
Ļņ100 mL FeI2ČÜŅŗÖŠÖš½„ĶØČėCl2£¬»įŅĄ“ĪÉś³ÉI2”¢Fe3£«”¢IO3-£¬ĘäÖŠFe3£«”¢I2µÄĪļÖŹµÄĮæĖęn(Cl2)µÄ±ä»ÆČēĶ¼ĖłŹ¾£¬Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ

£Ø1£©ÓÉĶ¼æÉÖŖ£¬I£”¢Fe2£«”¢I2ČżÖÖĮ£×ӵĻ¹ŌŠŌÓÉĒæµ½ČõµÄĖ³ŠņĪŖ________£¾________£¾________£»
£Ø2£©µ±n(Cl2)£½0.12 molŹ±£¬ČÜŅŗÖŠµÄĄė×ÓÖ÷ŅŖĪŖ________________________________£¬
“ÓæŖŹ¼ĶØČėCl2µ½n(Cl2)£½0.12 molŹ±µÄ×Ü·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ______________________£»
£Ø3£©µ±ČÜŅŗÖŠn(Cl£)”Ćn(IO3-)£½8”Ć1Ź±£¬ĶØČėµÄCl2ŌŚ±ź×¼×“æöĻĀµÄĢå»żĪŖ________”£
£Ø1£©I£””Fe2£«””I2
£Ø2£©Fe2£«”¢Fe3£«”¢Cl£””5FeI2£«6Cl2=5I2£«2FeCl3£«3FeCl2
£Ø3£©8.96 L
”¾½āĪö”æ£Ø1£©øł¾ŻĶ¼ĻńæÉÖŖ£ŗŹ×ĻČI£±»Ńõ»Æ£¬Č»ŗóŹĒFe2£«£¬ĖłŅŌ»¹ŌŠŌĖ³ŠņĪŖI££¾Fe2£«£¾I2”££Ø2£©ÓÉĶ¼ĻńæÉÖŖn(I2)£½0.1 mol£¬ĖłŅŌn(FeI2)£½0.1 mol£¬n(I£)£½0.2 mol”£µ±ĶØČė0.12 mol Cl2¼“0.24 mol ClŹ±£¬I£Č«²æ±»Ńõ»Æ£¬Fe2£«ÓŠ0.04 mol±»Ńõ»Æ£¬ĖłŅŌČÜŅŗÖŠµÄĄė×ÓÖ÷ŅŖÓŠ£ŗFe2£«”¢Fe3£«”¢Cl££¬ĘäĪļÖŹµÄĮæ·Ö±šĪŖ0.06 mol”¢0.04 mol”¢0.24 mol£¬
I2ĪŖ0.1 mol”£
n(I2)”Ćn(FeCl3)”Ćn(FeCl2)£½0.1”Ć0.04”Ć0.06£½5”Ć2”Ć3
·½³ĢŹ½ĪŖ5FeI2£«6Cl2=5I2£«2FeCl3£«3FeCl2”£
£Ø3£©Fe2£« ”« 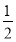 Cl2””””I£”””«””
Cl2””””I£”””«””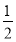 Cl2””I2”«2IO3-”«5Cl2
Cl2””I2”«2IO3-”«5Cl2
0.1 mol 0.05 mol 0.2 mol 0.1 mol x 2x 5x
ÓÉĢāŅāµĆ£ŗ
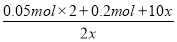 £½8
£½8
x£½0.05 mol
V(Cl2)£½(0.05 mol£«0.1 mol£«5”Į0.05 mol)”Į22.4 L”¤mol£1£½8.96 L”£



