
“ŲÄŃõ»ÆĢśµÄ»ÆѧĆū³ĘŹĒŃõ»ÆĢś£ØFe2O3£©£¬ĖüŹĒµē×Ó”¢µēŠÅ¹¤ŅµµÄ“ÅŠŌ²ÄĮĻ£¬¹¤ŅµÉĻ²ÉÓĆÉś²śīŃ°×·ŪµÄĻĀ½ÅĮĻ£Øŗ¬“óĮæFeSO4ČÜŅŗ£©Öʱø“ŲÄŃõ»ÆĢśµÄ²½ÖčČēĻĀ£ŗ
¢ŁĻņĻĀ½ÅĮĻ£Øŗ¬“óĮæFeSO4ČÜŅŗ£©ÖŠ¼ÓČėÉŁĮæ2 mo1”¤L-1½ŠH2SO4ŗĶĢśĘ¤£»
¢ŚĻņ¢ŁÖŠĖłµĆČÜŅŗÖŠ¼ÓŠõÄż¼ĮŗĶĖ®£¬¾¹żĀĖ³żČ„Īüø½ĮĖŌÓÖŹµÄŠõÄż¼Į£»
¢Ū½«¢ŚĖłµĆµÄĀĖŅŗÅØĖõ½į¾§µĆµ½¾§ĢåA£»
¢Ü½«¾§ĢåAČÜÓŚĖ®£¬²¢¼ÓČėNH4HCO3£¬²śÉśCO2ĘųĢåĶ¬Ź±µĆµ½FeCO3³ĮµķŗĶĪŽÉ«ČÜŅŗC£»
¢Ż½«FeCO3³ĮµķĻ“µÓ”¢ŗęøɲ¢ģŃÉÕ”££ØģŃÉÕÖŠµÄ±ä»ÆĪŖ£ŗFeCO3=FeO+CO2”ü; 4FeO+O2=2 Fe2O3£©
¾ŻÉĻŹöŠÅĻ¢»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ÓĆ18.4mo1”¤L-1µÄH2SO4ÅäÖĘ500mL 2 mo1”¤L-1H2SO4£¬ĖłŠč²£Į§ŅĒĘ÷³ż
mLĮæĶ²”¢²£Į§°ō”¢ÉÕ±”¢500mI”£ČŻĮæĘæĶā£¬»¹ŠčŅŖ ”£
£Ø2£©²½Öč¢ŁÖŠ2mo1”¤L-1H2SO4ŗĶĢśĘ¤µÄ×÷ÓĆ·Ö±šĪŖ ”£
£Ø3£©¾§ĢåAµÄ»ÆѧŹ½ĪŖ £¬¼ģŃéČÜŅŗCÖŠČÜÖŹŃōĄė×ӵķ½·ØŹĒ ”£
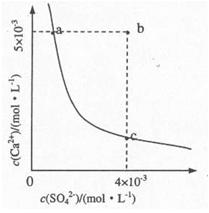
£Ø4£©ĻņČÜŅŗCÖŠ¼ÓČėCaCl2ČÜŅŗÄܵƵ½CaSO4³Įµķ£¬³£ĪĀĻĀKSP£ØCaSO4£©=9x10-6£¬³£ĪĀĻĀCaSO4ŌŚĖ®ÖŠµÄ³ĮµķČܽāĘ½ŗāĒśĻßČēĶ¼ĖłŹ¾”£
¢Łaµć¶ŌÓ¦µÄKSP cµć¶ŌÓ¦µÄKSPĢī”°“óÓŚ”±”¢”°Š”ÓŚ”±»ņ”°µČÓŚ”±£©£»
¢ŚČōÓÉbµć±äµ½aµćĻĀĮŠ“ėŹ©æÉŠŠµÄŹĒ ”£
A£®¼ÓČėŹŹĮæCaCl2 B£®¼ÓČėŹŹĮæBaCl2£®
C£®¼ÓČĖŹŹĮæNa2SO4 D£®Õō·¢

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
