·“Ó¦·½³ĢŹ½æÉÖŖ£¬µ±µē×Ó×ŖŅĘ0.1molŹ±£¬ĻūŗÄŃõ»Æ¼ĮµÄĪļÖŹµÄĮæĪŖ
£»¢ŚÅØH2SO4±ķĻÖ³öĄ“µÄŠŌÖŹŹĒ£ŗĖįŠŌ”¢Ńõ»ÆŠŌ£»øł¾Ż·“Ó¦·½³ĢŹ½æÉÖŖ£¬µ±µē×Ó×ŖŅĘ0.1molŹ±£¬ĻūŗÄŃõ»Æ¼ĮµÄĪļÖŹµÄĮæĪŖ![]() =0.05mol£»£Ø4£©¶ØČŻŹ±ŃŪ¾¦ÓėæĢ¶ČĻßĻąĘ½£¬²»ÄÜø©ŹÓ»ņŃöŹÓ£»¶ØČŻŹ±£¬ŃŪ¾¦ŃöŹÓæĢ¶ČĻߣ¬ČÜŅŗµÄŅŗĆę³¬¹żæĢ¶ČĻߣ¬ČÜŅŗµÄĢå»żĘ«“ó£¬ÅضČĘ«Š”£¬¹Ź“š°øĪŖ£ŗC£» Ę«µĶ£»ÅäÖĘ480mL 0.5mol/L µÄNaOHČÜŅŗŹ±£¬ÓÉӌƻӊøĆ¹ęøńµÄČŻĮæĘ棬±ŲŠėÅäÖĘ500mL 0.5mol/L µÄNaOHČÜŅŗ£¬Ó¦³ĘČ”NaOH 0.5L”Į0.5mol/L”Į40g/mol=10.0g”£
=0.05mol£»£Ø4£©¶ØČŻŹ±ŃŪ¾¦ÓėæĢ¶ČĻßĻąĘ½£¬²»ÄÜø©ŹÓ»ņŃöŹÓ£»¶ØČŻŹ±£¬ŃŪ¾¦ŃöŹÓæĢ¶ČĻߣ¬ČÜŅŗµÄŅŗĆę³¬¹żæĢ¶ČĻߣ¬ČÜŅŗµÄĢå»żĘ«“ó£¬ÅضČĘ«Š”£¬¹Ź“š°øĪŖ£ŗC£» Ę«µĶ£»ÅäÖĘ480mL 0.5mol/L µÄNaOHČÜŅŗŹ±£¬ÓÉӌƻӊøĆ¹ęøńµÄČŻĮæĘ棬±ŲŠėÅäÖĘ500mL 0.5mol/L µÄNaOHČÜŅŗ£¬Ó¦³ĘČ”NaOH 0.5L”Į0.5mol/L”Į40g/mol=10.0g”£


 ŌĶĮæģ³µĻµĮŠ“š°ø
ŌĶĮæģ³µĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æĻĀĮŠĖµ·ØÕżČ·µÄŹĒ
A. Na2O2ÖŠŅõŃōĄė×ӵĵÄøöŹż±ČĪŖ1”Ć1
B. ŌŖĖŲÖÜĘŚ±ķÖŠ£¬°üŗ¬ŌŖĖŲÖÖŹż×ī¶ąµÄ×åŹĒµŚ¢óB×å
C. NaClŗĶHClĘų»ÆŹ±£¬æĖ·ž×÷ÓĆĮ¦ĶźČ«ĻąĶ¬
D. ½šŹō»ÆŗĻĪļŅ»¶ØŹĒĄė×Ó»ÆŗĻĪļ£¬²»ŗ¬½šŹōŌŖĖŲµÄ»ÆŗĻĪļÓŠæÉÄÜŹĒĄė×Ó»ÆŗĻĪļ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æ¢ńijČÜŅŗÖŠÖ»æÉÄÜŗ¬ÓŠFe2£«”¢Mg2£«”¢Cu2£«”¢NH4+”¢Al3£«”¢Cl£”¢OH£”¢CO32£”£µ±¼ÓČėŅ»ÖÖµ»ĘÉ«¹ĢĢå²¢¼ÓČČŹ±£¬ÓŠ“Ģ¼