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014øßæ¼»Æѧ¶žĀÖø“Ļ°ĻŽŹ±¼Æѵ ×ØĢā14×ŪŗĻŹµŃéÓėĢ½¾æĮ·Ļ°¾ķB£Ø½āĪö°ę£© ĢāŠĶ£ŗŹµŃéĢā
ĪŖĮĖĢ½¾æSO2ÓėNa2O2µÄ·“Ó¦ŹĒ·ńĄąĖĘÓŚCO2ÓėNa2O2µÄ·“Ó¦£¬¼×Ķ¬Ń§Éč¼ĘĮĖČēĶ¼ĖłŹ¾µÄŹµŃé×°ÖĆ£¬»Ų“šĻĀĮŠĪŹĢā£ŗ

£Ø1£©ŅĘæŖĆŽ»Ø£¬½«“ų»šŠĒµÄľĢõ·ÅŌŚCŹŌ¹ÜæŚ£¬Ī“¼ūľĢõø“Č¼£¬¼×Ķ¬Ń§Ņņ“ĖČĻĪŖSO2ÓėNa2O2µÄ·“Ó¦²»Ķ¬ÓŚCO2”£Ēė°“¼×Ķ¬Ń§µÄ¹ŪµćŠ“³ö·“Ó¦µÄ»Æѧ·½³ĢŹ½ ”£
£Ø2£©ŅŅĶ¬Ń§ČĻĪŖĪŽĀŪ·“Ó¦ŌĄķČēŗĪ£¬×īÖÕ¶¼ÓŠO2²śÉś£¬ŅŅĶ¬Ń§µÄĄķÓÉŹĒ ”£°“ÕÕŅŅĶ¬Ń§µÄ¹Ūµć£¬øĆ×°ÖĆŠč×öµÄøĽųŹĒ
ӣ
£Ø3£©¼ŁÉčNa2O2ĶźČ«·“Ó¦£¬·“Ó¦ŗóB×°ÖĆÖŠ¹ĢĢåÉś³ÉĪļæÉÄÜŹĒ£ŗ¢ŁNa2SO3£»¢ŚNa2SO4£»¢ŪNa2SO3ŗĶNa2SO4”£
ĒėÉč¼ĘŹµŃé·½°ø¼ģŃ飬Š“³öŹµŃé²½ÖčŅŌ¼°Ō¤ĘŚĻÖĻóŗĶ½įĀŪ£¬Ķź³ÉĻĀ±ķ£ŗ
ĻŽŃ”ŹŌ¼Į£ŗ2 mol”¤L£1 HClČÜŅŗ£¬1 mol”¤L£1 HNO3ČÜŅŗ£¬1 mol”¤L£1 BaClČÜŅŗ£¬1 mol”¤L£1 Ba£ØNO3£©2ČÜŅŗ£¬0.01 mol”¤L£1 KMnO4ĖįŠŌČÜŅŗ”£
ŹµŃé²½Öč | Ō¤ĘŚĻÖĻóŗĶ½įĀŪ |
²½Öč1£ŗČ”BÖŠµÄÉŁĮæ¹ĢĢåѳʷӌŹŌ¹ÜÖŠ£¬µĪ¼Ó×ćĮæÕōĮóĖ®£¬Čܽā£¬Č»ŗóȔɣĮæ“ż²āŅŗ·Ö±šÖĆÓŚ¢ń”¢¢ņŹŌ¹ÜÖŠ | ¹ĢĢåĶźČ«Čܽā |
²½Öč2£ŗĶł¢ńŹŌ¹ÜÖŠ¼ÓČė £¬ŌŁµĪ¼Ó | £¬ |
ŌņÖ¤Ć÷Éś³ÉĪļÖŠŗ¬Na2SO4 |
|
²½Öč3£ŗĶł¢ņŹŌ¹ÜÖŠ |
|
| Čō £¬ |
ŌņÖ¤Ć÷Éś³ÉĪļÖŠÓŠNa2SO3£»Čō |
|
|
|
ŌņĖµĆ÷Éś³ÉĪļ֊ƻӊNa2SO3”£ |
|
£Ø4£©Éś³ÉĪļÖŠŃĒĮņĖįÄĘŗ¬ĮæµÄ²ā¶Ø£ŗ
¢ŁČ”a gÉś³ÉĪļÅäÖĘ³É100 mLČÜŅŗ£¬Č”10.00 mLøĆČÜŅŗӌ׶ŠĪĘæÖŠ£¬¼ÓČė¼øµĪµķ·ŪČÜŅŗ×÷ÖøŹ¾¼Į£¬ÓĆ0.010 0 mol”¤L£1µāĖ®½ųŠŠµĪ¶Ø£¬µĪ¶ØÖÕµćĻÖĻóĪŖ ”£¼ĒĀ¼Źż¾Ż£¬ÖŲø“µĪ¶Ø2“Ī£¬Ę½¾łĻūŗĵāĖ®20.00 mL”£
¢Ś¼ĘĖć£ŗÉś³ÉĪļÖŠŃĒĮņĖįÄʵÄÖŹĮæ·ÖŹżĪŖ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014øßæ¼»Æѧ¶žĀÖø“Ļ°ĻŽŹ±¼Æѵ ×ØĢā12ÓŠ»śĪļµÄ½į¹¹ÓėŠŌÖŹĮ·Ļ°¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ĻĀĮŠŠšŹöÕżČ·µÄŹĒ£Ø””””£©