“ŲÄŃõ»ÆĢśµÄ»ÆѧĆū³ĘŹĒŃõ»ÆĢś£ØFe2O3£©£¬ĖüŹĒµē×Ó”¢µēŠÅ¹¤ŅµµÄ“ÅŠŌ²ÄĮĻ£¬¹¤ŅµÉĻ²ÉÓĆÉś²śīŃ°×·ŪµÄĻĀ½ÅĮĻ£Øŗ¬“óĮæFeSO4ČÜŅŗ£©Öʱø“ŲÄŃõ»ÆĢśµÄ²½ÖčČēĻĀ£ŗ
¢ŁĻņĻĀ½ÅĮĻ£Øŗ¬“óĮæFeSO4ČÜŅŗ£©ÖŠ¼ÓČėÉŁĮæ2 mo1”¤L-1½ŠH2SO4ŗĶĢśĘ¤£»
¢ŚĻņ¢ŁÖŠĖłµĆČÜŅŗÖŠ¼ÓŠõÄż¼ĮŗĶĖ®£¬¾¹żĀĖ³żČ„Īüø½ĮĖŌÓÖŹµÄŠõÄż¼Į£»
¢Ū½«¢ŚĖłµĆµÄĀĖŅŗÅØĖõ½į¾§µĆµ½¾§ĢåA£»
¢Ü½«¾§ĢåAČÜÓŚĖ®£¬²¢¼ÓČėNH4HCO3£¬²śÉśCO2ĘųĢåĶ¬Ź±µĆµ½FeCO3³ĮµķŗĶĪŽÉ«ČÜŅŗC£»
¢Ż½«FeCO3³ĮµķĻ“µÓ”¢ŗęøɲ¢ģŃÉÕ”££ØģŃÉÕÖŠµÄ±ä»ÆĪŖ£ŗFeCO3=FeO+CO2”ü; 4FeO+O2=2 Fe2O3£©
¾ŻÉĻŹöŠÅĻ¢»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ÓĆ18.4mo1”¤L-1µÄH2SO4ÅäÖĘ500mL 2 mo1”¤L-1H2SO4£¬ĖłŠč²£Į§ŅĒĘ÷³ż
mLĮæĶ²”¢²£Į§°ō”¢ÉÕ±”¢500mI”£ČŻĮæĘæĶā£¬»¹ŠčŅŖ ”£
£Ø2£©²½Öč¢ŁÖŠ2 mo1”¤L-1H2SO4ŗĶĢśĘ¤µÄ×÷ÓĆ·Ö±šĪŖ ”£
£Ø3£©¾§ĢåAµÄ»ÆѧŹ½ĪŖ £¬¼ģŃéČÜŅŗCÖŠČÜÖŹŃōĄė×ӵķ½·ØŹĒ ”£
£Ø4£©ĻņČÜŅŗCÖŠ¼ÓČėCaCl2ČÜŅŗÄܵƵ½CaSO4³Įµķ£¬³£ĪĀĻĀKSP£ØCaSO4£©=9x10-6£¬³£ĪĀĻĀCaSO4ŌŚĖ®ÖŠµÄ³ĮµķČܽāĘ½ŗāĒśĻßČēĶ¼ĖłŹ¾”£
¢Łaµć¶ŌÓ¦µÄKSP cµć¶ŌÓ¦µÄKSPĢī”°“óÓŚ”±”¢”°Š”ÓŚ”±»ņ”°µČÓŚ”±£©£»
¢ŚČōÓÉbµć±äµ½aµćĻĀĮŠ“ėŹ©æÉŠŠµÄŹĒ ”£
A£®¼ÓČėŹŹĮæCaCl2 B£®¼ÓČėŹŹĮæBaCl2£®
C£®¼ÓČĖŹŹĮæNa2SO4 D£®Õō·¢
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
