
���� ��1��������ƽʹ�õ���ȷ��������
��2����������һ�����ʵ���Ũ����Һ�����Ʋ���ѡ����Ҫ������©��IJ��裻
��3���������������ʵ����ʵ�������Һ�����Ӱ�죬����C=$\frac{n}{V}$������������
��� �⣺��1����ƽ����ȷʹ�ò��裺a����ƽ�ŵ�ˮƽ̨�ϣ������Ƶ������˵���̶ȣ�b������ƽ��ƽ����ĸʹָ��ָ���ֶ��̵����룬������ƫת�ĸ�����ͬ��c���������ƽ�����̣����������ƽ�����̣������������ڱ���ϵ�λ�ã�ֱ�������ָ�ƽ�⣬Ҫ�����Ӽ�ȡ���룬Ҫ������ţ�Ҫ�Ӵ�С��d���������=���������+�����Ӧ�Ŀ̶ȣ�e�������ģ�������ȷ��˳��Ϊ��B C F A E D B��
�ʴ�Ϊ��C F E D B��
��2������һ�����ʵ���Ũ����Һ�IJ������裺���㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȣ��ܽ�ʱ������Ӧ�����ձ��У��ò��������Ͻ��裻
�ʴ�Ϊ���ձ���������������ƿ��ϴ�ӡ����ݡ�ҡ�ȣ�
��3��A����������������ϣ�BaCl2���������Ͻ��г��������³�ȡ������ƫС�����ʵ����ʵ���ƫС����Һ��Ũ��ƫ�ͣ���Aѡ��
B��ѡ�õ�����ƿ������������ˮ�������ʵ����ʵ�������Һ��������������Ӱ�죬��Һ��Ũ�Ȳ���Ӱ�죬��B��ѡ��
C������ҡ�Ⱥ�Һ���½����ּ�ˮ���̶��ߣ�������Һ�����ƫ����Һ��Ũ��ƫ�ͣ���Cѡ��
D���������ƹ����У�����ƿ�����ᵼ����Һ��ϲ����ȣ���Һ���������ƫ���ƫС����Һ��Ũ�ȿ���ƫ�ͻ�ƫ�ߣ���D��ѡ��
��ѡAC��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���Ϥ��ƽʹ�÷���������һ�����ʵ���Ũ����Һ�����ǽ���ؼ���ע���������ķ����ͼ��ɣ���Ŀ�Ѷ��еȣ�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
| A�� | CO2 | B�� | MgCl2 | C�� | Na2O2 | D�� | NaOH |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
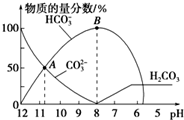
| A�� | ��A����ʾ����Һ�У�c��Na+��+c��H+��=2c��CO32-��+c��HCO3-��+c��OH-�� | |
| B�� | ��B����ʾ����Һ�У�Ũ������������Na+ | |
| C�� | ����Һ��pHΪ7ʱ����Һ�����������20mL | |
| D�� | 25��ʱ��CO32-ˮ�ⳣ��Kh=2��10-4mol•L-1������Һ��c��HCO3-����c��CO32-��=2��1ʱ����Һ��pH=10 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ���߶�Ӧ���������������ˮ | |
| B�� | ���ߵĵ��ʷ����ڿ����о�ֻ���������� | |
| C�� | �Ʊ�AlCl3��FeCl3��CuCl2�����ܲ��ý���Һֱ�����ɵķ��� | |
| D�� | ���AlCl3��FeCl3��CuCl2�Ļ����Һʱ��������������Cu��Fe��Al |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �ڢۢߢ��� | B�� | �٢ڢۢݢ�� | C�� | �ڢݢޢߢ� | D�� | �ڢܢߢ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ���й��ۼ��Ļ�����һ��Ϊ���ۻ����� | |
| B�� | �������Ӽ��Ļ�����һ��Ϊ���ӻ����� | |
| C�� | ���ȷ�Ӧ�У��ɻ�ѧ�������������������ȷ�Ӧ�У��ɻ�ѧ�����ѷų����� | |
| D�� | ���ȷ�Ӧ�У��»�ѧ���γ��������������ȷ�Ӧ�У��»�ѧ���γɷų����� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com