¤ŠŌĘųĢå·Å³öŗĶ°×É«³Įµķ²śÉś£¬¼ÓČėµ»ĘÉ«¹ĢĢåµÄĪļÖŹµÄĮæ£Øŗį×ų±ź£©ÓėĪö³öµÄ³ĮµķŗĶ²śÉśĘųĢåµÄĪļÖŹµÄĮæ£Øׯ×ų±ź£©µÄ¹ŲĻµČēĻĀĶ¼ĖłŹ¾”£øƵ»ĘÉ«ĪļÖŹ×öŃęÉ«·“Ó¦ŹµŃéĻŌ»ĘÉ«”£æÉÖŖČÜŅŗÖŠŗ¬ÓŠµÄĄė×ÓŹĒ________________£»Ėłŗ¬Ąė×ÓµÄĪļÖŹµÄĮæÅضČÖ®±ČĪŖ____________£»Ėł¼ÓµÄµ»ĘÉ«¹ĢĢåŹĒ______________”£

¢ņ£®ŹµŃéŹŅ²ÉÓƵĪ¶Ø·Ø²ā¶ØijĖ®ŃłÖŠŃĒĮņĖįŃĪŗ¬Įæ£ŗ

£Ø1£©µĪ¶ØŹ±£¬KIO3ŗĶKI×÷ÓĆĪö³öI2£¬Ķź³É²¢ÅäĘ½ĻĀĮŠĄė×Ó·½³ĢŹ½£ŗ__IO3££«____I££«____===____I2£«____H2O”£
£Ø2£©·“Ó¦£Ø1£©ĖłµĆI2µÄ×÷ÓĆŹĒ_________________________”£
£Ø3£©µĪ¶ØÖÕµćŹ±£¬100mLµÄĖ®Ńł¹²ĻūŗÄx mL±ź×¼ČÜŅŗ”£ČōĻūŗÄ1mL±ź×¼ČÜŅŗĻąµ±ÓŚSO32£µÄÖŹĮæ1g£¬ŌņøĆĖ®ŃłÖŠSO32£µÄŗ¬ĮæĪŖ________mg”¤L£1”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æ»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ÓĆ”°£¾”±»ņ”°£¼”±ĢīŠ“ĻĀ±ķ”£
µŚŅ»µēĄėÄÜ | ČŪµć | ·Šµć(Ķ¬Ń¹) | ¼üÄÜ |
P____S | MgO____CaS | CF4____SiCl4 | H£Cl____ H£Br |
£Ø2£©Ņ»ÖÖÖĘČ”NH2OHµÄ·“Ó¦ĪŖ2NO2£+4SO2 + 6H2O+ 6NH3 = 4SO![]() +6NH4++2NH2OH”£
+6NH4++2NH2OHӣ
¢ŁN”¢H”¢OµÄµēøŗŠŌ“ÓŠ”µ½“óµÄĖ³ŠņĪŖ____”£
¢ŚNH3·Ö×ÓµÄæռ乹ŠĶĪŖ____”£
¢ŪNH2OH¼«Ņ×ČÜÓŚH2O£¬³żŅņĪŖĖüĆĒ¶¼ŹĒ¼«ŠŌ·Ö×ÓĶā£¬»¹ŅņĪŖ____”£
£Ø3£©ÅäŗĻĪļK[PtCl3(NH3)]ÖŠ[PtCl3(NH3)]£µÄ½į¹¹æÉÓĆŹ¾ŅāĶ¼±ķŹ¾ĪŖ____£Ø²»æ¼ĀĒæռ乹ŠĶ£©”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æFeCl3Ź“æĢŅŗÓÉFeCl3ŗĶŃĪĖįµČ×é³É£¬æÉÓĆÓŚŹ“æĢĶ¼°ĶŗĻ½šµČ”£
