
��100��ʱ����0.200 mol�������������������2L��յ��ܱ������У�ÿ��һ��ʱ��Ը������ڵ����ʽ��з������õ����±���
ʱ�� | 0 | 20 | 40 | 60 | 80 | 100 |
c(N2O4) | 0.100 | c1 | 0.050 | c3 | a | b |
c(NO2) | 0.000 | 0.060 | c2 | 0.120 | 0.120 | 0.120 |
����գ�
��1���÷�Ӧ�Ļ�ѧ����ʽΪ_______________���ﵽƽ��ʱ������������ת����Ϊ________%��
��2��20sʱ������������Ũ��c1=_____ ___mol��L-1����0��20sʱ����ڣ�������������ƽ����Ӧ����Ϊ_____ ___��
��3��������ͬ���������������г�����Ƕ����������壬Ҫ�ﵽ����ͬ����ƽ��״̬�����������ij�ʼŨ��Ϊ_____ ___��
 ��ĩ1�����ʽ���������ϵ�д�
��ĩ1�����ʽ���������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ��ɽ��ʡ�߶���ѧ�����в��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
�������ʵķ����м��ЦҼ������Цм�����
��HCl ��H2O ��N2 ��H2O2 ��C2H4 ��C2H2
A���٢ڢ� B���ۢܢݢ� C���ۢݢ� D���٢ۢ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016�����ʡ������ѧ�ڵ�һ���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
����˼ά�ǻ�ѧ�����г��õ�һ��˼ά�����������й����ӷ���ʽ��������ȷ����
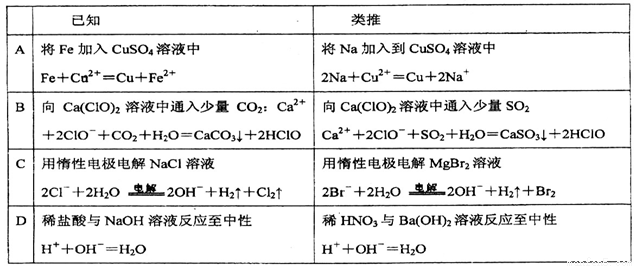
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ������ʡ�߶���ѧ�����в��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
�ۺ���ͼ�жϣ�����˵����ȷ����

A��װ�â��װ�â���ǽ�����ת��Ϊ��ѧ�ܵ�װ��
B��װ�â��װ�â���������Ӧ����O2��2H2O��4e��===4OH��
C��װ�â��װ�â��������е������Ӿ����Ҳ��ձ��ƶ�
D���ŵ�����У�װ�â�����ձ���װ�â��Ҳ��ձ�����Һ��pH������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ������ʡ��һ��ѧ�����в��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
һ����������������ȷֽ�Ļ�ѧ����ʽΪ��5NH4NO3��2HNO3��4N2��9H2O���ڷ�Ӧ�б������뱻��ԭ�ĵ�ԭ����֮��Ϊ
A��5��3 B��5��4 C��1��1 D��3��5
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016�����ʡ������ѧ�����в��Ի�ѧ�Ծ��������棩 ���ͣ�ʵ����
ij��ѧ��ȤС��ͬѧչ����Ư���������� (NaClO2)���о���
ʵ�����ȡNaClO2����
��֪��NaClO2������Һ���¶ȵ���38 ��ʱ�����ľ�����NaClO2��3H2O������38 ��ʱ�����ľ�����NaClO2������60 ��ʱNaClO2�ֽ��NaClO3��NaCl��������ͼ10��ʾװ�ý���ʵ�顣

��1��װ�â۵�������____________________��
��2��װ�â��в���ClO2�Ļ�ѧ����ʽΪ_________________________��װ�â����Ʊ�NaClO2�Ļ�ѧ����ʽΪ______________________��
��3����װ�âܷ�Ӧ�����Һ���NaClO2����IJ�������Ϊ��
�ټ�ѹ��55�������ᾧ���ڳ��ȹ��ˣ���____________���ܵ���60�����õ���Ʒ��
ʵ��ⶨij����������Ʒ�Ĵ��ȡ�
�������ʵ�鷽����������ʵ�飺
��ȷ��ȡ��������������Ʒm g���ձ��У�������������ˮ�����ĵ⻯�ؾ��壬�ٵ���������ϡ���ᣬ��ַ�Ӧ(��֪��ClO2-��4I����4H��===2H2O��2I2��Cl��)�������û��Һ���250 mL������Һ��
����ȡ25.00 mL������Һ����ƿ�У��Ӽ��ε�����Һ����c mol��L��1 Na2S2O3��Һ�ζ������ζ��յ㡣�ظ�2�Σ����ƽ��ֵΪV mL(��֪��I2��2S2O32-===2I����S4O62-)��
��4���ﵽ�ζ��յ�ʱ������Ϊ___________________��
��5������Ʒ��NaClO2����������Ϊ_______________(�ú�m��c��V�Ĵ���ʽ��ʾ)��
��6���ڵζ�������ȷ���������£���ʵ���ý��ƫ�ߣ�ԭ�������ӷ���ʽ��ʾΪ________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016�����ʡ������ѧ�ڵ������¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
����˵������ȷ����
A���������ˮ��HClO��Ũ�ȿ�����ˮ��������̼��Ʒ�ĩ
B���γ��������������Ҫ��SO2�͵��������CO2���������ЧӦ����Ҫ����
C������й©���Ծȷ�������ʪë����պ�д���ˮ��ë����ס�ڱDz�����Ƶ͵ĵط�����
D�������е�N2��ת��ΪNO��NO��ת��ΪNO2������ȣ�������������Ȼ���п�ת��ΪSO2�Ⱥ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016�����ʡ��ˮ�и�����ѧ�ڵ����β��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
Ԫ�����ڱ��Т�AԪ�ذ���C��Si��Ge��Sn��Pb�ȣ���֪Sn��+4���ȶ�����Pb��+2���ȶ��������ѧ֪ʶ���ж����з�Ӧ��(��Ӧ������)��ȷ����
��Pb+2Cl2=PbCl4
��Sn+2Cl2=SnCl4
��SnCl2+Cl2=SnCl4
��PbO2+4HCl=PbCl4+2H2O
��Pb3O4+8HCl=3PbCl2+Cl2��+4H2O
A���ڢۢ� B���ڢۢ� C���٢ڢ� D���٢ڢܢ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ����������������и�һ10���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
ij�Ȼ�þ��Һ���ܶ�Ϊ1.18 g��cm��3������þ���ӵ���������Ϊ5.1%,300 mL����Һ�������ӵ����ʵ���Ϊ
A��0.37 mol B��0.63 mol
C��0.74 mol D��1.5 mol
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com