
A��B��C��D��EΪԪ�����ڱ��ж�����Ԫ���γɵ��������ӣ�A��B��C��D�������ӵĵ���������E�ĵ�������8��
��1��A��B��ϳɵĻ�������м������Ӽ����й��ۼ���A��C��ϳɵĻ�������������ˮ�������ڼ�ˮ��Һ����ɫ����Һ��CԪ�������ڱ��е�λ��Ϊ _ �������ˮ��Һ��Ӧ�����ӷ���ʽΪ__________________________________��
��2��B��E��ϳɻ������������������֮��Ϊ2��1��A��D��ϳɻ����ﶡ���������Ӧ������ɫ��ζ�����壬����ĵ���ʽΪ�� _ ��1molD���������� _ mol����
��3����һ��Һ̬���⻯�����죬�������E�ĵ�������ͬ���ǡ������ߺš��ɴ�����ʱʹ�õĸ���ȼ��֮һ���ṹ�������ָ÷��ӽṹ��ֻ�е������백���ƣ�����ĽṹʽΪ ___��
��1����3���ڢ�A Al(OH)3+OH-=AlO2-+2H2O
��2�� 11mol
11mol
��3��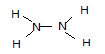
���������������1����������Ϊ10�����ӵ������ӣ�N3-��O2-��F-��OH-��NH2-�� �����ӣ�Na+��Mg2+��Al3+��NH4+��H3O+����������Ϊ18�����ӵ������ӣ�S2-��O22-��Cl-��HS-�� �����ӣ�K+��Ca2+��N2H5+��N2H62+�����������⣬A��B��C��D����10e-���ӣ�EΪ18e-���ӣ�A��B�γɵĻ�������м������Ӽ����й��ۼ���10e-�������γɵĻ�����������Ӽ����й��ۼ�������Ϊ���λ��������A��C��ϳɵĻ�������������ˮ��10e-�����н�ϳ�������ˮ�Ļ�������Mg��OH��2��Al��OH��3���ٸ��������ڼ�ˮ��Һ����ɫ����Һ��ȷ����ΪAl��OH��3����Ϊǿ��NaOH����AΪOH-��BΪNa+��CΪAl3+����C�����ڱ��е�λ�õ������ڣ���A�塣Al��OH��3��NaOH��Ӧ����ʽΪAl��OH��3+NaOH�TNaAlO2+2H2O����д���ӷ���ʽΪAl��OH��3+OH-=AlO2-+2H2O����2���������⣬A��OH-��D��H3O+����ϳɻ����ﶡ����˶�ΪH2O��B��Na+��B��E�γɵĻ������������������֮��Ϊ2��1��E����ΪS2-��O22-������Ϊ�������Ӧ������ɫ��ζ���壬���Ա�ֻ��ΪNa2O2������ʽ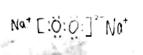 1mol H3O+��11mol���ӡ�
1mol H3O+��11mol���ӡ�
��3���������⣬��������ΪҺ̬���⻯���ͬʱҲΪ18���ӣ�Nԭ�Ӻ�����7�����ӣ�Hԭ�Ӻ�����1�����ӣ���˻���������Ӧ��2��Nԭ�Ӻ�4��Hԭ�ӣ��ʻ�ѧʽΪN2H4���ṹ�����õ��÷�����ֻ���е��������Ե�ԭ�Ӻ͵�ԭ�ӡ���ԭ�Ӻ���ԭ��֮�䶼���γ�һ�Թ��õ��Ӷԣ��ʵ���ʽΪ ���ṹʽ
���ṹʽ ��
��
���㣺���⿼��10���Ӻ�18������������ʽ���ṹʾ��ͼ�����ӷ���ʽ


 �ο�����������100��ϵ�д�
�ο�����������100��ϵ�д� �Űٷֿ�ʱ����ϵ�д�
�Űٷֿ�ʱ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
A��B��D��EΪԭ��������������Ķ�����Ԫ�ء�A��B��ɵ��������ʹʪ��ĺ�ɫʯ����ֽ������A��D����ͬһ���壬E��������������������Ӳ�����
��1��B��Ԫ�����ڱ���λ���� ������Ԫ�صļ����ӣ��뾶������ �������ӷ��ţ�������D��¶�ڿ��������ղ����� ��
��2��B��һ��������BO2��ˮ��Ӧ�Ļ�ѧ����ʽΪ�� ��
��3����B������������ˮ���ﷴӦ�����ҡ���0.1 mol��L-1����Һ�У���������Ũ���ɴ�С��˳���� ��
