
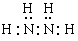 ��2��N2H4+2H2O2=N2+4H2O��3��N2H4+2HCl= N2H6Cl2��
��2��N2H4+2H2O2=N2+4H2O��3��N2H4+2HCl= N2H6Cl2��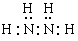 ����2��N2H4��Һ̬˫��ˮ��Ӧ�����������ֲ���Ⱦ��������̬���ʣ��÷�Ӧ�Ļ�ѧ����ʽΪN2H4+2H2O2=N2+4H2O����3���������⽫N2H4ͨ����������ʱ��������Ӧ�Ļ�ѧ����ʽΪN2H4+2HCl= N2H6Cl2��2H4�ķ���ʽ���ƶϡ��ṹ�����ʵ�֪ʶ��
����2��N2H4��Һ̬˫��ˮ��Ӧ�����������ֲ���Ⱦ��������̬���ʣ��÷�Ӧ�Ļ�ѧ����ʽΪN2H4+2H2O2=N2+4H2O����3���������⽫N2H4ͨ����������ʱ��������Ӧ�Ļ�ѧ����ʽΪN2H4+2HCl= N2H6Cl2��2H4�ķ���ʽ���ƶϡ��ṹ�����ʵ�֪ʶ��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��X�Ƿǽ���Ԫ�� |
| B��X��+2�ۻ������+4�ۻ������ȶ� |
| C��XO2����ǿ������ |
| D��X���ȶ�����̬�⻯�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| A��1.1 | B��1.2 | C��1.5 | D��1.8 |
�鿴�𰸺ͽ���>>
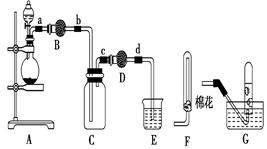
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
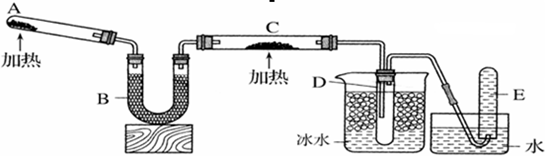
| A����Ӧ���������ͻ�ԭ�������ʵ���֮��Ϊ2��3 |
| B��װ��B�м�������ʿ����Ǽ�ʯ�һ���ˮ�Ȼ��� |
| C��װ��D��Һ�����ʹ����ĺ�ɫʯ����ֽ���� |
| D��Ϊ��ֹװ��D�з������������Ը��������ſ������ռ����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
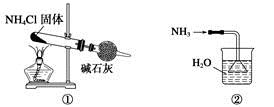
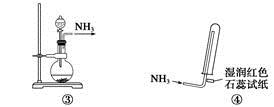
| A�����ǰ�������װ�� | B�����ǰ�������װ�� |
| C�����ǰ�������װ�� | D�����ǰ����ռ�������װ�� |
�鿴�𰸺ͽ���>>
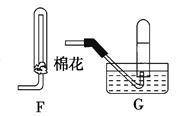
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

| ʵ����� | ���� |
| ��.��Bװ�����ƣ�ʹ̼�����ϡ����Ӵ� | �������� |
| ��.��Cװ���в�����ɫ����ʱ�����̽�Bװ������ | |
| ��.��Aװ����ͭ˿����ϡ�����У���Aװ������ | Aװ���в�����ɫ���� Eװ���п�ʼʱ����dz����ɫ���� |
| ��.��Fװ����Eװ���й������ | Eװ����������ɫ���� |
| ��.һ��ʱ��� | Cװ���а�ɫ�����ܽ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
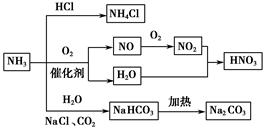
| A��NH4Cl��NaHCO3���dz��õĻ��� |
| B��NH4Cl��HNO3��Na2CO3����ʱ���ֽ� |
| C��NH3��NO2��һ�������¿ɷ���������ԭ��Ӧ |
| D��NH3�ϳ�NaHCO3�ĸ������ˮ��Һ������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A���μ�ϡNaOH��Һ����ʪ���ɫʯ����ֽ�����Թܿڣ���ֽ��������ԭ��Һ��һ����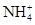 |
| B����װ��Fe��NO3��2��Һ���Թ��м���ϡH2SO4�����ڹܿڹ۲쵽����ɫ���� |
| C��������Fe���м���ϡHNO3����ַ�Ӧ����KSCN��Һ����Һ�ʺ�ɫ |
| D��46 g NO2��N2O4��������к���ԭ������Ϊ3NA�����еķ�������ΪNA |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com