£Ø16·Ö£©“ŲÄŃõ»ÆĢśµÄ»ÆѧĆū³ĘŹĒŃõ»ÆĢś£ØFe2O3£©£¬ĖüŹĒµē×Ó”¢µēŠÅ¹¤ŅµµÄ“ÅŠŌ²ÄĮĻ£¬¹¤ŅµÉĻ²ÉÓĆÉś²śīŃ°×·ŪµÄĻĀ½ÅĮĻ£Øŗ¬“óĮæFeSO4µÄ·ĻŅŗ£©ĪŖŌĮĻĄ“Öʱø“ŲÄŃõ»ÆĢś”£
ŅŃÖŖģŃÉÕÖŠµÄ»Æѧ·“Ó¦·½³ĢŹ½ĪŖ£ŗFeCO3 ”ś FeO + CO2”ü£¬4FeO + O2 ”ś 2Fe2O3
£Ø1£©ÓĆ98%µÄH2SO4µÄÅäÖĘ500mLµÄ20%µÄH2SO4£¬ĖłŠč²£Į§ŅĒĘ÷ŹĒ
A”¢²£Į§°ō B”¢ÉÕ± C”¢Ā©¶· D”¢250mLČŻĮæĘæ
E”¢500mLČŻĮæĘæ F”¢½ŗĶ·µĪ¹Ü
£Ø2£©ÅØĖõ½į¾§ŗóµĆµ½µÄ¾§ĢåŹĒ £ØĢī»ÆѧŹ½£©£¬AÖŠČÜÖŹŹĒ £»
“ĖČÜŅŗÖŠø÷Ąė×ÓµÄÅØ¶Č±Č½Ļ“óŠ”ĪŖ£ŗ ”£
£Ø3£©20%H2SO4ŗĶĢśĘ¤µÄ×÷ÓĆ·Ö±šŹĒ ”£
£Ø4£©¼ģŃéĀĖŅŗÖŠŗ¬ÓŠNH4+µÄ·½·ØŹĒ
£Ø5£©Š“³ö²½Öč”°ŗĻ³É”±ÖŠ·¢ÉśµÄ»Æѧ±ä»Æ£ØÓĆ»Æѧ·½³ĢŹ½±ķŹ¾£©£ŗ
ӣ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2011½ģ¹ć¶«Ź”ĆÆĆūŹŠøßČżµŚŅ»“ĪÄ£Äāæ¼ŹŌ£ØĄķ×Ū£©»Æѧ²æ·Ö ĢāŠĶ£ŗĢīæÕĢā
“ŲÄŃõ»ÆĢśµÄ»ÆѧĆū³ĘŹĒŃõ»ÆĢś£ØFe2O3£©£¬ĖüŹĒµē×Ó”¢µēŠÅ¹¤ŅµµÄ“ÅŠŌ²ÄĮĻ£¬¹¤ŅµÉĻ²ÉÓĆÉś²śīŃ°×·ŪµÄĻĀ½ÅĮĻ£Øŗ¬“óĮæFeSO4ČÜŅŗ£©Öʱø“ŲÄŃõ»ÆĢśµÄ²½ÖčČēĻĀ£ŗ
¢ŁĻņĻĀ½ÅĮĻ£Øŗ¬“óĮæFeSO4ČÜŅŗ£©ÖŠ¼ÓČėÉŁĮæ2 mo1”¤L-1½ŠH2SO4ŗĶĢśĘ¤£»
¢ŚĻņ¢ŁÖŠĖłµĆČÜŅŗÖŠ¼ÓŠõÄż¼ĮŗĶĖ®£¬¾¹żĀĖ³żČ„Īüø½ĮĖŌÓÖŹµÄŠõÄż¼Į£»
¢Ū½«¢ŚĖłµĆµÄĀĖŅŗÅØĖõ½į¾§µĆµ½¾§ĢåA£»
¢Ü½«¾§ĢåAČÜÓŚĖ®£¬²¢¼ÓČėNH4HCO3£¬²śÉśCO2ĘųĢåĶ¬Ź±µĆµ½FeCO3³ĮµķŗĶĪŽÉ«ČÜŅŗC£»
¢Ż½«FeCO3³ĮµķĻ“µÓ”¢ŗęøɲ¢ģŃÉÕ”££ØģŃÉÕÖŠµÄ±ä»ÆĪŖ£ŗFeCO3="FeO+CO2”ü; " 4FeO+O2="2" Fe2O3£©
