
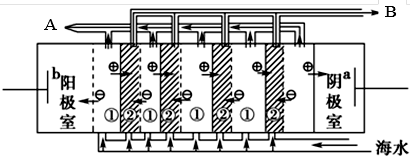



| ƒкЉґ | Єя÷–њќ≥ћ | ƒкЉґ | ≥х÷–њќ≥ћ |
| Єя“ї | Єя“ї√вЈ—њќ≥ћЌ∆Љц£° | ≥х“ї | ≥х“ї√вЈ—њќ≥ћЌ∆Љц£° |
| Єяґю | Єяґю√вЈ—њќ≥ћЌ∆Љц£° | ≥хґю | ≥хґю√вЈ—њќ≥ћЌ∆Љц£° |
| Єя»э | Єя»э√вЈ—њќ≥ћЌ∆Љц£° | ≥х»э | ≥х»э√вЈ—њќ≥ћЌ∆Љц£° |
њ∆ƒњ£ЇЄя÷–їѓ—І јі‘і£Ї≤їѕк ћв–Ќ£Їµ•—°ћв
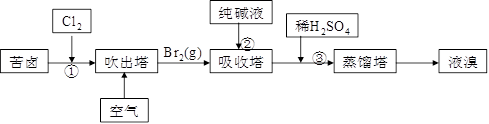
| A£Ѓ µ—й “÷∆NH3 | B£Ѓ≥э»•CO2÷–µƒHCl | C£ЃЉм—йK2CO3÷–µƒK£Ђ | D£Ѓ ’ЉѓNO |
≤йњіір∞ЄЇЌљвќц>>
њ∆ƒњ£ЇЄя÷–їѓ—І јі‘і£Ї≤їѕк ћв–Ќ£Їµ•—°ћв
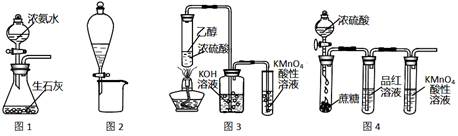
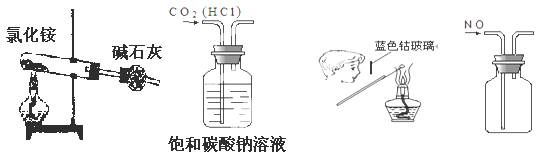
| A£ЃЌЉ1Ћщ Њ„∞÷√њ…÷∆»°∞±∆ш |
| B£ЃЌЉ2Ћщ Њ„∞÷√њ…Ј÷јлCH3CH2OHЇЌCH3COOC2H5їмЇѕ“Ї |
| C£ЃЌЉ3Ћщ Њ„∞÷√њ…÷∆»°““ѕ©≤Ґ—й÷§∆д“„±ї—хїѓ |
| D£ЃЌЉ4Ћщ Њ„∞÷√њ…Ћµ√ч≈®H2SO4Њя”–Ќ—ЋЃ–‘°Ґ«њ—хїѓ–‘£ђSO2Њя”–∆ѓ∞„–‘°Ґїє‘≠–‘ |
≤йњіір∞ЄЇЌљвќц>>
њ∆ƒњ£ЇЄя÷–їѓ—І јі‘і£Ї≤їѕк ћв–Ќ£Ї µ—йћв
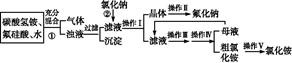
 °°°°NH4F+ °°°°H2SiO3°э+°°°°CO2°ь+°°°°°°°°
°°°°NH4F+ °°°°H2SiO3°э+°°°°CO2°ь+°°°°°°°° ≤йњіір∞ЄЇЌљвќц>>
њ∆ƒњ£ЇЄя÷–їѓ—І јі‘і£Ї≤їѕк ћв–Ќ£Ї µ—йћв
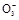 —хїѓ≥…I
—хїѓ≥…I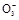 ,±їH2O2їтO2µ»—хїѓ≥…I2;
