
��ϩ�IJ���ͨ����������һ�����ҵ�ʯ�ͻ���ˮƽ������ϩΪ��Ҫԭ�Ϻϳ���Ҫ���л�������·������ͼ��ʾ����ش��������⡣
��1����ϩ�Ľṹ��ʽ___________������ʽ_______________��
��2��A�������������ŵ�������__________________��
��3����Ӧ�ڵķ�Ӧ������_______________________��
��4����Ӧ�۵Ļ�ѧ����ʽ��___________________________________��
��5������ϩΪԭ�Ͽɺϳ��л��߷��ӻ�����D����Ӧ�ٵĻ�ѧ����ʽ��
____________________________________________________________��
��6�����������У�����ͨ����ϩ�ӳɷ�Ӧ�õ�����_____������ţ���
a. CH3CH3 b. CH3CHCl2 c. CH3CH2Br
��10�֣���1��CH2=CH2��1�֣� 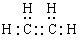 ��1�֣�
��1�֣�
��2���ǻ���1�֣���3���ӳɷ�Ӧ��1�֣�
��4��2CH3CH2OH+O2 2CH3CHO+ 2H2O��2�֣���д��������������1�֣�
2CH3CHO+ 2H2O��2�֣���д��������������1�֣�
��5��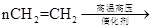
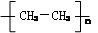 ��2�֣�д��һ�����������֣���д������1�֣�
��2�֣�д��һ�����������֣���д������1�֣�
��6��ac��2�֣���ѡΪ0�֣���ѡΪ1�֣�
���������������ϩ����̼̼˫�����ܷ����Ӿ۷�Ӧ�����ɾ���ϩ������D�Ǿ���ϩ����ϩҲ�ܺ�ˮ�����ӳɷ�Ӧ�����Ҵ�����A���Ҵ����Ҵ�����������Ӧ����B������B����ȩ����ȩ�����������ᡣ
��1����ϩ����̼̼˫�����ṹ��ʽ��CH2��CH2������ʽ��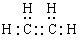 ��
��
��2��A���Ҵ������еĹ��������ǻ���
��3����Ӧ�����Ҵ���ˮ�����ӳɷ�Ӧ�����Ҵ����л���Ӧ�����Ǽӳɷ�Ӧ��
��4����Ӧ�����Ҵ��Ĵ�����������ȩ����Ӧ�Ļ�ѧ����ʽ��2CH3CH2OH��O2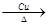 2CH3CHO��2H2O��
2CH3CHO��2H2O��
��5����Ӧ������ϩ�ļӾ۷�Ӧ����Ӧ�Ļ�ѧ����ʽ�� nCH2��CH2
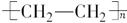 ��
��
��6����ϩ����̼̼˫���������������ӳɷ�Ӧ�������飬a��ȷ�����廯�ⷢ���ӳɷ�Ӧ���������飬c��ȷ��ѡ��b�е��л���������ԭ������ͬһ��̼ԭ���ϣ�������������ϩ�����ӳɷ�Ӧ�õ�����ѡac��
���㣺�����л�����ƶϡ��л���Ӧ���͡������ŵ��ж��Լ�����ʽ����д
�����������ǻ���������Ŀ��飬�漰ϩ���������������������ת�����ѶȲ����Ƕ��л�֪ʶ���ۺ����á��л��ƶ��Ǹ߿��еij������ͣ�ÿ��߿��бؿ�����������ij��Ӧ��ϢҪ��ѧ������Ӧ�ã��ܽϺõĿ��鿼�����Ķ�����ѧ������˼ά���������ȵ����ͣ���Ҫ�ص����ա�


 ���Ž�������С״Ԫϵ�д�
���Ž�������С״Ԫϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ϩ�����ںϳ�ɱ�������߳��ũҩ������ʽΪC3H5Br2Cl����Ӧ�ù㷺��DAP��֬��
��֪���봼�ɷ���������������Ӧ��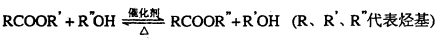
��1��ũҩC3H5Br2Cl������ÿ��̼ԭ���Ͼ�����±ԭ�ӡ�
A�Ľṹ��ʽ��__________________��A�����������ŵ�������____________________��
��ϩ��A�ķ�Ӧ������_______________��A��C3H5Br2CI�ķ�Ӧ������_____________��
��2��Aˮ��ɵõ�D����ˮ�ⷴӦ�Ļ�ѧ����ʽΪ��
_______________________________________________________________________.
��3��C�����ܶ�����ͬ״̬�¼����ܶȵ�6.25����S�и�Ԫ�ص����������ֱ�Ϊ��̼60%����8%����32%. S�Ľṹ��ʽΪ_________________________________��
��4������˵����ȷ����______________������ĸ���ţ���
a��C�ܷ����ۺϷ�Ӧ����ԭ��Ӧ��������Ӧ
b��C����2������������ͬ���칹����4��
c��D������IJ�����B������ͬ����Է�������
d��E���з�����ζ���������Ҵ�
��5��E��ˮ����ᆳ�������յõ��״���B�����߾���ѭ��������DAP��֬���Ʊ������н��״���H����IJ���������__________________��
��6��F�ķ���ʽΪC10H10O4. ��DAP����Ϊ���Ķ�Ԫȡ���������ȡ���������ڶ�λ���õ��屽���ϵ�һ��ȡ����ֻ�����֡�����D��F��Ӧ����DAP����Ļ�ѧ����ʽΪ��
_______________________________________________________________________.
��7��ʵ������2-�����Ʊ���ϩʱ������������SO2�� CO2��ˮ������ijͬѧ�������Լ�
�������������壬�������ͨ���Լ���˳����_______________������ţ���
�ٱ���Na2SO3��Һ������KMnO4��Һ ��ʯ��ˮ ����ˮCuSO4��Ʒ����Һ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪����CHO+(C6H5)3P=CH��R ��CH=CH��R + (C6H5)3P=O��R����ԭ�ӻ�ԭ���ţ�W��һ���л��ϳ��м��壬�ṹ��ʽΪ��HOOC��CH=CH��CH=CH��COOH����ϳɷ������£�
���У� �ֱ����һ���л���ϳɹ�������������ͷ�Ӧ��������ȥ��
�ֱ����һ���л���ϳɹ�������������ͷ�Ӧ��������ȥ��
X��W��һ�������·�Ӧ����������N��N����Է�������Ϊ168��
��ش��������⣺
��1��W�ܷ�����Ӧ�������� ������д��ĸ��ţ�
| A��ȡ����Ӧ | B��ˮ�ⷴӦ | C��������Ӧ | D���ӳɷ�Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
A��B��Ϊ��Ҫ���л�����ԭ�ϡ�
��֪��

��ش��������⣺
��1��A�����ں��б������������� ��
��2��B������ֻ��һ����ԭ�ӣ�2 mol B��Ӧ����1 mol C�Ļ�ѧ����ʽ�ǣ�
��
��3��B��һ��ͬ���칹���ܷ���������Ӧ���÷�Ӧ�Ļ�ѧ����ʽ�ǣ�
��
��4��D��������3��������ṹ��ʽ�� ��
��5��F�Ľṹ��ʽ�� ��F��L��Ӧ����G�Ǽӳɷ�Ӧ��L�Ľṹ��ʽ ��
��6��A��B��Ӧ����J�Ļ�ѧ����ʽ�� ��
��7��K�����ں�����Ԫ�����ɷ���ˮ�ⷴӦ����ṹ��ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
A-G��Ϊ�л����������AΪ��±��������Է�������Ϊ216��̼����������Ϊ22.2%�����ת����ϵ���£�
��֪��
��ش�
��1��C�ĺ˴Ź���������_______�����շ塣
��2���١��ܵķ�Ӧ���ͷֱ�Ϊ________��______��
