
��10�֣�2006����������ӵ���ܲ�������25��ֻ��﮵���������Բ��������Ľ�����Դ���������൱��ġ��������ӵ�ػ��վ�����Ҫ���壬������Ҫ�ص���յ����������ϣ�����Ҫ�ɷ�Ϊ����ﮣ�LiCoO2����������Ȳ�ڣ�һ��̿�ڣ��������Լ��л�ճ�Ӽ���ij���չ����������£�
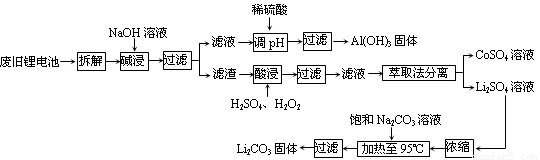
��1���������ջ��յ��IJ����� ��
��2���Ͼɵ�ؿ������ڷŵ粻��ȫ��������ԭ��̬��ﮣ�Ϊ�˰�ȫ�Բ�����Ҫ����_______________��
��3�����ʱ��Ҫ��Ӧ�����ӷ���ʽΪ_____________________��
��4�����ʱ��Ӧ�Ļ�ѧ����ʽΪ___________��������������H2SO4��H2O2�Ļ��ҺҲ�ܴﵽ�ܽ��Ŀ�ģ�������֮����_______________��
��5������Li2CO3�Ļ�ѧ��Ӧ����ʽΪ____________����֪Li2CO3��ˮ�е��ܽ�������¶����߶���С�����һ������ʱӦ____________��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��10�֣�2006����������ӵ���ܲ�������25��ֻ��﮵���������Բ��������Ľ�����Դ���������൱��ġ��������ӵ�ػ��վ�����Ҫ���壬������Ҫ�ص���յ����������ϣ�����Ҫ�ɷ�Ϊ����ﮣ�LiCoO2����������Ȳ�ڣ�һ��̿�ڣ��������Լ��л�ճ�Ӽ���ij���չ����������£�
��1���������ջ��յ��IJ����� ��
��2���Ͼɵ�ؿ������ڷŵ粻��ȫ��������ԭ��̬��ﮣ�Ϊ�˰�ȫ�Բ�����Ҫ����_______________��
��3�����ʱ��Ҫ��Ӧ�����ӷ���ʽΪ_____________________��
��4�����ʱ��Ӧ�Ļ�ѧ����ʽΪ___________��������������H2SO4��H2O2�Ļ��ҺҲ�ܴﵽ�ܽ��Ŀ�ģ�������֮����_______________��
��5������Li2CO3�Ļ�ѧ��Ӧ����ʽΪ____________����֪Li2CO3��ˮ�е��ܽ�������¶����߶���С�����һ������ʱӦ____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010�꽭��ʡ�������Ĵ�ģ�⿼�Ի�ѧ���� ���ͣ�ʵ����
��10�֣�2006����������ӵ���ܲ�������25��ֻ��﮵���������Բ��������Ľ�����Դ���������൱��ġ��������ӵ�ػ��վ�����Ҫ���壬������Ҫ�ص���յ����������ϣ�����Ҫ�ɷ�Ϊ����ﮣ�LiCoO2����������Ȳ�ڣ�һ��̿�ڣ��������Լ��л�ճ�Ӽ���ij���չ����������£�

��1���������ջ��յ��IJ����� ��
��2���Ͼɵ�ؿ������ڷŵ粻��ȫ��������ԭ��̬��ﮣ�Ϊ�˰�ȫ�Բ�����Ҫ����_______________��
��3�����ʱ��Ҫ��Ӧ�����ӷ���ʽΪ_____________________��
��4�����ʱ��Ӧ�Ļ�ѧ����ʽΪ___________��������������H2SO4��H2O2�Ļ��ҺҲ�ܴﵽ�ܽ��Ŀ�ģ�������֮����_______________��
��5������Li2CO3�Ļ�ѧ��Ӧ����ʽΪ____________����֪Li2CO3��ˮ�е��ܽ�������¶����߶���С�����һ������ʱӦ____________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com