,±їH2O2їтO2µ»—хїѓ≥…I2;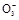 ƒ№±їHS
ƒ№±їHS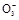 їє‘≠≥…I2°£
їє‘≠≥…I2°£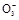 ÷–µƒƒƒ“ї÷÷°£
÷–µƒƒƒ“ї÷÷°£| µ—й≤љ÷и | ‘§∆Џѕ÷ѕу”лљб¬џ |
| ≤љ÷и1:»°…ўЅњ…ѕ ц»№“Ї„Ґ»л ‘є№÷–,µќ»лЉЄµќµнЈџ»№“Ї’сµі | »ф»№“Їѕ‘°°°°°°°°,‘тЉў…и1≥…ЅҐ;Јс‘т,Љў…и1≤ї≥…ЅҐ,‘ўљш––≤љ÷и2 |
| ≤љ÷и2: | »ф»№“Їѕ‘јґ…Ђ,‘тЉў…и2≥…ЅҐ,Јі”¶µƒјл„”Јљ≥ћ љќ™ °° ; Јс‘т,Љў…и2≤ї≥…ЅҐ,‘ўљш––≤љ÷и3 |
| ≤љ÷и3: | |
≤йњіір∞ЄЇЌљвќц>>
њ∆ƒњ£ЇЄя÷–їѓ—І јі‘і£Ї≤їѕк ћв–Ќ£Ї µ—йћв
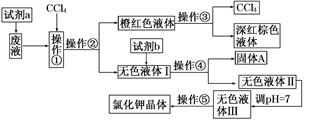
| A£ЃЁЌ»°°Ґєэ¬Ћ°ҐЈ÷“Ї°Ґєэ¬Ћ°Ґ’фЈҐљбЊІ |
| B£ЃЁЌ»°°ҐЈ÷“Ї°Ґ’фЅу°Ґєэ¬Ћ°Ґ’фЈҐљбЊІ |
| C£ЃЈ÷“Ї°ҐЁЌ»°°Ґєэ¬Ћ°Ґєэ¬Ћ°Ґ’фЈҐљбЊІ |
| D£ЃЁЌ»°°ҐЈ÷“Ї°ҐЈ÷“Ї°Ґєэ¬Ћ°Ґ’фЈҐљбЊІ |
≤йњіір∞ЄЇЌљвќц>>
њ∆ƒњ£ЇЄя÷–їѓ—І јі‘і£Ї≤їѕк ћв–Ќ£Їµ•—°ћв
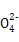 °ҐN
°ҐN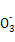 °ҐCl-÷–µƒ4÷÷јл„”,ЋщЇђјл„”µƒќп÷ µƒЅњЊщќ™1 mol°£»фѕтЄ√»№“Ї÷–Љ”»лєэЅњµƒѕ°ЅтЋб,”–∆ш≈Ё≤ъ…ъ,«“»№“Ї÷–“хјл„”÷÷ја≤ї±д(≤їњЉ¬«ЋЃµƒµзјлЇЌјл„”µƒЋЃљв)°£ѕ¬Ѕ–ЋµЈ®≤ї’э»Јµƒ «( )
