
��18�֣�B��һ����Ԫ��״�������˴Ź�������ֻ��һ���壻H�ĺ˴Ź���������3���壬�����֮��Ϊ2��2��1��I��һ�ֺϳ�����֬����Ҫԭ�ϡ���֪R���������й�����ת����ϵ����Ϣ���£�
�밴Ҫ��ش��������⣺
��1��A�IJ�����������Ϊ _______________���۷�Ӧ����Ϊ____________________��
��2��B�Ľṹ��ʽΪ __________________��H��ϵͳ������Ӧ����Ϊ________________��
��3��д�����з�Ӧ�Ļ�ѧ����ʽ��
��____________________________________________________________________��
�� ____________________________________________________________________��
��4��д��˳���Ľṹ��ʽ_____________________________��˳�����Ըߣ��ͺ��Ժã����л������������͵��£��ϳ�·�����£�
���ȹ���(CH3)2SiCl2 ��������� (CH3)2Si(OH)2
��������� (CH3)2Si(OH)2 ����
����
��д�����۵Ļ�ѧ����ʽ__________________________________________________________��
��5���л���J��F��Ϊͬ���칹�壬�Ҿ�����ͬ�Ĺ������������Ŀ��д�����з���������J�Ľṹ��ʽ��������F����_________________________________________________________________��
��1�� -Br(��ԭ��)��1�֣� ��ԭ��Ӧ��1�֣���2��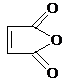 ��2�֣�1��4-��������2�֣�
��2�֣�1��4-��������2�֣�
��3���� 2HOOCCH2CHOHCOOH 2H2O+
2H2O+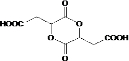 ��2�֣�
��2�֣�
��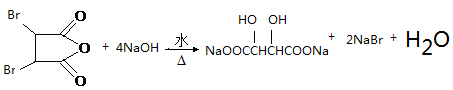 (2�֣�
(2�֣�
��4��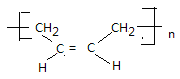 ��2�֣�
��2�֣�
n(CH3)2Si(OH)2��(n-1)H2O + ��2�֣�
��2�֣�
��5�� ��2�� 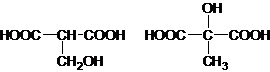
�������������B��һ����Ԫ��״�������˴Ź�������ֻ��һ���壬�������巢���ӳɷ�Ӧ��˵�������к���̼̼˫����ӦΪ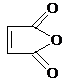 ��Bˮ������C��C�Ľṹ��ʽΪHOOCCH��CHCOOH��C��ˮ������Ӧ����F������C��F��Ħ��������֪��C��ˮ�����ӳɷ�Ӧ����F������F�Ľṹ��ʽΪ��HOOCCH2CH��OH��COOH��C�����������ӳɷ�Ӧ����D��D�Ľṹ��ʽΪHOOCCH2CH2COOH����������Ϣ֪��H�Ľṹ��ʽΪHOCH2CH2CH2CH2OH��IΪCH2��CHCH��CH2��GΪ
��Bˮ������C��C�Ľṹ��ʽΪHOOCCH��CHCOOH��C��ˮ������Ӧ����F������C��F��Ħ��������֪��C��ˮ�����ӳɷ�Ӧ����F������F�Ľṹ��ʽΪ��HOOCCH2CH��OH��COOH��C�����������ӳɷ�Ӧ����D��D�Ľṹ��ʽΪHOOCCH2CH2COOH����������Ϣ֪��H�Ľṹ��ʽΪHOCH2CH2CH2CH2OH��IΪCH2��CHCH��CH2��GΪ ��AΪ
��AΪ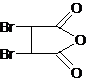 ��EΪNaOOCCHOHCHOHCOONa����
��EΪNaOOCCHOHCHOHCOONa����
��1��AΪ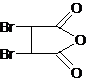 ������������Ϊ-Br����Ӧ�����Ȼ����ǻ�������ȥ���ķ�Ӧ������ǻ�ԭ��Ӧ��
������������Ϊ-Br����Ӧ�����Ȼ����ǻ�������ȥ���ķ�Ӧ������ǻ�ԭ��Ӧ��
��2�������Ϸ�����֪BΪ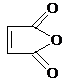 ��H�Ľṹ��ʽΪHOCH2CH2CH2CH2OH��Ϊ1��4-��������
��H�Ľṹ��ʽΪHOCH2CH2CH2CH2OH��Ϊ1��4-��������
��3����Ӧ�ߵĻ�ѧ����ʽΪ2HOOCCH2CHOHCOOH 2H2O+
2H2O+ ����Ӧ����±������ˮ�ⷴӦ����Ӧ�Ļ�ѧ����ʽΪ
����Ӧ����±������ˮ�ⷴӦ����Ӧ�Ļ�ѧ����ʽΪ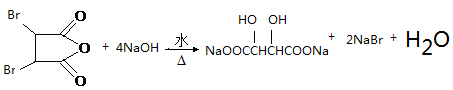 ��
��
��4��˳��1,3������ϩ�����Ӿ۷�Ӧ������˳��ϩ���ṹ��ʽΪ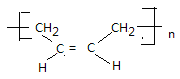 ���������������2���ǻ����������۷�Ӧ���ɹ�����Ӧ�Ļ�ѧ����ʽΪn(CH3)2Si(OH)2��(n-1)H2O +
���������������2���ǻ����������۷�Ӧ���ɹ�����Ӧ�Ļ�ѧ����ʽΪn(CH3)2Si(OH)2��(n-1)H2O + ��
��
��5��F�Ľṹ��ʽΪ��HOOCCH2CH��OH��COOH����Ӧ�ĺ�����ͬ�����ŵ�ͬ���칹����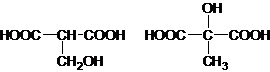 ��
��
���㣺�����л����ƶϡ������š��л���Ӧ���͡�������ͬ���칹���жϡ��Լ�����ʽ��д�ĵ�


 ������ȫ�̼����ĩ���100��ϵ�д�
������ȫ�̼����ĩ���100��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
Ϊ�ⶨij�л�������A�Ľṹ����������ʵ�飺
(һ)����ʽ��ȷ����
��1�����л���A�����������г��ȼ�գ�ʵ���ã�����5.4 g H2O��8.8 g CO2����������6.72 L(��״����)����������и�Ԫ�ص�ԭ�Ӹ�������________��
��2���������Dzⶨ���л����������Է����������õ���ͼ1��ʾ����ͼ��������Է�������Ϊ________�������ʵķ���ʽ��________��
��3�����ݼۼ����ۣ�Ԥ��A�Ŀ��ܽṹ��д���ṹ��ʽ______________________��
(��)�ṹʽ��ȷ����
��4���˴Ź��������ܶ��л�������в�ͬλ�õ���ԭ�Ӹ�����ͬ�ķ�ֵ(�ź�)�����ݷ�ֵ(�ź�)����ȷ����������ԭ�ӵ��������Ŀ�����磺���ȼ���(Cl��CH2��O��CH3)��������ԭ����ͼ2�����ⶨ���л���A�ĺ˴Ź�������ʾ��ͼ��ͼ3����A�Ľṹ��ʽΪ________��
ͼ1 ͼ2 ͼ3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(8��)�л���A��B��C��D���������ʡ�
��1������֧���Ļ�����A�ķ���ʽΪC4H6O2��A����ʹBr2�����Ȼ�̼��Һ��ɫ��1molA
��1mol NaHCO3����ȫ��Ӧ����A�Ľṹ��ʽ�� ��
д����A������ͬ�����ŵ�A������ͬ���칹��Ľṹ��ʽ
��
��2��������B����C��H��O����Ԫ�أ�������Ϊ60������̼����������Ϊ60%�������������Ϊ13.33%��B�ڴ���Cu�������±�������C��C�ܷ���������Ӧ����B�Ľṹ��ʽ�� ��
��3��D��NaOHˮ��Һ�м��ȷ�Ӧ��������A�����κ�B����Ӧ��Ӧ�Ļ�ѧ����ʽ��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��10�֣����ס�������ס��˴Ź�������������ѳ�Ϊ�о��л������Ҫ��ɲ��֡�
(1) ���������У���˴Ź��������и����ķ�ֵֻ��һ������
A��CH3CH3 B��CH3COOH C��CH3COOCH3 D��CH3OCH3
(2)������A��B�ķ���ʽ����C2H4Br2, A�ĺ˴Ź�������ͼ��ͼ��ʾ����A�Ľṹ��ʽΪ ��
��Ԥ��B�ĺ˴Ź���������Ӧ����_______���壨�źţ���
(3)��������и�ԭ����Ŀ��Ϊ��N(C)��N(H)��N(O)=1��2��1���Ի�����C�����������ɵõ���ͼ��ʾ������ͼ���ɵó������ʽΪ ����������к�����������ɵó��������Ϊ������������д���ƣ���ȷ��������C�Ĺ�����ʱ������Ϊ ����С���û�С�����Ҫ���к����������д��������C����������������ͭ��Ӧ�Ļ�ѧ����ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(10��)�����沽���� �ϳ�
�ϳ� (�����Լ��ͷ�Ӧ��������ȥ)��
(�����Լ��ͷ�Ӧ��������ȥ)��
��ش��������⣺
(1)�ֱ�д��B��D�Ľṹ��ʽ��B_________��D_________��
(2)��Ӧ�١�����������ȥ��Ӧ���� ��(���������)
(3)��������Ǣޡ��߷�Ӧ�����ڷ�Ӧ�ݣ��õ���E���ܵĽṹ��ʽΪ��
(4)��д��C��D��Ӧ�Ļ�ѧ����ʽ(ע����Ӧ����)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����8�֣�
�����л��������������ԡ�
���������գ�
(1)���ӡ����״��������ᡢ̼���������ǿ������˳��Ϊ��_____________________________________�������� ������______������ͬ�����ԡ��������ԡ������ԡ���
������______������ͬ�����ԡ��������ԡ������ԡ���
(2)�����������ʵ�����Է�����Һ��ijһ���Һ�����ӡ����״���������ͱ����������ʣ�����뷽������ͼ��
��֪�����״��������ᡢ��������ˮ��
A��B��C�ֱ��ǣ�A_______ B_________ C_____________
���������У������Ƚ������������������������ʷֿ�����Ӧ����________��
(3)���� ת��Ϊ
ת��Ϊ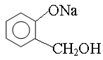 ����Ӧ����____________��
����Ӧ����____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������ABS��֬���ṹ��ʽ���£��ϳ�ʱ�������ֵ��塣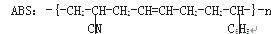
ʽ��- C6H5�DZ����������ֵ���Ľṹ��ʽ�ֱ��ǣ�
�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����15�֣�ҩ��Z����������������ϵͳ�Ժ���Ǵ��ȣ�����X��1,4-������ͪ���Ҷ�����ͪ����Y�������ᣩΪԭ�Ϻϳɣ�����ͼ��
��1��������X���� �ֻ�ѧ������ͬ����ԭ�ӡ�
��2������˵����ȷ���� ��
| A��X�Ƿ��㻯���� | B��Ni����Y����5molH2�ӳ� |
| C��Z�ܷ����ӳɡ�ȡ������ȥ��Ӧ | D��1mol Z������5mol NaOH��Ӧ |
 �����Ӽ���ˮ���ã�һ�������£�M����1����OH����ȥ��Ӧ�õ��ȶ�������N������ʽΪC6H8O2������N�Ľṹ��ʽΪ ����֪ϩ��ʽ���ȶ����ᷢ���������ţ����磺
�����Ӽ���ˮ���ã�һ�������£�M����1����OH����ȥ��Ӧ�õ��ȶ�������N������ʽΪC6H8O2������N�Ľṹ��ʽΪ ����֪ϩ��ʽ���ȶ����ᷢ���������ţ����磺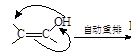
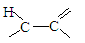 ����
����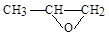 ���������Ʒ�Ӧ�ٵķ�Ӧ����������Ľṹ��ʽΪ ��дһ�֣���Y��ͬ���칹��ܶ��֣������б�����������������ȡ�������ҷ��ǻ���λ�ú���Ŀ�����䣩����������ͬ���칹���� �֡�
���������Ʒ�Ӧ�ٵķ�Ӧ����������Ľṹ��ʽΪ ��дһ�֣���Y��ͬ���칹��ܶ��֣������б�����������������ȡ�������ҷ��ǻ���λ�ú���Ŀ�����䣩����������ͬ���칹���� �֡��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij�о�С���Լױ�Ϊ��Ҫԭ�ϣ���������·�ߺϳ�ҽҩ�м���F��Y��
��֪����)  ������
������ ��
��
��ش��������⣺
��1��д��Y�к��������ŵ����� ��
��2�������й�F��˵����ȷ���� ��
| A������ʽ��C7H7NO2Br | B�����������ᷴӦ������NaOH��Һ��Ӧ |
| C���ܷ���������Ӧ | D��1 mol F����������2 mol NaOH |
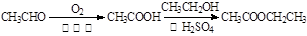
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com