
��14�֣�ijУ��һ��ѧ�о���ѧϰС����ճ������е�������;�����˵��飬�˽�����ɹ㷺��Ӧ���������Ư�ס�ˮ���ɱ���������ȡ�
(1)������������Ư�ס�������ԭ������������ˮ�����γɾ���________�ԵĴ����ᣬ��ط�Ӧ�����ӷ���ʽΪ________________________��
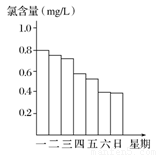
(2)�ڵ���ij������Ӿ���ļ���ˮ�������ʱ��С���Ա�˽������Ӿ��ÿ��һ����8��00��ˮ��Ȼ��ͨ������������ͨ����Ӿ��ˮ�ĺ�����������0.5 mg/L��1.0 mg/L ֮��ʱ��Ч����á���ͼ�Ǹ�С��ⶨ��ÿ��19��00ʱ��Ӿ����ˮ�ĺ����������ļ���ʹ����Ӿ�ز�����_________________��
(3)����Ϊ�ļ�����������ȡ�����ǿ��________��˵��һ��������____________ ____________��
(4)�ڶ���Ӿ��ˮ��ͨ����������ʱ������������й©��Ӧ�����ر������ޣ���Ӧ��ȡ�����Ծȷ���________��(ѡ�����)
A. ��ʪ���ë����ס�ڱ�����ʹ�
B. �ý�ʪС�մ�����ˮ��ë����ס�ڱ�����ߴ�
C. �ý�ʪŨ��ˮ��ë����ס�ڱ�����������ȫ��
D. �ý�ʪʳ��ˮ��ë����ס�ڱ�˳��������ȫ��
(5)С����Ӿ��ͨ��ʹ��Ư��Һ(NaClO��Һ)����������������ˮ���Ծٳ�ʹ��Ư��Һ��������������һ������________________________��
��14�֣���1��ǿ����
Cl2+H2O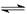 H++Cl-+HClO
H++Cl-+HClO
��2����������������
��3�������ġ������� ����ǿ�ң�HClO���ֽ⣬���º������½�����
��4��B ��5��NaClO���ȶ������ڴ洢������
����������1����������ˮ����ˮ��Ӧ���ɾ���ǿ�����ԵĴ����ᣬ����ʽΪCl2+H2O H++Cl-+HClO��
H++Cl-+HClO��
��2������ͼ���֪����������������������Ӿ��ˮ�ĺ�����С��0.5 mg/L�����Դ�ʱʹ���Dz������ġ�
��3�����ڴ�����ȶ��������ֽ⣬��˵��º������½����ԣ����Ը���ͼ����жϣ�Ӧ���������ġ���������
��4���������ܶȴ��ڿ����ģ�����Ӧ����ߴ��ܣ�A����ȷ�����������ڼ��У�������̫ǿ�ḯʴƤ��������ѡ��B��ȷ��C����ȷ����ΪŨ��ˮ���д̼��ԡ�DҲ����ȷ��Ӧ��������ܣ���ѡB��
��5�����������壬��Ư��Һ��Һ�壬���ڴ洢�����䣬��NaClO���ȶ���


 ֥�鿪���γ�������ϵ�д�
֥�鿪���γ�������ϵ�д� ����ѧ��ţ��Ӣ��ϵ�д�
����ѧ��ţ��Ӣ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 ijУ��һ��ѧ�о���ѧϰС����ճ������е�������;�����˵��飬�˽�����ɹ㷺��Ӧ���������Ư�ס�ˮ���ɱ���������ȣ�
ijУ��һ��ѧ�о���ѧϰС����ճ������е�������;�����˵��飬�˽�����ɹ㷺��Ӧ���������Ư�ס�ˮ���ɱ���������ȣ��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ijУ��һ��ѧ�о���ѧϰС����ճ������е�������;�����˵��飬�˽�����ɹ㷺��Ӧ���������Ư�ס�ˮ���ɱ���������ȡ�
(1)������������Ư�ס�������ԭ������Ϊ����ˮ�����γɾ���______�ԵĴ����ᣬ��ط�Ӧ�Ļ�ѧ����ʽΪ____________________________��

(2)�ڵ���ij������Ӿ���ļ���ˮ�������ʱ��С���Ա�˽������Ӿ��ÿ��һ����8��00��ˮ��Ȼ��ͨ������������ͨ����Ӿ����ˮ�ĺ�����������0.5 mg��L��1��1.0 mg��L��1֮��ʱ��Ч����á���ͼ�Ǹ�С��ⶨ��ÿ��19��00ʱ��Ӿ����ˮ�ĺ��������ļ���ʹ����Ӿ�ز���ȫ��______��
(3)����Ϊ�ļ�����������ȡ�����ǿ��______��˵��һ��������
__________________________(��Ҫ�ķ���ʽ������)��
(4)�ڶ���Ӿ��ˮ��ͨ����������ʱ������������й©ʱ��Ӧ�����ر������ޣ���Ӧ��ȡ�����Ծȷ���______��
A����ʪ���ë����ס�ڱ�����ʹ�
B���ý�ʪС�մ�����ˮ��ë����ס�ڱ�����ߴ�
C���ý�ʪŨ��ˮ��ë����ס�ڱ�����������ȫ��
D���ý�ʪʳ��ˮ��ë����ס�ڱ�˳��������ȫ��
(5)С����Ӿ��ͨ��ʹ��Ư��Һ(NaClO��Һ)����������������ˮ���Ծٳ�ʹ��Ư��Һ��������������һ������__________________���û�ѧ����ʽ˵����ҵ���������Ư��Һ��________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(15��)ijУ��һ��ѧ�о���ѧϰС����ճ������е�������;�����˵��飬�˽�����ɹ㷺��Ӧ���������Ư�ס�ˮ���ɱ���������ȡ�
(1)������������Ư�ס�������ԭ������Ϊ����ˮ�����γɾ���______�ԵĴ����ᣬ��ط�Ӧ�Ļ�ѧ����ʽΪ____________________________��
(2)�ڵ���ij������Ӿ���ļ���ˮ�������ʱ��С���Ա�˽������Ӿ��ÿ��һ����8��00��ˮ��Ȼ��ͨ������������ͨ����Ӿ����ˮ�ĺ�����������0.5 mg��L��1��1.0 mg��L��1֮��ʱ��Ч����á���ͼ�Ǹ�С��ⶨ��ÿ��19��00ʱ��Ӿ����ˮ�ĺ��������ļ���ʹ����Ӿ�ز���ȫ��______��
(3)����Ϊ�ļ�����������ȡ�����ǿ��______��˵��һ��������
__________________________(��Ҫ�ķ���ʽ������)��
(4)�ڶ���Ӿ��ˮ��ͨ����������ʱ������������й©ʱ��Ӧ�����ر������ޣ���Ӧ��ȡ�����Ծȷ���______��
A����ʪ���ë����ס�ڱ�����ʹ�
B���ý�ʪС�մ�����ˮ��ë����ס�ڱ�����ߴ�
C���ý�ʪŨ��ˮ��ë����ס�ڱ�����������ȫ��
D���ý�ʪʳ��ˮ��ë����ס�ڱ�˳��������ȫ��
(5)С����Ӿ��ͨ��ʹ��Ư��Һ(NaClO��Һ)����������������ˮ���Ծٳ�ʹ��Ư��Һ��������������һ������__________________���û�ѧ����ʽ˵����ҵ���������Ư��Һ��________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�긣��ʡ�������и�һ��ѧ�����п��Ի�ѧ�Ծ����������� ���ͣ������
��11�֣�ijУ��һ��ѧ�о���ѧϰС����ճ������е�������;�����˵��飬�˽�����ɹ㷺��Ӧ���������Ư�ס�ˮ���ɱ���������ȡ�
��1��������������Ư�ס�������ԭ������Ϊ����ˮ�����γɾ���Ư�ס��������õ�_________����ط�Ӧ�����ӷ���ʽΪ____________________________��
��2)�ڵ���ij������Ӿ���ļ���ˮ�������ʱ��С���Ա�˽������Ӿ��ÿ��һ����8��00��ˮ��Ȼ��ͨ������������ͨ����Ӿ����ˮ�ĺ���������Ч�ȣ�������0.5 mg��L��1��1.0 mg��L��1֮��ʱ��Ч����á���ͼ�Ǹ�С��ⶨ��ÿ��19��00ʱ��Ӿ����ˮ�ĺ��������ļ���ʹ����Ӿ�ز���ȫ��________________��
(3)����Ϊ�ļ�����������ȡ�����ǿ��___________________________��˵��һ��������____________________________________________________(��Ҫ�ķ���ʽ������)��
(4)�ڶ���Ӿ��ˮ��ͨ����������ʱ������������й©ʱ��Ӧ�����ر������ޣ���Ӧ��ȡ�����Ծȷ���______��
| A����ʪ���ë����ס�ڱ�����ʹ� |
| B���ý�ʪС�մ�����ˮ��ë����ס�ڱ�����ߴ� |
| C���ý�ʪŨ��ˮ��ë����ס�ڱ�����������ȫ�� |
| D���ý�ʪʳ��ˮ��ë����ס�ڱ�˳��������ȫ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ������ʡ��ǿ��������ѧ��һ��ѧ�����л�ѧ�Ծ����������� ���ͣ�ʵ����
��14�֣�ijУ��һ��ѧ�о���ѧϰС����ճ������е�������;�����˵��飬�˽�����ɹ㷺��Ӧ���������Ư�ס�ˮ���ɱ���������ȡ�
(1)������������Ư�ס�������ԭ������������ˮ�����γɾ���________�ԵĴ����ᣬ��ط�Ӧ�����ӷ���ʽΪ________________________��
(2)�ڵ���ij������Ӿ���ļ���ˮ�������ʱ��С���Ա�˽������Ӿ��ÿ��һ����8��00��ˮ��Ȼ��ͨ������������ͨ����Ӿ��ˮ�ĺ�����������0.5 mg/L��1.0 mg/L ֮��ʱ��Ч����á���ͼ�Ǹ�С��ⶨ��ÿ��19��00ʱ��Ӿ����ˮ�ĺ����������ļ���ʹ����Ӿ�ز�����_________________��
(3)����Ϊ�ļ�����������ȡ�����ǿ��________��˵��һ��������____________ ____________��
(4)�ڶ���Ӿ��ˮ��ͨ����������ʱ������������й©��Ӧ�����ر������ޣ���Ӧ��ȡ�����Ծȷ���________��(ѡ�����)
| A����ʪ���ë����ס�ڱ�����ʹ� |
| B���ý�ʪС�մ�����ˮ��ë����ס�ڱ�����ߴ� |
| C���ý�ʪŨ��ˮ��ë����ס�ڱ�����������ȫ�� |
| D���ý�ʪʳ��ˮ��ë����ס�ڱ�˳��������ȫ�� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com