


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ʡ�ϲ�һ�С��ϲ�ʮ��2011�������һ��������ѧ���� ���ͣ�022
��ҵ��������������(��Ҫ�ɷ���Al2O3������������Fe2O3��SiO2)��ȡұ��������Ҫԭ����������Ȼ����е�⣮������������ͼ��

(1)��Һ1��Ҫ�����Թ�����ԭ��B��ԭ��B�Ļ�ѧʽ��________��д��������йط�Ӧ�����ӷ�________��________��
(2)���ʡȥ����٣����ܽ��������ֱ�Ӽ���ԭ��B��ʼ�������ջ����������������ʲôӰ�죿________��
(3)����������������н��У����۵��������Ͼ���ʯī������________���IJ����淴Ӧ�Ľ�����Ҫ���ϲ��䣬Ϊʲô��________
(4)������������������ĩ����ȼ�������³��������Ӹֹ죬��Ҫ�������˸÷�Ӧ________�����Ҹ÷�Ӧ�����ٶȿ졢�豸���ף�����Ұ����ҵ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

(1)��Һ1��Ҫ�����Թ�����ԭ��B��ԭ��B�Ļ�ѧʽ��__________________________��
(2)���ʡȥ����٣����ܽ��������ֱ�Ӽ���ԭ��B��ʼ�������ջ����������������ʲôӰ�죿_______________________________________��
(3)����������������н��У����۵��������Ͼ���ʯī������_____________���IJ����淴Ӧ�Ľ�����Ҫ���ϲ��䣬Ϊʲô��_______________________________________��
(4)������������������ĩ����ȼ�������³��������Ӹֹ죬��Ҫ�������˸÷�Ӧ_____________�����Ҹ÷�Ӧ�����ٶȿ졢�豸���ף�����Ұ����ҵ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ҵ����������������Ҫ�ɷ���Al2O3������������Fe2O3��SiO2����ȡұ��������Ҫԭ����������Ȼ����е�⡣������������ͼ��
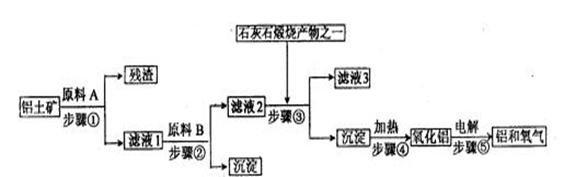
��1����Һ1��Ҫ�����Թ�����ԭ��B��ԭ��B�Ļ�ѧʽ���������� ��д��������йط�Ӧ�����ӷ� �� ��
��2�����ʡȥ����٣����ܽ��������ֱ�Ӽ���ԭ��B��ʼ�������ջ����������������ʲôӰ�죿���� ��
��3������������������н��У����۵��������Ͼ���ʯī�������� ���IJ����淴Ӧ�Ľ�����Ҫ���ϲ��䣬Ϊʲô������������
��4��������������������ĩ����ȼ�������³��������Ӹֹ죬��Ҫ�������˸÷�Ӧ������ ������ �����Ҹ÷�Ӧ�����ٶȿ졢�豸���ף�����Ұ����ҵ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ʡ�ϲ�һ���ϲ�ʮ��2010-2011ѧ��������Ͽ��� ���ͣ��ƶ���
��ҵ����������������Ҫ�ɷ���Al2O3������������Fe2O3��SiO2����ȡұ��������Ҫԭ����������Ȼ����е�⡣������������ͼ��

��1����Һ1��Ҫ�����Թ�����ԭ��B��ԭ��B�Ļ�ѧʽ���������� ��д��������йط�Ӧ�����ӷ� �� ��
��2�����ʡȥ����٣����ܽ��������ֱ�Ӽ���ԭ��B��ʼ�������ջ����������������ʲôӰ�죿���� ��
��3������������������н��У����۵��������Ͼ���ʯī�������� ���IJ����淴Ӧ�Ľ�����Ҫ���ϲ��䣬Ϊʲô������������
��4��������������������ĩ����ȼ�������³��������Ӹֹ죬��Ҫ�������˸÷�Ӧ������ ������ �����Ҹ÷�Ӧ�����ٶȿ졢�豸���ף�����Ұ����ҵ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com