��3�����й���F��˵����ȷ����_______ (��ѡ����ĸ����
a��1 mol F�����������Ʒ�Ӧ�������2 mol H2
b��1 mol F��ȫȼ������8.5 mol O2
c����������Cu(OH)2��Ӧ����ש��ɫ����
d������NaHCO3��Ӧ����CO2
��4��д�����з�Ӧ�Ļ�ѧ����ʽ
��Ӧ��___________________________________;
��Ӧ��___________________________________
��5����������������ͬ���칹����_______�֣���д����һ�ֵĽṹ��ʽ_______��
i����E��Ϊͬϵ�� ii����Է���������E��28
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������ʳΪ�죬ʳ��Ϊ�ȡ���ʳƷ��ȫһֱ������ע�Ļ��⡣������������( )��ȥ���������������������Ŀǰ�ѱ����á��ϳɹ��������������������£�
)��ȥ���������������������Ŀǰ�ѱ����á��ϳɹ��������������������£�
��ش��������⣺
��1���١��ڵķ�Ӧ���ͷֱ���______��______��
��2��A�Ľṹ��ʽΪ______��
��3��д����Ӧ�۵Ļ�ѧ����ʽ______��
��4��д�����ַ�������Ҫ��Ĺ�����������ͬ���칹��Ľṹ��ʽ��____________��
�ٷ�����ֻһ�ֺ��������ţ��ں˴Ź���������3�ַ壬���ź�ǿ��֮��Ϊ1:2:2��
��5�����������Ա���ϩ��1��3������ϩ��һ�������¹��ۺϳɵģ�д���÷�Ӧ�Ļ�ѧ����ʽ__________________��
��6���Ա���ϩΪԭ�ϣ�������ѡ������ƺϳ�| �����̡�ʾ�����£�
�����̡�ʾ�����£�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����3���������A��CH2=CH2�� B��CH3COOH�� C��
��1��������A��B�еĹ����ŵ����Ʒֱ��� �� ��
��2����ʹ������Ȼ�̼��Һ��ɫ��Ӧ�Ļ�ѧ����ʽΪ�� ��
�÷�Ӧ����Ϊ�� ��
��3�������Ҵ�����������Ӧ�Ļ�ѧ����ʽΪ�� ��
��4�� ��Ũ���������£���Ũ���ᷴӦ�Ļ�ѧ����ʽΪ�� ��
��Ũ���������£���Ũ���ᷴӦ�Ļ�ѧ����ʽΪ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
(17��)��ŵ����������ʹ,��Ҫ�������ʪ�Թؽ��ס���ð��ͷʹ�����ơ���ŵ�����ɻ������--��˾ƥ�ֺͻ������---����Ϣʹ�ϳɡ���ϳ�·�����£�
��֪��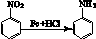
��1��������A��FeCl3������ɫ��Ӧ����Է�������С��100�� A�ķ���ʽΪ ��
��2�����������ʾB�к���C-H����O-H����C-O����C=O���ĺ������շ壬��B�б�����������ȡ������B�Ľṹ��ʽΪ ��
��3��E�Ĺ����������� ����Ӧ��--���в�����ȡ����Ӧ���� ��
��4��1 mol�������������NaOH��Һ���ȣ��������NaOH mol��
��5��д��C���������Ļ�ѧ����ʽ ��
��6���������������Ļ�������ͬ���칹���� �֡�
�� ���ڷ����廯����
�ں���-NH2��-COOH������-CH3
д�����к���һ��ȡ������ͬ���칹��һ�����������ɸ߾��ﻯѧ����ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����ʵ��װ�û������ȷ����
| A��ͼ�ף���ȥCl2�л��е�HCl | B��ͼ�ң���Ũ��������һ��Ũ�ȵ�ϡ���� |
| C��ͼ���������������ƹ��� | D��ͼ����ʵ�����Ʊ����� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com