A£®ĀČŅŅĻ©µÄ½į¹¹¼ņŹ½ĪŖCH2CHCl
B£®µ°°×ÖŹµÄĖ®½ā²śĪļĪŖ°±»łĖį£¬µķ·ŪµÄĖ®½ā²śĪļĪŖĘĻĢŃĢĒ£¬¾łŹōÓŚ“æ¾»Īļ
C£®ŅŅĻ©ÓėĖ®ŌŚŅ»¶ØĢõ¼žĻĀ·¢Éś¼Ó³É·“Ó¦£¬Ęä²śĪļŹĒ“æ¾»Īļ
D£®ŗ¬5øöĢ¼Ō×ÓµÄÓŠ»śĪļ£¬Ćæøö·Ö×ÓÖŠ×ī¶ąæÉŠĪ³É4øöĢ¼Ģ¼µ„¼ü
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014øßæ¼»Æѧ¶žĀÖø“Ļ°ĻŽŹ±¼Æѵ ×ØĢā10·Ē½šŹōŌŖĖŲµ„ÖŹ¼°»ÆŗĻĪļĮ·Ļ°¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŹµŃéĢā
Ä³ÖŠŃ§»ÆѧŹµŃ銔×éĪŖĮĖÖ¤Ć÷ŗĶ±Č½ĻSO2ŗĶĀČĖ®µÄĘư׊Ō£¬Éč¼ĘĮĖČēĶ¼ĖłŹ¾×°ÖĆ£ŗ
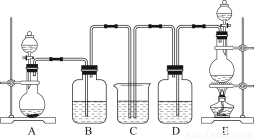
£Ø1£©ŹµŃéŹŅ³£ÓĆ×°ÖĆEÖʱøCl2£¬Öø³öøĆ·“Ó¦ÖŠÅØŃĪĖįĖł±ķĻÖ³öµÄĢŲÖŹÓŠ________________”£
£Ø2£©·“Ó¦æŖŹ¼ŗ󣬷¢ĻÖB”¢DĮ½øö¼ÆĘųĘæÖŠµÄĘ·ŗģČÜŅŗ¶¼ĶŹÉ«£¬Ķ£Ö¹ĶØĘųŗó£¬ÓĆČČĖ®Ō”øųB”¢DĮ½øö¼ÆĘųĘæ¼ÓČČ”£Į½øö¼ÆĘųĘæÖŠµÄĻÖĻó·Ö±šĪŖ£ŗB____________________£»D____________________”£
£Ø3£©×°ÖĆCµÄ×÷ÓĆŹĒ______________________________________________________”£
£Ø4£©øĆŹµŃ銔×éµÄ¼×”¢ŅŅĮ½Ī»Ķ¬Ń§ĄūÓĆÉĻŹöĮ½øö·¢Éś×°ÖĆ°“Ķ¼ĖłŹ¾×°ÖĆ¼ĢŠų½ųŠŠŹµŃé£ŗ
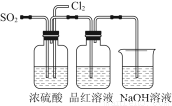
ĶØĘųŅ»¶ĪŹ±¼äŗ󣬼×Ķ¬Ń§ŹµŃé¹ż³ĢÖŠĘ·ŗģČÜŅŗ¼øŗõ²»ĶŹÉ«£¬¶ųŅŅĶ¬Ń§µÄŹµŃéĻÖĻóŹĒĘ·ŗģČÜŅŗĖꏱ¼äµÄĶĘŅʱäµĆŌ½Ą“Ō½Ē³”£
ŹŌøł¾ŻøĆŹµŃé×°ÖĆŗĶĮ½ĆūĶ¬Ń§µÄŹµŃé½į¹ū»Ų“šĪŹĢā£ŗ
¢ŁÖø³öŌŚĘųĢå½ųČėĘ·ŗģČÜŅŗÖ®Ē°£¬ĻČ½«SO2ŗĶCl2ĶعżÅØĮņĖįÖŠµÄÄæµÄ£ŗ________________________________________________________________________________________________________________________________________________”£