£Ø1£©¼ÓČėŃĪĖįæÉŅÖÖĘFe3+¼°Éś³ÉµÄCu2+µÄĖ®½ā£¬ Fe3+Ė®½āµÄĄė×Ó·½³ĢŹ½ĪŖ____”£
£Ø2£©FeCl3ČÜŅŗŹ“æĢĶµÄ»Æѧ·½³ĢŹ½ĪŖ____”£
£Ø3£©²ā¶Øij·ĻŹ“æĢŅŗÖŠCu2+ŗ¬ĮæµÄŹµŃé²½ÖčČēĻĀ£ŗĮæČ”25.00mL·ĻŹ“æĢŅŗ£¬¼ÓČė×ćĮæµÄNH4HF2£ØŃŚ±ĪFe3+£¬Ļū³żøÉČÅ£©£¬¼ÓČėÉŌ¹żĮæKIČÜŅŗ£Ø2Cu2++4I£=2CuI”ż+ I2£©£¬ŌŚ°µ“¦¾²ÖĆ5min£¬Č»ŗóÓĆ0.02000mol”¤L£1Na2S2O3±ź×¼ČÜŅŗµĪ¶Ø£ØI2+2S2O![]() =2I£+S4O
=2I£+S4O![]() £©£¬½Ó½üÖÕµćŹ±£¬¼ÓČėµķ·ŪŗĶ10mL10%NH4SCNČÜŅŗ£ØCuIæÉĪüø½ÉŁĮæI2£¬ CuSCN²»Īüø½I2£©£¬¼ĢŠųµĪ¶ØÖĮÖÕµć£¬ĻūŗÄNa2S2O3ČÜŅŗ20.00mL”£
£©£¬½Ó½üÖÕµćŹ±£¬¼ÓČėµķ·ŪŗĶ10mL10%NH4SCNČÜŅŗ£ØCuIæÉĪüø½ÉŁĮæI2£¬ CuSCN²»Īüø½I2£©£¬¼ĢŠųµĪ¶ØÖĮÖÕµć£¬ĻūŗÄNa2S2O3ČÜŅŗ20.00mL”£
¢Ł²»¼ÓNH4HF2»įµ¼ÖĀ²āµĆµÄĶµÄŗ¬Įæ____£ØĢī£ŗ”°Ę«øß”±”°Ę«µĶ”±»ņ”°²»±ä”±£©”£
¢ŚĶعż¼ĘĖćČ·¶ØøĆ·ĻŹ“æĢŅŗÖŠĶµÄŗ¬Įæ£Øµ„Ī»g”¤L£1£¬Š“³ö¼ĘĖć¹ż³Ģ£©____”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æŅŃÖŖA”¢B”¢C”¢D”¢E”¢F”¢G”¢H”¢I¾ÅÖÖĪļÖŹ£¬ĘäÖŠA”¢B”¢D”¢EŃęÉ«·“Ó¦¾łĪŖ×ĻÉ«(Ķø¹żĄ¶É«īܲ£Į§)£¬G”¢FŹĒµ„ÖŹ£¬ĘäÓą¾łĪŖ»ÆŗĻĪļ£¬HŹĒŅ»ÖÖµ»ĘÉ«¹ĢĢ壬ĖüĆĒµÄ×Ŗ»Æ¹ŲĻµČēĶ¼ĖłŹ¾”£Ēė»Ų“š£ŗ
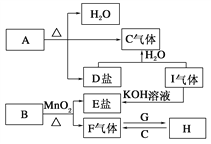
£Ø1£©»³öGĄė×Ó½į¹¹Ź¾ŅāĶ¼________£»
£Ø2£©Š“³öB”¢IµÄ»ÆѧŹ½B________£¬I________£»
£Ø3£©Š“³öHµÄŅ»ÖÖÓĆĶ¾__________________________________£»
