
��Ҫ����գ�
��1����������֪�Ļ������У����������ǵ� ��Ԫ���γɵĻ����
��2����ϵͳ����������������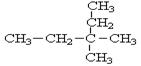 ��
��
��3��ijʵ��С�������뱽�ͱ��ӣ����������м����Թ�����Ũ����������Һ���÷�Ӧ�Ļ�ѧ����ʽΪ ���ٽ���Ӧ��Ļ����ת�Ƶ���Һ©���У������ã�Ȼ���Һ���ӷ�Һ©���ų��²�Һ�壬�����������ᣬ�ٽ���ֳ����ݣ���һ����Һ�м������Ȼ�����Һ�����ֵ������� ������һ����Һ�м���Ũ��ˮ����Ӧ�Ļ�ѧ����ʽΪ  ���ӷ�Һ©���Ͽڵ�����Һ���� ��
���ӷ�Һ©���Ͽڵ�����Һ���� ��
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(8��)����S2-��SO32-��NH4+��Al3+����Na+��SO42-��Fe3+��HCO3-��Cl-�����ӣ��밴Ҫ����գ�
��1����ˮ��Һ�У�������ˮ��ʼ��Ե��� ��
��2����ˮ��Һ�У�������ˮ������Ե��� ��
��3�����������Խ�ǿ����Һ��������ڣ������ڼ��Խ�ǿ����Һ��������ڵ������� ��
��4���Ȳ��������Խ�ǿ����Һ��������ڣ��ֲ����ڼ��Խ�ǿ����Һ��������ڵ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014��ӱ�ʡ�߶�9���¿���ѧ�Ծ��������棩 ���ͣ������
��8�֣�����S2-��SO32-��NH4+��Al3+��HPO42-��Na+��SO42-��AlO2-��Fe3+��HCO3-��Cl-�����ӣ��밴Ҫ����գ�
��1����ˮ��Һ�У�������ˮ��ʼ��Ե��� ��
��2����ˮ��Һ�У�������ˮ������Ե��� ��
��3�����������Խ�ǿ����Һ��������ڣ������ڼ��Խ�ǿ����Һ��������ڵ������� ��
��4���Ȳ��������Խ�ǿ����Һ��������ڣ��ֲ����ڼ��Խ�ǿ����Һ��������ڵ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013�����ʡ�߶�3�¿��Ի�ѧ�Ծ��������棩 ���ͣ������
(8��)����S2-��SO32-��NH4+��Al3+����Na+��SO42-��Fe3+��HCO3-��Cl-�����ӣ��밴Ҫ����գ�
��1����ˮ��Һ�У�������ˮ��ʼ��Ե��� ��
��2����ˮ��Һ�У�������ˮ������Ե��� ��
��3�����������Խ�ǿ����Һ��������ڣ������ڼ��Խ�ǿ����Һ��������ڵ������� ��
��4���Ȳ��������Խ�ǿ����Һ��������ڣ��ֲ����ڼ��Խ�ǿ����Һ��������ڵ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010-2011ѧ��㶫ʡ�������Ĵ��ۺϲ������ۻ�ѧ���� ���ͣ������
(8��)����S2-��SO32-��NH4+��Al3+����Na+��SO42-��Fe3+��HCO3-��Cl-�����ӣ��밴Ҫ����գ�
��1����ˮ��Һ�У�������ˮ��ʼ��Ե��� ��
��2����ˮ��Һ�У�������ˮ������Ե��� ��
��3�����������Խ�ǿ����Һ��������ڣ������ڼ��Խ�ǿ����Һ��������ڵ������� ��
��4���Ȳ��������Խ�ǿ����Һ��������ڣ��ֲ����ڼ��Խ�ǿ����Һ��������ڵ������� ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com