¢ŚŹŌ·ÖĪö¼×Ķ¬Ń§ŹµŃé¹ż³ĢÖŠ£¬Ę·ŗģČÜŅŗ²»ĶŹÉ«µÄŌŅņŹĒ____________________________________________________________________________________________________________________________________________________________________________________________________________________”££Ø½įŗĻĄė×Ó·½³ĢŹ½ĖµĆ÷£©
¢ŪÄćČĻĪŖŅŅĶ¬Ń§ŹĒŌõŃł×öµ½ČĆĘ·ŗģČÜŅŗ±äµĆŌ½Ą“Ō½Ē³µÄ£æ
________________________________________________________________________ӣ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014øßæ¼»Æѧ¶žĀÖø“Ļ°ĻŽŹ±¼Æѵ ×ØĢā10·Ē½šŹōŌŖĖŲµ„ÖŹ¼°»ÆŗĻĪļĮ·Ļ°¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
ĻĀĮŠŹµŃéŹĀŹµĖłµĆ³öµÄĻąÓ¦½įĀŪŗĻĄķµÄŹĒ£Ø””””£©
ŹµŃéŹĀŹµ½įĀŪ
A£®Cl2µÄĖ®ČÜŅŗæÉŅŌµ¼µēCl2ŹĒµē½āÖŹ
B£®½«Č¼×ŵÄĆ¾ĢõÉģČėŹ¢ÓŠCO2µÄ¼ÆĘųĘæÖŠ¼ĢŠųČ¼ÉÕ»¹ŌŠŌ£ŗMg>C
C£®SO2æÉŅŌŹ¹ĖįŠŌKMnO4ČÜŅŗĶŹÉ«SO2¾ßÓŠĘư׊Ō
D£®½«Ģś·Ū·ÅČėĻ”HNO3ÖŠ³ä·Ö·“Ó¦ŗ󣬵ĪČėKSCNČÜŅŗĪŽĆ÷ĻŌĻÖĻóĻ”HNO3²»Äܽ«FeŃõ»Æ³ÉFe3£«
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014øßæ¼»Æѧ¶žĀÖ×ØĢāĶ»ĘĘ ×ØĢāĖÄŃõ»Æ»¹Ō·“Ó¦Į·Ļ°¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
½«Ņ»¶ØĮæµÄFeŗĶFe2O3µÄ»ģŗĻĪļ·ÅČė250 mL”¢1.8 mol”¤L£1µÄHNO3ČÜŅŗÖŠ£¬µ±¹ĢĢå»ģŗĻĪļĶźČ«Čܽāŗó£¬ŌŚ±ź×¼×“æöĻĀÉś³É1.12 L NO(HNO3µÄ»¹Ō²śĪļ½ö“ĖŅ»ÖÖ)£¬ŌŁĻņ·“Ó¦ŗóµÄČÜŅŗÖŠ¼ÓČė1.0 mol”¤L£1 NaOHČÜŅŗ£¬ČōŅŖŹ¹ĢśŌŖĖŲĶźČ«³Įµķ£¬Ėł¼ÓČėµÄNaOHČÜŅŗµÄĢå»ż×īÉŁÓ¦ĪŖ(””””)
A£®300 mL B£®400 mL C£®450 mL D£®500 mL
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014øßæ¼»Æѧ¶žĀÖ×ØĢāĶ»ĘĘ ×ØĢāŹ®µē½āÖŹČÜŅŗĮ·Ļ°¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