£Ø4£©Š“³öAŹÜČČ·Ö½āµÄ»Æѧ·½³ĢŹ½_________________________£»
£Ø5£©Čō10gµÄC6H12O6ŌŚŃõĘųÖŠ³ä·ÖČ¼ÉÕ£¬½«²śĪļČ«²æÓė×ćĮæµÄH³ä·Ö·“Ó¦£¬·“Ó¦ŗó¹ĢĢåŌöÖŲ____________g”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æijŹµŃ銔×é¶Ō֊ѧæĪ±¾ÖŠæÉÉś³ÉĒāĘųµÄ·“Ó¦½ųŠŠĮĖŃŠ¾æ,×ܽį³öČżøöæÉŅŌÉś³ÉH2µÄ·“Ó¦:¢ŁZn+ŃĪĖį;¢ŚNa+Ė®;¢ŪAl+NaOHČÜŅŗ”£ĪŖµćČ¼ÉĻŹöČżøö·“Ӧɜ³ÉµÄH2,ĖūĆĒÉč¼ĘĮĖČēÓŅĶ¼ĖłŹ¾µÄ×°ÖĆĶ¼:Ēė»Ų“šĻĀĮŠĪŹĢā:
![]()
£Ø1£©Š“³öAlŗĶNaOHČÜŅŗ·“Ó¦µÄĄė×Ó·½³ĢŹ½_______________________________”£
£Ø2£©ŌŚµćČ¼H2Ö®Ē°±ŲŠėĻČ½ųŠŠ____________________________________________”£

£Ø3£©ŹµŃ銔×éŌŚµćČ¼ÓĆÉĻŹö×°ÖĆÖʵƵÄH2Ź±,¢Ł¢ŪŹµŃé»ńµĆ³É¹¦,¢ŚČ“Ź§°ÜĮĖ”£ĖūĆĒ·ÖĪöČĻĪŖŹ§°ÜµÄŌŅņŹĒNaÓėH2OµÄ·“Ó¦ĖŁĀŹĢ«æģ,NaµÄÓĆĮæÓÖĢ«ÉŁ”£ÓŚŹĒĖūĆĒ×¼±øŌö¼ÓÄʵÄÓĆĮæ,æɽĢŹ¦ĖµĢ«Ī£ĻÕ,ÄćČĻĪŖ²śÉśĪ£ĻÕµÄŌŅņŹĒ___________________________”£
£Ø4£©ŹµŃ銔×é²éŌÄÄĘ”¢±½(Ņ»ÖÖ²»ČÜÓŚĖ®µÄŅŗĢ¬ÓŠ»śĪļ)”¢Ė®µÄĆܶȷֱšĪŖ0.97 g”¤mL-1”¢0.88 g”¤mL-1”¢1.00 g”¤mL-1,²¢¾Ż“Ė¶ŌŹµŃé½ųŠŠĮĖøĽų”£ŌŚøĽųŗóµÄŹµŃéÖŠH2µÄÉś³ÉĖŁĀŹ____________”£(Ģī”°¼õĀż”±»ņ”°¼Óæģ”±)
£Ø5£©½«4.6gNaŗĶ8.1gAlĶ¶Čėµ½×ćĮæµÄĖ®ÖŠ£¬ĖłµĆČÜŅŗµÄĢå»żĪŖ200mL£¬ŌņÉś³É±ź×¼×“æöĻĀµÄH2µÄĢå»żŹĒ___________£æøĆČÜŅŗÖŠČÜÖŹµÄĪļÖŹµÄĮæÅØ¶ČŹĒ__________________£æ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æ»ÆѧÓėÉś²ś”¢Éś»ī”¢Éē»įĆÜĒŠĻą¹Ų£¬ĻĀĮŠĖµ·ØÖŠ²»ÕżČ·µÄŹĒ£Ø £©
