
|
�����£���H2O2��Һ�еμ�����FeSO4��Һ���ɷ�������������Ӧ��2Fe2+��H2O2��2H+ | |
| [����] | |
A�� |
H2O2 �������Ա�Fe3+ǿ���仹ԭ�Ա�Fe2+�� |
B�� |
��H2O2�ֽ�����У���Һ��pH���½� |
C�� |
��H2O2�ֽ�����У�Fe2+��Fe3+���������ֲ��� |
D�� |
H2O2����������Ҫ����Fe2+ |

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
�������⣨H2O2���ֳ�˫��ˮ������������һ��Һ�壬���ѵ��룬���ֽ⡣��Ϊһ�־��仯����Ʒ����ҵ������100����ǰ�㿪ʼ��������������������������������̬������������Ҫ������������������ܵ����ǵ�������������Ϊ��ɫ��������Ϊ�����桢���䡢ʹ�õķ��㣬��ҵ�ϲ��á�������������ת��Ϊ��̬�Ĺ�̼���ƾ��壨�仯ѧʽΪ2Na2CO3��3H2O2)���þ������Na2CO3��H2O2��˫�����ʡ��������������������չ������£���0��5 ���£������͵�Na2CO3��Һ�У��ȼ���H2O2���ȶ�����MgCl2��Na2SiO3)������ַ�Ӧ���ټ�������������˷��룬���ù�̼���ƾ��塣
�����������ϣ�����������⣺
��1�����������е�֪ʶ��д��H2O2�Ľṹʽ__________����̬ʱ����__________���塣
��2������ΪH2O2����Ϊ��ɫ����������Ҫԭ���ǣ�____________________________��
��3������֪����ϡ�����ͭ��Ӧ������ϡ�����м���H2O2�����ʹͭ˳���ܽ⣬д���÷�Ӧ�����ӷ���ʽ_______________________________________��
��4���ȶ����ܷ�Ӧ���������ォ��̼�������Ӱ���ס���÷�Ӧ�Ļ�ѧ����ʽΪ��________
___________________________________________��
��5�������������������____________________________________________��
��6���������ʲ���ʹ��̼����ʧЧ����______________��
A.MnO2 B.H2S C.CH3COOH D.NaHCO3
(7)���з�̪��NaOH��Һ�еμ�˫��ˮ����Һ�ɺ�ɫ������ɫ����Ҫԭ����˫��ˮ��________�ԡ�
��8����˫��ˮ���뾭�ữ�ĸ��������Һ�У���Һ���Ϻ�ɫ�����ˣ���ʱ˫��ˮ���ֳ�________�ԣ�����H218O2��˫��ˮ���ữ�ĸ�����ط�Ӧ��18O������������________�
��9�����õ��ͻ�����ɫ��λ��PbSO4�������ڣ�PbS������˫��ˮ�������ָֻ�ԭò���йط�Ӧ�Ļ�ѧ����ʽΪ_________________________________������0.1 mol��PbS���뷴Ӧ�����ڴ˷�Ӧ�з���ת�Ƶĵ���Ϊ________mol��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ���Ϻ��г����ظ�����ѧ����ĩ��һģ�����Ի�ѧ�Ծ��������棩 ���ͣ������
��������ĵ���ƽ�ⳣ�����±���
|
���� |
HCOOH |
HClO |
H2CO3 |
H2SO3 |
|
����ƽ�ⳣ�� ��25�棩 |
|
|
|
|
��1�����¶���ͬʱ���������Kiֵ�����Ե����ǿ���Ĺ�ϵΪ��________________________��
��2���������ӷ���ʽ��ȷ����
A��2ClO�� + H2O + CO2 �� 2HClO + CO32��
B��2HCOOH + CO32�� �� 2HCOO�� + H2O + CO2��
C��H2SO3 + 2HCOO�� �� 2HCOOH + SO32��
D��Cl2 + H2O+2CO32�� �� 2HCO3�� + Cl�� + ClO��
��3�������£�pH=3��HCOOH��Һ��pH=11��NaOH��Һ�������Ϻ���Һ������Ũ���ɴ�С��˳��Ϊ������������������������������������������������������������������
������(H2SeO3)Ҳ��һ�ֶ�Ԫ���ᣬ��������һ����ɫ���壬������ˮ���н�ǿ�������ԡ�
��4������������Һ�в���ͨ��SO2 ��������ɫ���ʣ�д���÷�Ӧ�Ļ�ѧ����ʽ������������������������ ��������������������������������������������
��5������������30%��H2O2���ȿ��Ƶ�����(H2SeO4)����Ӧ����ʽ���£�
H2SeO3 + H2O2 �� H2SeO4+H2O������˵������ȷ���ǡ���������������������������������
A��H2O2�������������ǻ�ԭ��
B��H2O �Ȳ������������ֲ��ǻ�ԭ����
C��H2SeO4���������������ǻ�ԭ����
D�������ԣ�H2SeO3��H2SeO4
���ᣨH6TeO6����һ�ֺ������ᣬ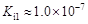 �������������Ա����ỹҪǿ�������Խ����У�����ɽ�HI������I2������ʽ���£�
�������������Ա����ỹҪǿ�������Խ����У�����ɽ�HI������I2������ʽ���£�
���� ��HI+���� ��H6TeO6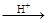 ���� ��TeO2+�� ����Te+���� ��I2+�� ����H2O
���� ��TeO2+�� ����Te+���� ��I2+�� ����H2O
��6������Ӧ�����ɵ�TeO2��Te�����ʵ���֮��Ϊ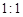 ������ƽ������ѧ����ʽ��
������ƽ������ѧ����ʽ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��
�����������ϣ�����������⣺
(1)���������е�֪ʶ��д��H2O2�Ľṹʽ______________________��
(2)����ΪH2O2����Ϊ��ɫ����������Ҫԭ���ǣ�________________��
(3)����֪����ϡ�����ͭ��Ӧ������ϡ�����м���H2O2�����ʹͭ˳���ܽ⣬д���÷�Ӧ�����ӷ���ʽ_____________________��
(4)�ȶ����ܷ�Ӧ���������ォ��̼�������Ӱ���ס���÷�Ӧ�Ļ�ѧ����ʽΪ��___________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com