¾ŻÉĻŹöŠÅĻ¢»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ÓĆ18.4mo1”¤L-1µÄH2SO4ÅäÖĘ500mL 2 mo1”¤L-1H2SO4£¬ĖłŠč²£Į§ŅĒĘ÷³ż
mLĮæĶ²”¢²£Į§°ō”¢ÉÕ±”¢500mI”£ČŻĮæĘæĶā£¬»¹ŠčŅŖ ”£
£Ø2£©²½Öč¢ŁÖŠ2 mo1”¤L-1H2SO4ŗĶĢśĘ¤µÄ×÷ÓĆ·Ö±šĪŖ ”£
£Ø3£©¾§ĢåAµÄ»ÆѧŹ½ĪŖ £¬¼ģŃéČÜŅŗCÖŠČÜÖŹŃōĄė×ӵķ½·ØŹĒ ”£
£Ø4£©ĻņČÜŅŗCÖŠ¼ÓČėCaCl2ČÜŅŗÄܵƵ½CaSO4³Įµķ£¬³£ĪĀĻĀKSP£ØCaSO4£©=9x10-6£¬³£ĪĀĻĀCaSO4ŌŚĖ®ÖŠµÄ³ĮµķČܽāĘ½ŗāĒśĻßČēĶ¼ĖłŹ¾”£
¢Łaµć¶ŌÓ¦µÄKSP cµć¶ŌÓ¦µÄKSPĢī”°“óÓŚ”±”¢”°Š”ÓŚ”±»ņ”°µČÓŚ”±£©£»
¢ŚČōÓÉbµć±äµ½aµćĻĀĮŠ“ėŹ©æÉŠŠµÄŹĒ ”£
| A£®¼ÓČėŹŹĮæCaCl2 | B£®¼ÓČėŹŹĮæBaCl2£® |
| C£®¼ÓČĖŹŹĮæNa2SO4 | D£®Õō·¢ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2011½ģ¹ć¶«Ź”ĮłŠ£øßČżÉĻѧʌµŚ¶ž“ĪĮŖæ¼£ØĄķ×Ū£©»Æѧ²æ·Ö ĢāŠĶ£ŗŹµŃéĢā
£Ø16·Ö£©“ŲÄŃõ»ÆĢśµÄ»ÆѧĆū³ĘŹĒŃõ»ÆĢś£ØFe2O3£©£¬ĖüŹĒµē×Ó”¢µēŠÅ¹¤ŅµµÄ“ÅŠŌ²ÄĮĻ£¬¹¤ŅµÉĻ²ÉÓĆÉś²śīŃ°×·ŪµÄĻĀ½ÅĮĻ£Øŗ¬“óĮæFeSO4µÄ·ĻŅŗ£©ĪŖŌĮĻĄ“Öʱø“ŲÄŃõ»ÆĢś”£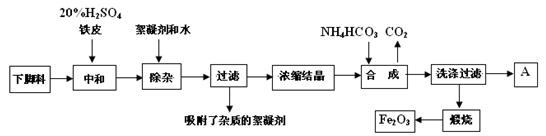

ŅŃÖŖģŃÉÕÖŠµÄ»Æѧ·“Ó¦·½³ĢŹ½ĪŖ£ŗFeCO3 ”ś FeO + CO2”ü£¬4FeO + O2 ”ś 2Fe2O3
£Ø1£©ÓĆ98%µÄH2SO4µÄÅäÖĘ500mLµÄ20%µÄH2SO4£¬ĖłŠč²£Į§ŅĒĘ÷ŹĒ
| A£®²£Į§°ō | B£®ÉÕ± | C£®Ā©¶· | D£®250mLČŻĮæĘæ |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com