Ė®ŹĒÉśĆüµÄŌ“ČŖ”¢¹¤ŅµµÄŃŖŅŗ”¢³ĒŹŠµÄĆüĀö”£ŅŖ±£»¤ŗĆŗÓĮ÷£¬ŗÓĖ®ŹĒÖ÷ŅŖµÄŅūÓĆĖ®Ō“£¬ĪŪČ¾ĪļĶعżŅūÓĆĖ®æÉÖ±½Ó¶¾ŗ¦ČĖĢ壬Ņ²æÉĶعżŹ³ĪļĮ“ŗĶ¹ąøČÅ©Ģļ¼ä½ÓĪ£¼°½”æµ”£
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
(1)“æĖ®ŌŚ100 ”ꏱ£¬pH£½6£¬øĆĪĀ¶ČĻĀ1 mol”¤L£1µÄNaOHČÜŅŗÖŠ£¬ÓÉĖ®µēĄė³öµÄ
c(OH£)£½________ mol”¤L£1”£
(2)25 ”ꏱ£¬ĻņĖ®µÄµēĄėĘ½ŗāĢåĻµÖŠ¼ÓČėÉŁĮæĢ¼ĖįÄĘ¹ĢĢ壬µĆµ½pHĪŖ11µÄČÜŅŗ£¬ĘäĖ®½ā·½³ĢŹ½ĪŖ__________£¬ÓÉĖ®µēĄė³öµÄc(OH£)£½__________ mol”¤L£1”£
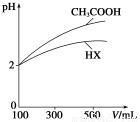
(3)Ģå»ż¾łĪŖ100 mL”¢pH¾łĪŖ2µÄCH3COOHÓėŅ»ŌŖĖįHX£¬¼ÓĖ®Ļ”ŹĶ¹ż³ĢÖŠpHÓėČÜŅŗĢå»żµÄ¹ŲĻµČēÉĻĶ¼ĖłŹ¾£¬ŌņHXµÄµēĄėĘ½ŗā³£Źż________(Ģī”°“óÓŚ”±”¢”°Š”ÓŚ”±»ņ”°µČÓŚ”±)CH3COOHµÄµēĄėĘ½ŗā³£Źż”£ĄķÓÉŹĒ__________________________________”£
(4)µēĄėĘ½ŗā³£ŹżŹĒŗāĮæČõµē½āÖŹµēĄė³Ģ¶ČĒæČõµÄĪļĄķĮ攣ŅŃÖŖ£ŗ
»ÆѧŹ½ | µēĄė³£Źż(25 ”ę) |
HCN | K£½4.9”Į10£10 |
CH3COOH | K£½1.8”Į10£5 |
H2CO3 | K1£½4.3”Į10£7”¢K2£½5.6”Į10£11 |
¢Ł25 ”ꏱ£¬ÓŠµČÅØ¶ČµÄNaCNČÜŅŗ”¢Na2CO3ČÜŅŗŗĶCH3COONaČÜŅŗ£¬ČżČÜŅŗµÄpHÓɓ󵽊”µÄĖ³ŠņĪŖ____________________________”£
¢ŚĻņNaCNČÜŅŗÖŠĶØČėÉŁĮæµÄCO2£¬·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ__________________
(5)25 ”ꏱ£¬ŌŚCH3COOHÓėCH3COONaµÄ»ģŗĻČÜŅŗÖŠ£¬Čō²āµĆpH£½6£¬ŌņČÜŅŗÖŠc(CH3COO£)£c(Na£«)£½________ mol”¤L£1(Ģī¾«Č·Öµ)£¬c(CH3COO£)/c(CH3COOH)£½________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014øßæ¼»Æѧ¶žĀÖ×ØĢāĶ»ĘĘ ×ØĢāŹ®Ėij£¼ūÓŠ»śĪļ¼°ĘäÓ¦ÓĆĮ·Ļ°¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŃ”ŌńĢā
