
| n |
| V |
| 0.05mol |
| 10mol/L |



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ���㽭ʡ��������������ѧ��һ5���¿����ƻ�ѧ�Ծ����������� ���ͣ�ʵ����
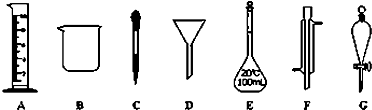
ʵ���ǻ�ѧ�о���һ����Ҫ�ֶΣ�������ͼ��ʾA~G���������������Ҫ����ա�
��1��д���������ƣ�B ��F
��2������ʵ��������õ�����G���� ��ѡ������ѡ��ı����ĸ����
a������ˮ��CC14�Ļ���� b������ˮ�;ƾ��Ļ���� c������ˮ����ɰ�Ļ����
��3��ʵ��������100mL 0.5mol/L��������Һ��
�����й�������E��ʹ�÷����У���ȷ���� ��ѡ������ѡ��ı����ĸ����
a��ʹ��ǰӦ����Ƿ�©Һ b��ʹ��ǰ������
c�������������ʷ�Ӧ���ܽ������ d������Һ��ֱ��ת�Ƶ�����ƿ��
����10mol/L��Ũ���� mL��ȡ�ø��������ʱ����Ҫ�õ����������е�A��
��ѡ�������ı����ĸ����
����ʵ��ʱ�������в��衰�������ȡ���ܽ��ת�ơ����ݡ����浽�Լ�ƿ�С��������ƣ����ݺ�Һ��λ�ã�������ͼ����ȷ���� ��ѡ������ѡ��ı����ĸ����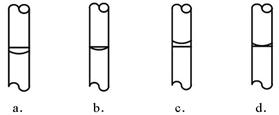
�������Լ�ƿ��ǩ����д��Ӧ���ݣ�ʢ���������ƺõ���Һ��[(��ǩ������ͼ)]��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ���㽭ʡ�����и�һ��ѧ����ĩ��ѧ�Ծ����������� ���ͣ������
ʵ���ǻ�ѧ�о���һ����Ҫ�ֶΣ�������ͼ��ʾA~G���������������Ҫ����ա�
��1��д���������ƣ�B ��F
��2������ʵ��������õ�����G���� ��ѡ������ѡ��ı����ĸ����
a������ˮ��CC14�Ļ���� b������ˮ�;ƾ��Ļ���� c������ˮ����ɰ�Ļ����
��3��ʵ��������100mL 0.5mol/L��������Һ��
�����й�������E��ʹ�÷����У���ȷ���� ��ѡ������ѡ��ı����ĸ����
a��ʹ��ǰӦ����Ƿ�©Һ b��ʹ��ǰ������
c�������������ʷ�Ӧ���ܽ������ d������Һ��ֱ��ת�Ƶ�����ƿ��
������10mol/L��Ũ���� mL��ȡ�ø��������ʱ����Ҫ�õ����������е�A�� ��ѡ�������ı����ĸ����
����ʵ��ʱ�������в��衰�������ȡ���ܽ��ת�ơ����ݡ����浽�Լ�ƿ�С��������ƣ����ݺ�Һ��λ�ã�������ͼ����ȷ���� ��ѡ������ѡ��ı����ĸ����

�������Լ�ƿ��ǩ����д��Ӧ���ݣ�ʢ���������ƺõ���Һ��[(��ǩ������ͼ)]��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com