°ҐCl-÷–µƒ4÷÷јл„”,ЋщЇђјл„”µƒќп÷ µƒЅњЊщќ™1 mol°£»фѕтЄ√»№“Ї÷–Љ”»лєэЅњµƒѕ°ЅтЋб,”–∆ш≈Ё≤ъ…ъ,«“»№“Ї÷–“хјл„”÷÷ја≤ї±д(≤їњЉ¬«ЋЃµƒµзјлЇЌјл„”µƒЋЃљв)°£ѕ¬Ѕ–ЋµЈ®≤ї’э»Јµƒ «( )| A£Ѓ»фѕтЄ√»№“Ї÷–Љ”»л„гЅњµƒNaOH»№“Ї,≥дЈ÷Јі”¶Їу,єэ¬Ћ°Ґѕіµ”°Ґ„∆…’,„о÷’Ћщµ√єћћеµƒ÷ Ѕњќ™72 g |
| B£Ѓ»фѕтЄ√»№“Ї÷–Љ”»лєэЅњµƒѕ°ЅтЋб,≤ъ…ъµƒ∆шће”цњ’∆шƒ№±д≥…Їм„Ў…Ђ |
| C£Ѓ»фѕтЄ√»№“Ї÷–Љ”»лєэЅњµƒѕ°ЅтЋбЇЌKSCN»№“Ї,»№“Їѕ‘—™Їм…Ђ |
| D£ЃЄ√»№“Ї÷–ЋщЇђµƒјл„” «:Fe2+°ҐNa+°ҐSO42-°ҐNO3- |
≤йњіір∞ЄЇЌљвќц>>
њ∆ƒњ£ЇЄя÷–їѓ—І јі‘і£Ї≤їѕк ћв–Ќ£Їµ•—°ћв
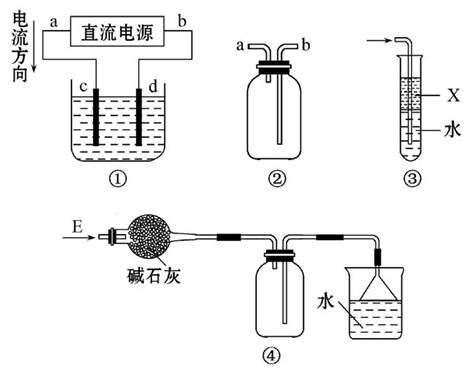
| A£Ѓ„∞÷√Ґў”√јіµзљв±•ЇЌ ≥—ќЋЃ£ђcµзЉЂ≤ъ…ъµƒ∆шћеƒ№ є ™»уµƒµнЈџKI ‘÷љ±дјґ |
| B£Ѓ„∞÷√ҐЏњ…”√”Џ ’ЉѓH2°ҐNH3°ҐCl2°ҐHCl°ҐNO2 |
| C£Ѓ„∞÷√Ґџ÷–Xќ™±љ£ђњ…”√”Џќь ’∞±∆шїт¬»∆ш |
| D£Ѓ„∞÷√Ґ№њ…”√”ЏЄ…‘п°Ґ ’Љѓ∞±∆ш£ђ≤Ґќь ’ґа”аµƒ∞±∆ш |
≤йњіір∞ЄЇЌљвќц>>
њ∆ƒњ£ЇЄя÷–їѓ—І јі‘і£Ї≤їѕк ћв–Ќ£Їћоњ’ћв

| ≥Ѕµнќп | Al£®OH£©3 | Fe£®OH£©3 | Fe£®OH£©2 | Mn£®OH£©2 | Cu£®OH£©2 |
| pH | 5.2 | 3.2 | 9.7 | 10.4 | 6.7 |
| ≥Ѕµнќп | Zn£®OH£©2 | CuS | ZnS | MnS | FeS |
| pH | 8.0 | £≠0.42 | 2.5 | 7 | 7 |
≤йњіір∞ЄЇЌљвќц>>
∞ўґ»÷¬–≈ - ЅЈѕ∞≤бЅ–±н - ‘ћвЅ–±н
Їю±± °ї•Ѕ™Ќшќ•Ј®ЇЌ≤їЅЉ–≈ѕҐЊў±®∆љћ® | Ќш…ѕ”–Ї¶–≈ѕҐЊў±®„®«ш | µз–≈’©∆≠Њў±®„®«ш | …жјъ Ј–йќё÷ч“е”–Ї¶–≈ѕҐЊў±®„®«ш | …ж∆у«÷»®Њў±®„®«ш
ќ•Ј®ЇЌ≤їЅЉ–≈ѕҐЊў±®µзї∞£Ї027-86699610 Њў±®” ѕд£Ї58377363@163.com