Ņŗ»ÆĘųµÄÖ÷ŅŖ³É·ÖŹĒ±ūĶ飬ĻĀĮŠÓŠ¹Ų±ūĶéµÄŠšŹö²»ÕżČ·µÄŹĒ(””””)
A£®ŹĒÖ±Į“Ģž£¬µ«·Ö×ÓÖŠ3øöĢ¼Ō×Ó²»ŌŚŅ»ĢõÖ±ĻßÉĻ
B£®ŌŚ¹āÕÕĢõ¼žĻĀÄܹ»ÓėĀČĘų·¢ÉśČ”“ś·“Ó¦
C£®±ūĶé±Č¶”ĶéŅ×Ņŗ»Æ
D£®1 mol±ūĶéĶźČ«Č¼ÉÕĻūŗÄ5 mol O2
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014øßæ¼»Æѧ¶žĀÖ×ØĢāĶ»ĘĘ ×ØĢāŹ®ĘßÓŠ»ś»Æѧ»ł“”Į·Ļ°¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
ĀČßĮøńĄ×(clopidogrel,1)ŹĒŅ»ÖÖÓĆÓŚŅÖÖĘŃŖŠ”°å¾Ū¼ÆµÄŅ©Īļ£¬øł¾ŻŌĮĻµÄ²»Ķ¬£¬øĆŅ©ĪļµÄŗĻ³ÉĀ·ĻßĶس£ÓŠĮ½Ģõ£¬ĘäÖŠŅŌ2?Āȱ½¼×Č©ĪŖŌĮĻµÄŗĻ³ÉĀ·ĻßČēĻĀ£ŗ
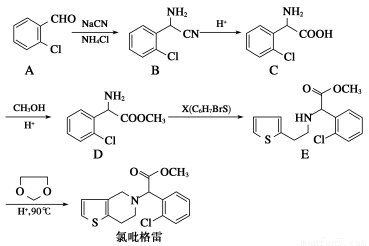
(1)·Ö×ÓDÖŠµÄ¹ŁÄÜĶÅĆū³ĘĪŖ________________”£XµÄ½į¹¹¼ņŹ½ĪŖ____________”£
(2)·Ö×ÓCæÉŌŚŅ»¶ØĢõ¼žĻĀ·“Ӧɜ³ÉŅ»ÖÖ²śĪļ£¬øĆ²śĪļ·Ö×ÓÖŠŗ¬ÓŠ3øöĮłŌŖ»·£¬Š“³öøĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½______________________________________
(3)D”śEµÄ·“Ó¦ĄąŠĶŹĒ________·“Ó¦”£
(4)Š“³öAŹōÓŚ·¼Ļć×å»ÆŗĻĪļµÄĖłÓŠĶ¬·ÖŅģ¹¹ĢåµÄ½į¹¹¼ņŹ½£ŗ__________”£
(5)ŅŃÖŖ£ŗCO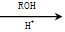 COROH
COROH COROR
COROR
ŌņÓÉŅŅ“¼”¢¼×“¼ĪŖÓŠ»śŌĮĻÖʱø»ÆŗĻĪļ £¬ŠčŅŖ¾ĄśµÄ·“Ó¦ĄąŠĶÓŠ__________(ĢīŠ“±ąŗÅ)”£¢Ł¼Ó³É·“Ó¦””¢ŚĻūČ„·“Ó¦””¢ŪČ”“ś·“Ó¦””¢ÜŃõ»Æ·“Ó¦””¢Ż»¹Ō·“Ó¦£¬Š“³öÖʱø»ÆŗĻĪļ
£¬ŠčŅŖ¾ĄśµÄ·“Ó¦ĄąŠĶÓŠ__________(ĢīŠ“±ąŗÅ)”£¢Ł¼Ó³É·“Ó¦””¢ŚĻūČ„·“Ó¦””¢ŪČ”“ś·“Ó¦””¢ÜŃõ»Æ·“Ó¦””¢Ż»¹Ō·“Ó¦£¬Š“³öÖʱø»ÆŗĻĪļ µÄ×īŗóŅ»²½·“Ó¦_______________________________________________”£
µÄ×īŗóŅ»²½·“Ó¦_______________________________________________”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com