A.ŗĻ½š²ÄĮĻÖŠæÉÄÜŗ¬ÓŠ·Ē½šŹōŌŖĖŲ
B.ĢśŌŚ³±ŹŖµÄæÕĘųÖŠ·ÅÖĆ£¬Ņ×·¢Éś»ÆѧøÆŹ“¶ųÉśŠā
C.ĀķæŚĢś(¶ĘĪż)µÄ±ķĆęŅ»µ©ĘĘĖš£¬ĢśøÆŹ“¼Óæģ
D.ÉńÖŪ10ŗÅ·É“¬ĖłÓĆĢ«ŃōÄܵē³Ų°åæɽ«¹āÄÜ×Ŗ»»ĪŖµēÄÜ£¬ĖłÓĆ×Ŗ»»²ÄĮĻŹĒµ„¾§¹č
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æQ”¢R”¢X”¢Y”¢ZĪåÖÖŌŖĖŲµÄŌ×ÓŠņŹżŅĄ“ĪµŻŌö”£ŅŃÖŖ£ŗ¢ŁZµÄŌ×ÓŠņŹżĪŖ29£¬ĘäÓąµÄ¾łĪŖ¶ĢÖÜĘŚÖ÷×åŌŖĖŲ£»YŌ×ӵļŪµē×Ó(ĶāĪ§µē×Ó)ÅŲ¼ĪŖmsnmpn£»¢ŚRŌ×ÓŗĖĶāL²ćµē×ÓŹżĪŖĘꏿ£»¢ŪQ”¢XŌ×Óp¹ģµĄµÄµē×ÓŹż·Ö±šĪŖ2ŗĶ4”£Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©Z2£«µÄŗĖĶāµē×ÓÅŲ¼Ź½ŹĒ________”£
£Ø2£©ŌŚ[Z(NH3)4]2£«Ąė×ÓÖŠ£¬Z2£«µÄæÕ¹ģµĄ½ÓŹÜNH3·Ö×ÓĢį¹©µÄ________ŠĪ³ÉÅäĪ»¼ü”£
£Ø3£©QÓėYŠĪ³ÉµÄ×ī¼ņµ„ĘųĢ¬Ēā»ÆĪļ·Ö±šĪŖ¼×”¢ŅŅ£¬ĻĀĮŠÅŠ¶ĻÕżČ·µÄŹĒ________”£
a£®ĪČ¶ØŠŌ£ŗ¼×>ŅŅ£¬·Šµć£ŗ¼×>ŅŅ b£®ĪČ¶ØŠŌ£ŗ¼×>ŅŅ£¬·Šµć£ŗ¼×<ŅŅ
c£®ĪČ¶ØŠŌ£ŗ¼×<ŅŅ£¬·Šµć£ŗ¼×<ŅŅ d£®ĪČ¶ØŠŌ£ŗ¼×<ŅŅ£¬·Šµć£ŗ¼×>ŅŅ
£Ø4£©Q”¢R”¢YČżÖÖŌŖĖŲµÄµŚŅ»µēĄėÄÜŹżÖµÓÉŠ”µ½“óµÄĖ³ŠņĪŖ________(ÓĆŌŖĖŲ·ūŗÅ×÷“š)”£
£Ø5£©QµÄŅ»ÖÖĒā»ÆĪļĻą¶Ō·Ö×ÓÖŹĮæĪŖ26£¬ĘäÖŠ·Ö×ÓÖŠµÄ¦Ņ¼üÓė¦Š¼üµÄ¼üŹżÖ®±ČĪŖ________£¬ĘäÖŠŠÄŌ×ÓµÄŌÓ»ÆĄąŠĶŹĒ________”£
£Ø6£©Ä³ŌŖĖŲŌ×ӵļŪµē×Ó¹¹ŠĶĪŖ3d54s1£¬øĆŌŖĖŲŹōÓŚ________ĒųŌŖĖŲ£¬ŌŖĖŲ·ūŗÅŹĒ________”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com