��4��25��ʱ��E������������ˮ����KSP=1.0��10-34��ʹ��0.1 mol��L-1E���ӵ���Һ��ʼ����������pHΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
A��B��C��D��E������ԭ���������ε����Ķ�����Ԫ�أ���֪������ֻ��һ���ǽ���Ԫ�أ�A��DԪ�ص�ԭ��������������ͬ��C��Eͬ���壬��EԪ��ԭ����������CԪ��ԭ����������2����B�������������ǵ��Ӳ�����������C��A���γ����ֳ�����Һ̬��������ң�����Է��������ұȼ״�16��
��1��EԪ�ص�����Ϊ�� ��D��ԭ�ӽṹʾ��ͼΪ ��
��2�����������к��еĻ�ѧ���� ������Թ��ۼ����Ǽ��Թ��ۼ�������
��3��A������B��C�γɵĻ�����ɺϳ�һ��������������ȼ�ϼ״���
��֪CO��g��+1/2O2��g��=CO2��g��
��H=��283 kJ/mol
H2��g��+1/2O2��g��===H2O��g�� ��H=��242 kJ/mol
CH3OH��g��+3/2O2��g��=CO2��g��+2H2O��g��
��H=��651 kJ/mol
д��A����ѡ������ϳɼ״����Ȼ�ѧ����ʽ�� ��
��4��EC2��C��D�γɵĻ������������ԭ��Ӧ�Ļ�ѧ����ʽΪ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������Ԫ��A��B��C��D��E��ԭ����������������Ԫ��������Ϣ�����
(1)Dԭ�ӽṹʾ��ͼΪ _______ ��A�����ڱ��е�λ��Ϊ ��
(2)B��C�γɵĻ�����ĵ���ʽΪ ��
(3)B��D�����ӵİ뾶��СΪ ��
(4)Ԫ�طǽ�����ǿ���Ƚ��кܶ��������B��E�ķǽ�����ǿ�����о������в����е��� (�����)��
a.�Ƚ����ֵ��ʵ���ɫ
b.�Ƚ����ֵ�����H2���ϵ����׳̶�
c.������Ԫ�������ڱ��е�λ��
d.�Ƚ϶�Ӧ�����ӵĻ�ԭ��
e.�Ƚ�����������Ӧˮ���������
(5)EԪ��������������Ԫ���е�һ��Ԫ���γɹ��ۻ���������е�ԭ�Ӹ�����Ϊ1��3����Է�������Ϊ120.5��������ʵĻ�ѧʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��14�֣�A��B��C��D��E��F��Ϊ������Ԫ�أ�ԭ���������ε�����AԪ��ԭ�Ӻ��������ӡ�BԪ��ԭ�������������Ǵ�����������2����CԪ���ǵؿ��Ǻ�������Ԫ�ء�D�Ƕ�����Ԫ���н�������ǿ��Ԫ�ء�E��F��λ�����ڣ�F������������ˮ����Ϊ��ǿ���ᡣ
��1���ƶ�B��Ԫ�����ڱ��е�λ�ã� ��
��2��д��A��C�γɵ�10���ӵ������ӻ�ѧʽ�� �����ö�Ӧ�Ļ�ѧ������գ���ͬ��
��3��E��F����Ԫ���зǽ����Խ�ǿ���� ���õ���ʽ��ʾD2C�γɹ���
��4��D��E�γɵ����ε�ˮ��Һ�У������ӵ�Ũ�ȴ�С˳��Ϊ�� ���Ӵ�С���У���
��5����������1molAԪ�صĵ�����CԪ�صĵ��ʻ��ϣ��ų�286kJ��������д����Ӧ���Ȼ�ѧ����ʽ��
��6������A��C��ԭ�Ӹ�����1:1��ɵĻ������֪����Һ��ʹ���Ը��������Һ��ɫ��������0.5mol����Һ�μӵ�100mL 2mol/L���Ը��������Һ�У���Һ��ɫǡ����ȥ���÷�Ӧ�����ӷ���ʽΪ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
X��һ����Ҫ�Ĺ�ҵԭ�ϣ� X��160��ֽ��Ʊ�������Ͱ�������Ӧ����ʽΪ��X  HCNO + NH3������ƽ�������������գ�
HCNO + NH3������ƽ�������������գ�
��1����������Ӧ���漰�ĸ�Ԫ���У�ԭ�Ӱ뾶����Ԫ����_________����ԭ�Ӻ�����ӹ�ռ��_______�������
��2��������������������Ԫ�ش���ͬ���ڣ�������������˵������Ԫ��C,N,O�ǽ����Եݱ���ɵ���ʵ��____________��
a������������Ӧˮ���������
b��������H2��Ӧ�����׳̶�
c�������γɻ�������Ԫ�صĻ��ϼ�
��ҵ���ڴ��������£���NH3��Ϊ��ԭ���������е�NOx��ԭ�����ĵ���ˮ����Ӧ����ʽ�ɱ�ʾΪ��
2NH3��g����NO��g����NO2��g�� 2N2��g����3H2O��g��
2N2��g����3H2O��g��
��3��һ�������¸÷�Ӧ���ݻ�Ϊ2L�������ڷ�Ӧ��20 minʱ�ﵽƽ�⣬����N2 0.4 mol����ƽ����Ӧ���ʦ�(NO)��__________mol/L��min��
��4�����÷�Ӧ�ﵽƽ��ʱ������˵��һ���������___________��
a�������ܶȲ��� b��������ɫ����
c����(NO)��= 2��(N2)�� d��N2��NH3��Ũ�����
��5��X���Դ���NH3��ԭ�����е�NOx��X�ķ���ģ����ͼ��ʾ��X����ʽΪ______________��
��6�����������ֽṹ��һ�ַ����ں�����������Ϊ���ᣬ��һ�ַ����ڲ�����������Ϊ�����ᣬ�����ֽṹ������ԭ���������Ѵﵽ�ȶ��ṹ��������Ҳ������״�ṹ����ֱ�д�������������Ľṹʽ��_______________��______________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪X��Y��Z��W���ֶ�����Ԫ�ص�ԭ������������������X��Y��Z��W���γɵij����������ڳ����¾�����̬�������ڱ���Z��W�������ڣ�Y������������ˮ���������⻯�ﷴӦ�����Σ���Y�ĺ˵������W��������������ͬ����ش��������⣺
��1��Z�����ӽṹʾ��ͼ�� ��
��2��X��Y��W�����һ�������ԭ�Ӹ���֮��Ϊ4��1��1���仯ѧʽ�к��еĻ�ѧ���� ��
��3��YX3���ӵĿռ乹���� ��Z���⻯����ȶ��Ա�W���⻯����ȶ���_____���ǿ������������
��4��Z2W2�����У�W��Z������8e-�ȶ��ṹ����Z 2W2�ĵ���ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
1886�꣬�¹���������Frieberg����ҵѧԺ��һλ�����ڷ����������������ֵ�һ���¿�ʯ����X��ʾ��XΪ���Ȼ������ʱ����������һ�ֵ�ʱδ֪����Ԫ�أ���Y��ʾ����X��Y�Ļ��ϼ�Ϊ+4������ͨ��ʵ����֤���Լ����ƶϡ���������X�к�����Ԫ�أ����ϼ�Ϊ+1������Ԫ�أ����ϼ�Ϊ-2��������Ag����������Ϊ76��53%�����������м���X���ɵõ�Ag2S��H2S��Y�ĵͼ����Y�Ļ��ϼ�Ϊ+2���������200���100kPa��������ȫת��16��0g��X��Ҫ0��557L�����������������������
��1������Y��Ħ��������ָ��Y��Ԫ�����ڱ��е�λ��
��2��д��X�Ļ�ѧʽ
��3��д��������X��Ӧ�Ļ�ѧ����ʽ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����������(BN)n��һ�����ϳɲ��ϣ����ʽΪ(BN)�ݣ����и�Ӳ�ȡ����µ��ص㣬���������������մɲ��ϡ�ĥ�ϡ���ĥ�оߵĺò��ϡ�����ɰ��Na2B4O7���������ڸ��¸�ѹ�·�Ӧ���Ի�á����磺 Na2B4O7 + 2CO(NH2)2 �� 4(BN) + Na2O + 2CO2
��1���������������ʽ��ʾ��ɰ�Ļ�ѧʽ ������(BN)n�Ⱦ������и���Ӳ�Ⱥ������Ե�ԭ���ǣ� ��
��21��������Ӧʽ�о���4�ֲ�ͬ�������ӵ�ԭ�ӣ���������Ԫ�������ڱ��д��ڵ� ���ڣ��� �塣
��3�������ڱȽ�N��O�ǽ��������ǿ������ʵ�� ��
| A������������Ӧˮ��������� | B��H2O(g) ��NH3(g)�ȶ� |
| C��������H2��Ӧ�����׳̶� | D��NO�е�Ԫ�������ۣ���Ԫ���Ը��� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com