
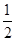 Cl2(g)=NaCl(s)�� ��H1
Cl2(g)=NaCl(s)�� ��H1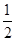 Cl2(g)=Cl(g)����H4��Cl(g)��e��=Cl��(g)�� ��H5
Cl2(g)=Cl(g)����H4��Cl(g)��e��=Cl��(g)�� ��H5| A����H����H1����H2����H3����H4����H5 |
| B����H����H1����H2����H3����H4����H5 |
| C����H����H1����H2����H3����H4����H5 |
| D����H����H2����H3����H4����H5����H1 |

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
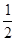 O2(g)=H2O(g)
O2(g)=H2O(g) 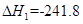 kJ��mol-1���� C(s)+
kJ��mol-1���� C(s)+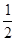 O2(g)="CO(g)"
O2(g)="CO(g)" 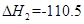 kJ��mol-1���ɴ˿�֪��̿��ˮ������Ӧ���Ȼ�ѧ����ʽΪ��C(s)+H2O(g)=CO(g)+H2(g)
kJ��mol-1���ɴ˿�֪��̿��ˮ������Ӧ���Ȼ�ѧ����ʽΪ��C(s)+H2O(g)=CO(g)+H2(g) 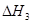 ,��
,��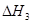 Ϊ
Ϊ| A��+131.3 kJ��mol-1 | B��-131.3 kJ��mol-1 |
| C��+352.3 kJ��mol-1 | D��-352.3 kJ��mol-1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 (NH4)2CO3 (aq) ��H1
(NH4)2CO3 (aq) ��H1 NH4HCO3 (aq) ��H2
NH4HCO3 (aq) ��H2 2NH4HCO3 (aq) ��H3
2NH4HCO3 (aq) ��H3
|
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 CH3OH(g) +H2O(g) ��H =��49.0 kJ��mol��1
CH3OH(g) +H2O(g) ��H =��49.0 kJ��mol��1 
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
 2NH3����Ӧ��1mol N2�ų�������Ϊ92.4kJ����Ͽ�1mol N��N�������յ������ǣ� ��
2NH3����Ӧ��1mol N2�ų�������Ϊ92.4kJ����Ͽ�1mol N��N�������յ������ǣ� ��| A��431kJ | B��945.6kJ | C��649kJ | D��869kJ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
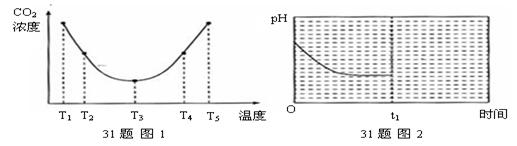
 4NO��g��+6 H2O��g����H��
4NO��g��+6 H2O��g����H��
 4NO��g��+6 H2 O��g���ﵽƽ��״̬���� ������ţ���
4NO��g��+6 H2 O��g���ﵽƽ��״̬���� ������ţ���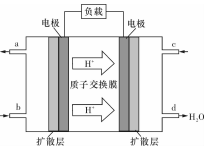
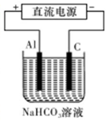
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
 2CO(g)����H="-221.0" kJ��mol-1
2CO(g)����H="-221.0" kJ��mol-1 2H2O(g)����H="-483.6" kJ��mol-1
2H2O(g)����H="-483.6" kJ��mol-1 CO(g)+H2(g)�Ħ�HΪ(����)
CO(g)+H2(g)�Ħ�HΪ(����)| A��+131.3 kJ��mol-1 | B��-131.3 kJ��mol-1 | C��-352.3 kJ��mol-1 | D��+262.6 kJ��mol-1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
 2SO3(g)��H="-197" kJ��mol-1��ʵ����4 mol SO2�μ�������Ӧ�ų�354 kJ��������SO2��ת������ӽ���( )
2SO3(g)��H="-197" kJ��mol-1��ʵ����4 mol SO2�μ�������Ӧ�ų�354 kJ��������SO2��ת������ӽ���( )| A��90% | B��80% | C��50% | D��40% |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 CO2(g) +3 H2 (g)�����CO2��CH3OH(g)��Ũ����ʱ��仯���±���ʾ��
CO2(g) +3 H2 (g)�����CO2��CH3OH(g)��Ũ����ʱ��仯���±���ʾ�� | ʱ�� ���� | 0 min | 10 min | 30 min | 60 min | 70 min |
| CO2(mol/L) | 0 | 0.2 | 0.6 | 0.8 | 0.8 |
| CH3OH(mol/L) | 1.0 | 0.8 | 0.4 | 0.2 | 0.2 |
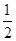 O2 (g)
O2 (g) CO2(g) + 2H2 (g) ?H1= ��192.9kJ/mol
CO2(g) + 2H2 (g) ?H1= ��192.9kJ/mol 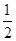 O2 (g)
O2 (g) H2 O(g) ?H2= ��120.9kJ/mol
H2 O(g) ?H2= ��120.9kJ/mol 
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com